Q: What would be the best starting material for the following chemical conversion? 1. CH3COCl in…
A: Introduction :- O-nitroaniline is orange solid. It has melting point 72 °C…
Q: C b. :O:
A: •RESONATING STRUCTURE:- Resonating structure describe about the delocalization of electron in…
Q: LOD 5D METHANOL CH40 D 4000 3347 8 3336 8 2945 18 2833 22 2522 77 2046 84 1460 47 3000 1116 62 1030…
A:
Q: Give the major organic product(s) of the following reactions or sequences of reactions. Show all…
A:
Q: need help on the following with explanation Please do both correctly neat and clean otherwise don't…
A: (1) German chemist and physicist Erich Hückel proposed a theory to help determine if a planar ring…
Q: Cuien H₂₂ (g) + F₂ (g) = 2HF (g) Find the partial pressure of HF? Crien An empty Steel Container is…
A:
Q: All chemical dangers in the workplace are addressed under the: A) Formaldehyde Standard B)…
A: All chemical are dangerous at the workplace, there for they monitored under some conditions and…
Q: a 0.4815 g sample of potassium iodide yields 0.7562 g of silver iodide gravimetrically, determine…
A: Mass of potassium iodide = 0.4815 g
Q: Check all of the compounds in the list below that are constitutional isomers. (Select all that…
A: Constitutional isomers are compounds having same molecular formula but different atom to atom…
Q: Show the full reaction mechanism and include its name
A:
Q: excess CH₂Br
A:
Q: If 50.0 mL of 0.435 M NaCl solution is diluted to 225.0 mL, what is the concentration of the diluted…
A: Given data is as follows: The initial volume of NaCl solution V1= 50.0 mL The initial concentration…
Q: Carbonic anhydrase is one of the fastest enzymes known. If 10 µg of this enzyme catalyzes the…
A:
Q: Based on their formulas, classify each of the following in the solid state as a molecular, ionic,…
A:
Q: Concerning the following alcohol, which one of the following statement is incorrect? OH This…
A: According to Zeitsev;s rule, during dehydration of an alcohol, a more substituted alkene is formed…
Q: Either the reactant (X) or the major organic product is missing from the equation below. Draw the…
A:
Q: Synthesize the following compounds from benzene. O CI OH OH NO₂ CI NO₂
A: Since you have posted multiple questions, as per guidelines we will solve the first two parts.please…
Q: Refer to the following ion of oxygen to answer the question(s) below. 2 e 8 e Reference: Ref 2-4 Oo…
A:
Q: 13. Propose an effective 3-step synthesis for the following transformation using the given reagents.…
A:
Q: Activity 3 Here is another Type 2 problem. The bullet points “walk you through” how to do it. …
A:
Q: Calculate the molarity of the two solutions. The first solution contains 0.200 mol of NaOH in 1.65 L…
A: Molarity is defined as the number of moles of solute present in per litre of solution. It is denoted…
Q: Using your half-reactions, write the balanced redox reaction in the data table above. show the work…
A: Given: Ecell of : Cu/Fe 0.450V Fe/Zn 0.475V Zn/Al 0.404V
Q: O 0841100 O 1021 g O 0045330 O 5.9090-5 g How many grams of tin metal will be plated out in an…
A: Given that - Ampere current = 4.308 A Time = 317.4 second We know that during electrolysis of tin…
Q: What is the [OH-] in a solution that has a [H3O+] = 1.0 × 10-8 M?
A:
Q: What is the [OH-] in a solution that has a [H3O+] = 1.0 × 10-5 M?
A: Given information is as follows: The concentration of H3O+ = 1.0×10-5 M The concentration of OH- = ?…
Q: Which compound of the following structures is aromatic? HH I II :0 III IV
A:
Q: Which reaction below would result in a net decrease in entropy?
A:
Q: 14.3 mL of a 2.5 molar nitric acid (HNO3) solution was needed to neutralize a 20 mL solution of…
A:
Q: For the following reaction, indicate the false statement. HCIO3 + F CIO3 + HF F" acts like a proton…
A: The false statement is Option e) HClO3 and F- are conjugate base pairs
Q: What is the [HF] in a solution with a pOH of 12.5? the answer is not listed 3.16 x 10:13 M 0.0316 M…
A:
Q: Consider the reaction scheme below. N Br₂ FeBr3 NaOH warm X AICI 3 Y CI
A:
Q: Students were responsible for conducting 4 trials of titration of 15 mL samples of phosphoric acid…
A: Using the stoichiometry i.e. the mole ratio of Sr(OH)2 to H3PO4 , concentration of H3PO4 can be…
Q: Nitration of methyl benzoate only occurs in presence of a Lewis acid. True False
A: We need to discuss nitration of methyl benzoate.
Q: Ribose, a carbohydrate with the formula shown, forms a cyclic hemiacetal, which, in principle, could…
A:
Q: Provide the major product of the SO₂H Cl₂ AICI
A: Given reaction is halogenation reaction. Cl substitutes ring hydrogen.
Q: In the laboratory a student finds that it takes 214 Joules to increase the temperature of 13.3 grams…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: 2. a) Calculate the pH of 0.12 mol/L ethanoic acid (K₂ = 1.7 x 105 mol/L). b) Calculate the mass of…
A: Since the given question is a multipart question, hence I solved first three parts according to…
Q: CUCN OR SN/HCI AIC (2) SNHC options. 80212 CCN Cent KMnOt → B HNO3 H2804 Zn(Hg) Hel Brzihn <CH₂H…
A:
Q: estion 51 Which of the following occurs during Fischer esterification? O a. The -OH from the…
A:
Q: What is the structure of compound C in the following chemical transformation? a) Br 50 O Compound A…
A: Given: Reactant is benzene. The reaction is a multistep reaction.
Q: Is this compound aromatic? Explain why?
A: We need to determine if the given compound is aromatic or not.
Q: A common demonstration in chemistry courses involves adding a tiny speck of manganese(IV) oxide to a…
A: In Decomposition reactions chemical species break up into simpler molecules. At very high…
Q: 3. Fill in the box with the major organic product of the first step of the reaction shown? xs CH₂l…
A: Here we are required to predict the product of the reaction
Q: CH3 H HCI CH3 H Epoxides are cleaved by acid as are other ethers, but milder conditions are…
A: There are two reactants - an epoxide and Hydrochloric acid.
Q: edu.co D- and L- designations are used to distinguish between the two possible enantiomers of the…
A:
Q: bject) ftantilludel.edux Mail-Francesca A Tantillo-Out x START HERE CHM150-251 Che X C app.101edu.co…
A: For any phase to exist in equilibrium, the point at the phase diagram should coincide between all…
Q: :0: :O: ز - ل :O:
A: The resonance hybrid is the intermediate of contributing resonating structutes.
Q: Using the molecular orbital model, identify the correct (valence) electron configurations for the…
A: Dear student , since you have posted multiple questions we will allow to solve only first question…
Q: type of radiation red light A few different types of electromagnetic radiation are listed in the…
A: In the electromagnetic spectrum radiations are arranged in the order of decreasing wavelength and…
Q: Indicate which factors affect the rate of a reaction. Select all that apply x Keq oooo k E₂…
A: Rate of reaction : It is the speed at ease of which reactant are converted to products or it is the…
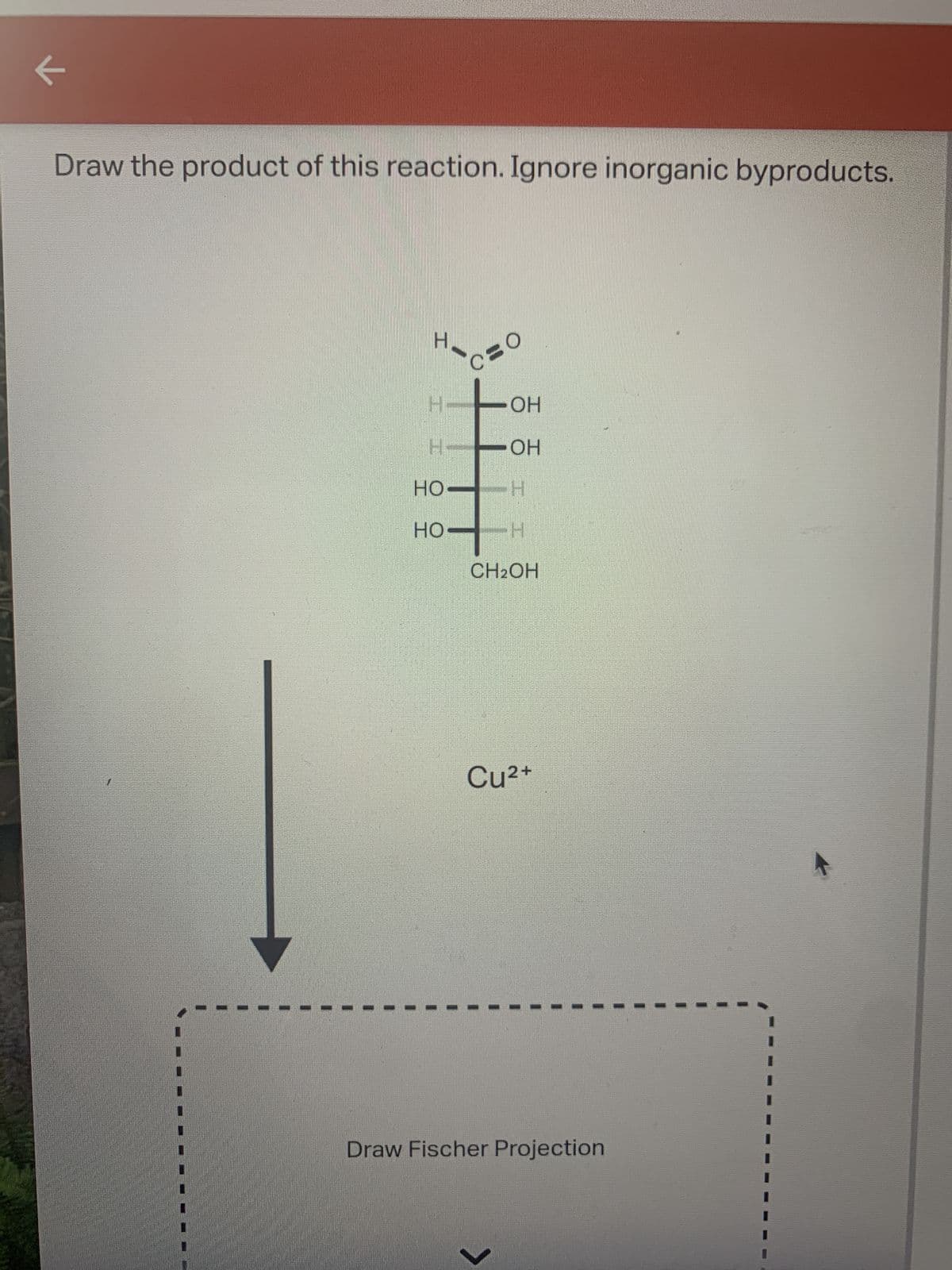
Step by step
Solved in 2 steps with 2 images

- Draw and write only the main product of the reaction (C) Only typed solutionDraw the organic product(s) formed when CH3CH2CH2OH is treated withfollowing reagent : [1] NaH; [2] CH3CH2BrClassify the reaction type. a. oxidation (Benedict's) b. reduction (hydrogenation) c. acetal formation d. hemiacetal hydrolysis e. acetal hydrolysis f. mutarotation g. hemiacetal formation
- Draw the major product of this reaction. Ignore inorganic byproducts. PCC, CH2Cl2Draw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH c.HCl + ZnCl2 d.HBr e.SOCl2, pyridine f.PBr3 g.TsCl, pyridine h. [1] NaH; [2] CH3CH2Br [1] i.TsCl, pyridine; [2] NaSH j.POCl3, pyridineDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. Ag2O, NH4OH

