Q: Write the mass balance for a K2CO3 solution if the species in the solution are K+, CO32, HCO3,…
A: Mass balance equation based on the law of conservation of mass.According to this all the species…
Q: Show how the dehalogenation of (1R,2R)-1,2-dibromo-1,2-diphenyl ethane with zinc affords…
A:
Q: 2N2(g) + O2(g) 2N₂O(g) 5th attempt Part 1 Use thermodynamic data to calculate the Kp for the…
A: The objective of the question is to calculate the equilibrium constant (Kp) for the given reaction…
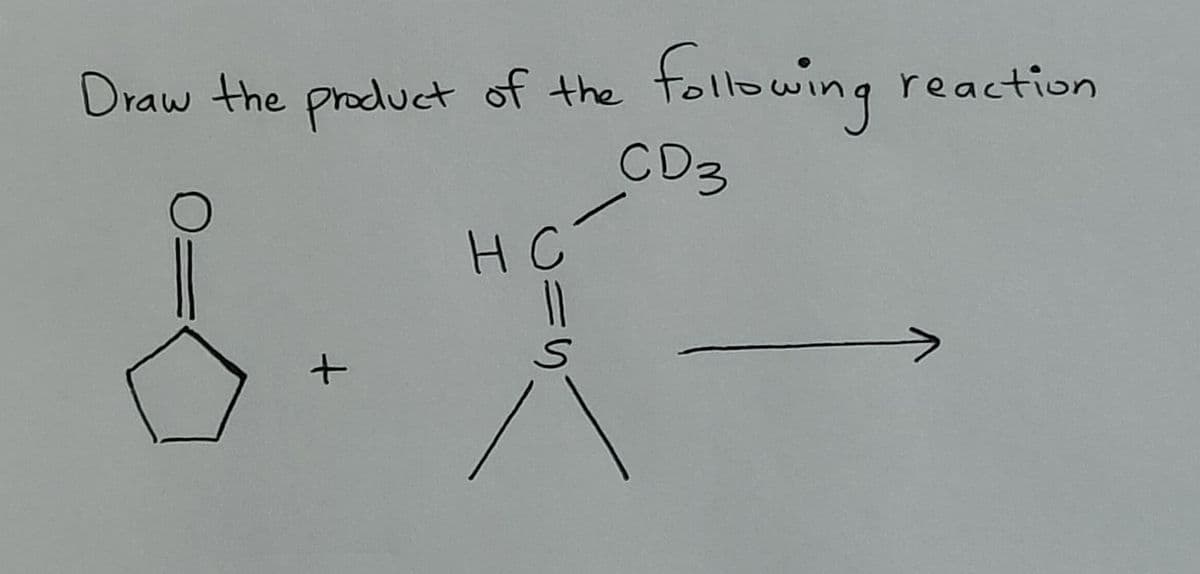
Q: Drawn out the fragmentation processes of the three alcohols below. + A. H-O-CH2 CH2 CH2 CH 3 B. H-O+…
A: In case of alcohols, the fragmentation takes place at the C-C bond which is adjacent to the-OH group…
Q: The rate constant for a certain reaction is k = 5.10×10−3 s−1 . If the initial reactant…
A: The objective of the question is to find the concentration of the reactant after 20.0 minutes given…
Q: A chemist measures the enthalpy change AH during the following reaction: 2 KCl(s) + 302(g) →2…
A: 2KCl(s) + 3O2(g) ----> 2KClO3(s) ∆H = 78. kJreaction∆H2KClO3(s) --> 2KCl(s) +…
Q: Which of the following would you expect to NOT undergo a Jahn-Teller distortion? Select all that…
A: Each of the given compounds is an octahedral complex. According to CFT, the orbital splitting…
Q: When we were doing the GC analysis of our product from dehydrobromination of 2- bromobutane, we…
A: Gas chromatography (GC) is a technique used in analytical chemistry to separate and analyze…
Q: How many planar nodes does this orbital have? Z y X
A: Answer:The nodal plane is a region in an orbital where probability of finding an electron is zero.
Q: 8. Use the following reactions and Draw the transition state in the following reactions: CH3 CH3 a)…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Two adjacent polyester molecules are shown below. List and describe the factors that influence the…
A: Transition temperature is the temperature at which an amorphous polymer changes from a hard/glassy…
Q: The osmotic pressure of a saturated solution of CuBr at 25°C is 3 torr. What is the solubility…
A: The objective of the question is to calculate the solubility product of for the given conditions.
Q: 5) A photon of energy 12.1 eV absorbed by hydrogen atom, originally in the ground state, raises the…
A: The photon energy given is = 12.1 eVSo, the energy is = -13.6eVThe quantum number of the state is :
Q: (A)Write the Hückel Hamiltonian matrix for benzene. (B)The pictures below represent a top view of…
A: The objective of the question is to write the Huckel Hamiltonian matrix for benzene and to label the…
Q: When 173. mg of a certain molecular compound X are dissolved in 100 g of cyclohexane (C6H12), the…
A: Answer:For a non-electrolyte and non-volatile solute dissolved in a solvent, depression in freezing…
Q: What are the effects of aerosols on climate change?
A: An aerosol is a colloid of fine solid particles or liquid droplets, in air or another gas.
Q: 1. Provide the mechanism. NaOEt EtOH CO₂Et
A: Given,The reaction:
Q: Draw the simplest curved arrow mechanism possible for the reaction shown below. You may need to…
A: Given is organic reaction. given starting compound is secondary carbocation. The given product is…
Q: the energy diagram below? O O O S2 E1 SN1 E2 Free energy (G) Transition states D Intermediates…
A: SN1A nucleophilic substitution reaction that occurs in two steps. The leaving group departs first,…
Q: 1. CrO, H₂SO4 2. Neutralizing work-up H
A: In the above given reaction an aldehyde is treated with CrO3 , H2SO4 followed by the neutralizing…
Q: The enolate of an ester can be treated with a ketone to give a beta-hydroxy ester. Draw a mechanism…
A:
Q: What is the major organic product of the following reaction? NH AICI NH HN HN HN IV
A: alkyl group can be added by an electrophillic aromatic substitution reaction called the…
Q: P1A.12* Balloons are still used to deploy sensors that monitor meteorological phenomena and the…
A: (a) Volume of balloon is:
Q: What reagents are required for the following transformation? ? H2O / H+ ○ H2O ○ H2O / -OH O-OH OH
A: We know, hydration of alkene produces alcohol, it is done in the presence of an acid and water. The…
Q: In an experiment, a 1.3440 g sample of dimethyl oxalate (C4H6 O4) is burned completely in a bomb…
A: The objective of this question is to calculate the molar heat of combustion of dimethyl oxalate…
Q: What can you predict about the stability of the structures shown? = OI is predicted to be more…
A: Stability is simply a measure of energy. Lower energy molecules are more stable than higher energy…
Q: Give detailed Solution..show work..don't give Handwritten answer..don't use Ai for answering this
A: The objective of this question is to calculate the amount of energy required to heat a certain…
Q: VO 4^-3 + C2 O 4^-2 ---->VO ^+1 + CO 2 (acidic) Solve and balance this redox equation
A: The objective of this question is to balance the given redox equation in an acidic medium. The redox…
Q: Calculate the pH when 20.0 mL of 0.150 M KOH is mixed with 26.0 mL o 0.300 M HBrO (Ka = 2.5 × 109)
A:
Q: Design a synthesis of pent-2-yne from trans-pent-2-ene. Complete the following reaction scheme. Step…
A: The objective of this question is to determine the structure of compound A and to identify the…
Q: Select the best reagents for each of the following transformations. You should only use each set of…
A: 1.) G2.) C3.)I4.)BExplanation:1.) this is a Baeyer Villiger reaction in which aldehyde/ketone…
Q: Cyclopentadienone is a highly reactive ketone. Draw an anti- aromatic resonance structure that…
A: The compound should have internal conjugation with a total no. of 4n pi electrons.Anti aromatic is…
Q: Propose a synthesis for the following molecules, starting from benzene or a named varian
A: The objective of this question is to propose syntheses for specific molecules starting from benzene…
Q: The hydrocarbon whose formula is shown was subjected to complete hydrogenation. Use the given data…
A: The objective of the question is to determine the number of rings and pi bonds in the reactant…
Q: What is the highest-energy intermediate in the reaction profile below? Transition states OE OF OD O…
A: The state corresponding to highest potential energy.
Q: Which of these reactions is less likely to produce an ester? .d A 。༼ ོ་ཡིན་བ B PhCOOOH H₂SO4,…
A: We have to choose the correct option.
Q: %Transmittance 100 M 4000 'H NMR 300 MHz -3344 2958 2931 2873 3000 2000 Wavenumber (cm-1) -1466…
A: The objective of the question is to analyze the given spectrum and predict the structure of the…
Q: How many different types of carbons does the following compound have?
A: 13C NMR spectroscopy is used to determine the number of distinct carbon atoms in a molecule and the…
Q: 4. Extraction: a) ( ) In what order (from lowest to highest R,) would you expect to find compound X,…
A: Thin layer chromatography (TLC) is a method of separating components of the mixture. It is based on…
Q: 1. Predict the major organic product for the following reaction shown. Include stereochemistry when…
A: This is an example of Diels-alder reaction. In this conjugated diene undergoes [4+2]cycloaddition…
Q: Please help, i need fast answer. don't provide handwritten solution ....
A: The objective of this question is to calculate the molar mass of a molecular compound X, given the…
Q: 10. gas is at a pressure of 5.09 MPa and temperature of 325 K. Determine Nitrous Oxide the…
A: The pressure is given as P = 5.09 MPatemperature T = 325 K
Q: 6. ( ) A 40 mL sample of a 0.2 M RNHz (kb = 3.7 x 10") is titrated with 0.2 M HC the pH after the…
A:
Q: What MS resolving power is needed to fully resolve the C8H8NO2 and C7H6N2O2 ions (at the base…
A: The objective of the question is to determine the resolving power needed to fully mass resolve the…
Q: unknown acid #: 28 Mass of unknown acid: 0.300g Molarity of NaOH: 0.1002 g Volume of NaOH at…
A: Titration is commonly used to measure and adjust the pH of solution. Acid-base titrations, in…
Q: The following is a Hill Plot for a receptor-ligand interaction: 8 6 (0-1) 601 4 2 0 -2 -4 -6 -6 4 -2…
A: Given:Hill plot for receptor-ligand interaction.Determine the fold difference.
Q: Provide the mechanism. NaOEt EtOH CO₂Et
A: This is an example of Robinson annulation reaction which is acombination of two reactions, Michael…
Q: Draw the organic product you would expect to isolate from the nucleophilic substitution reaction…
A: Nucleophilic substitution is a fundamental class of organic reactions in which a leaving group is…
Q: An increase in temperature will always cause AG to: Incorrect increase. change from negative to…
A: decrease. Explanation:An increase in temperature will not always cause ΔG to behave in a single…
Q: Draw the organic product you would expect to isolate from the nucleophilic substitution reaction…
A: Given reaction is an example of nucleophilic substitution reaction.The substitution reaction is a…
Please help with the following
Step by step
Solved in 1 steps with 2 images

- A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the Lindlar catalyst, 1 equivalent of Η2 is absorbed. On hydrogenation overa palladium catalyst, 3 equivalents of Η2 are absorbed. (a) How many degrees of unsaturation are present in the unknown structure? (b) How many triple bonds are Present? (c) How many double bonds are present? (d) How many rings are present? (e) Draw a structure that fits the data.Rank the leaving groups below from worst to best. -F -CH3 -Br -I -OHDraw HOCH2CH(CH3)2. Need answer fast i'll rate up
- Identify the organic functional group(s) of the reactant, the reaction type, and predict the functional group(s) of the product(s) then draw the major product(s) The reactant is:a. deoxytetroseb. alcohol pentosec. ketohexosed. aldotriosee. aldopentoseThe reaction type is:a. acetal formationb. oxidation (benedict's)c. hemiacetal formationd. acetal hydrolysise. reduction (hydrogenation)f. mutarotationThe product should be:a. alpha 1-4 disaccharideb. deoxyhexosec. carboxylic acid pentosed. alcohol pentosee. alpha pyranosef. no reaction56. If a trans alkene is reacted with OsO4 in H2S, the product is: Meso compound Hoffman product Markovnikov product Racemic mixtureIs THF a polar protic or aprotic?



