Q: A specimen of copper having a rectangular cross section 10 mm x 15 mm and is pulled in tension with…
A: When a material is loaded, internal stress is created inside the material and material gets deformed…
Q: 4. Balanced Chemical Equation Reaction Type: At completion of reactions: Grams of zinc metal Grams…
A: A single replacement reaction is defined as a reaction in which one element is substituted by…
Q: If an acetic acid in acetate buffer is 0.022 M in acetic acid and has a pH of 5.01 , what is the…
A: PH of acidic buffer can be determained by Henderson equation PH = Pka + log (salt / acid)
Q: 3. Calculate the pH of a buffer which is 0.2 mol/L with respect to ammonium sulphate and 0.1 mol/L…
A: Given -> Concentration of ammonium sulphate= 0.2 mole/L Concentration of NH3 = 0.1 mole/L Ka of…
Q: A chemist prepares a solution of copper(II) sulfate (CuSO4) by measuring out 5.4 μmol of copper(II)…
A: The given solution contains: The amount of solute is = 5.4 μmol The volume of the solution is = 200.…
Q: Provide IUPAC names for the following compounds. (a) OH (b)
A: We follow the below steps designed by IUPAC to assign IUPAC name to an organic molecule.
Q: Methyl salicylate (sp gr = 1.167) 60 mL Lidocaine 4% (w/w) Tween:Span mix (1:4 mix) Polypeg Base 10…
A: Since you have asked a question with multiple subparts, we will answer only 3 subparts for you. In…
Q: What is the [OH-] in a solution that has a [H3O+] = 1.0 × 10-5 M?
A: Given information is as follows: The concentration of H3O+ = 1.0×10-5 M The concentration of OH- = ?…
Q: Technetium-99m is a radioactive isotope that is widely used in medical imagining. Tc-99m decays to…
A:
Q: Which qualitative analysis test from this module can help distinguish between different kinds of…
A:
Q: Wt of precipitate (Silver chloride) MW of silver nitrate MW of silver chloride Meg silver nitrate…
A:
Q: b) In another experiment, the following equilibrium was established: 2 NO(g) + Cl₂(g) = 2 NOCI(g)…
A: Given an equilibrium reaction and initial moles of reactants is follows;2NO(g)+Cl2(g)⇌2NOCl(g)at…
Q: I ift ↑ Qlaccds X tab ΤΟΝ C caps lock H fn esc Re → O CHEMICAL REACTIONS Calculating molarity using…
A:
Q: A reaction has a AH° = 25 kJ/mol and a AS° = -43 J/K at 298 K. Calculate the equilibrium constant…
A:
Q: What descriptive term is applied to the type of diene represented by 1,5-octadiene ? O alkynyl diene…
A: Each type of diene is represented by the position of two double bonds present in the compound. a.…
Q: Reaction between ammonia and oxygen. Amount of nitrogen monoxide produced if 0.5g of ammonia is…
A:
Q: 4 The Haworth projection of D-glucose is shown. What type of cyclic sugar is formed? 4 6 CH₂OH 5 OH…
A: All common monosaccharides exists in two forms, either six membered cyclic hemiacetals or hemiketals…
Q: compound hydrogen bromide Br. bromine CH₂CL₂ dichloromethane H₂2₂ intermolecular forces (check all…
A: Intermolecular forces are the forces of attraction and repulsion between molecules that hold…
Q: An unknown substance is found to be soluble in water and gives a positive 2,4- DNP test. If you…
A: 2,4-DNP test is a chemical test which is performed to determine whether a compound contains aldehyde…
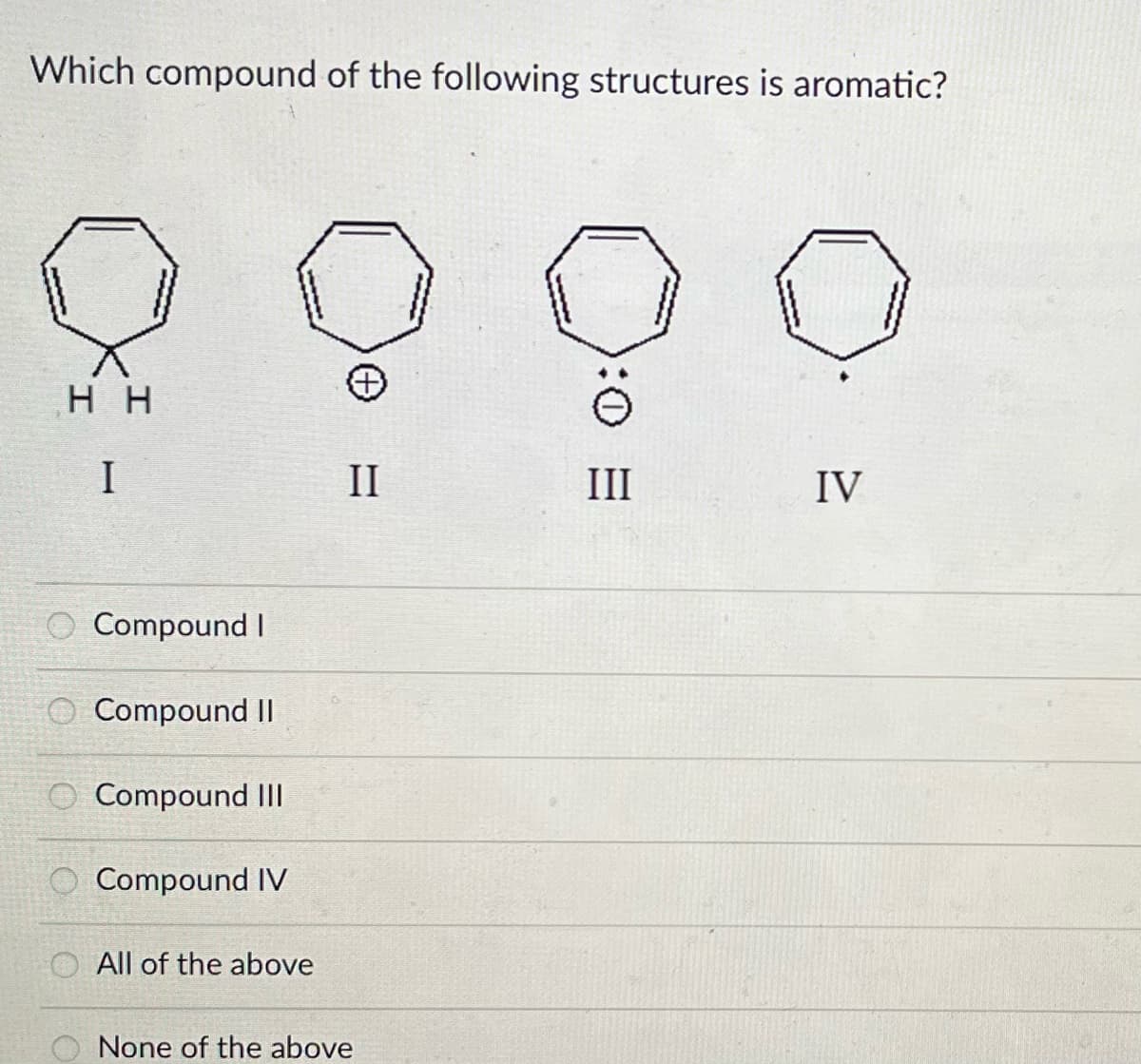
Q: Which of the following is aromatic? www www.
A: Aromatic compound which obey 4n+2 rule and it should be cyclic, conjugated and planar.
Q: What diene and dienophile are used in the Diels-Alder route to the compound shown? H3CO. + H. NC CN…
A:
Q: 5.69 Carvone is present in many essential oils. It is chiral, and its two enantiomers are shown…
A: carvone is a chiral molecule; its enantiomers have different physical and chemical properties.The…
Q: Use a sheet of paper to answer the following question. Take a picture of your answers and attach to…
A: The structure of 2-hexanone is given below. calculating the M/Z value of molecular ion 6×12 + 12 ×1…
Q: What is the molarity of a solution containing 2.75 moles of NaCl in 5.00 L of solution?
A: Definition of molarity- Molarity is the ratio of moles of solute to the volume of the solution.…
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A: The polarity of a bond depends on electronegativity difference between the two atoms sharing the…
Q: Identify the structure for the following H NMR spectrum OH 10 HO PPM
A: Please find your solution below : Spectroscopy is the study of interaction of radiation with matter.…
Q: What type of signal(s) would you observe in the mass and infrared spectrum of the following…
A:
Q: What reagents would you use to carry out the following reaction? (A) H₂ / Pt (B) (C) (D)…
A:
Q: 2.25 g of a mixture of NaCl and KCI salt was dissolved in 20.0 mL of distilled water. The freezing…
A:
Q: IDENTIFY THE OXIDATION NUMBER OF EACH ELEMENT IN THE GIVEN CHEMICAL FORMULA. -All answers to be…
A: Given,Sodium oxalate Na2C2O4Required, 1. Oxidation number of sodium in sodium…
Q: 5-methyl-2,6-heptadiene 4-methyl-1,3-heptadiene O2-methyl-2,4-heptadiene 5-methyl-2,3-heptadiene 0…
A: About dienes:
Q: 3. The solubility product constant (Ksp) of iron (III) chloride is 1.7 x 106. Calculate the molar…
A: The solubility product ( Ksp ) a sparingly soluble salt is defined as the product of the molar…
Q: What chemical species is primarily responsible for the cleaning action to remove volatile organic…
A: Volatile Organic molecules released as gases into atmosphere from certain solids or liquids and have…
Q: Mg(OH)₂ (magnesium hydroxide)) Questions: 1. What is the oxidation number of magnesium in magnesium…
A:
Q: Assess the validity of each statement and choose X if the statement is CORRECT, Choose Y if…
A:
Q: An electron transition fron n=5to n=3 in hydrogen atom. Whats the energy required for this…
A:
Q: OH i.) NaNH, ii.) CH₂Br д ОН |
A:
Q: a 0.4815 g sample of potassium iodide yields 0.7562 g of silver iodide gravimetrically, determine…
A: Mass of potassium iodide = 0.4815 g
Q: What descriptive term is applied to the type of diene represented by 2,4-hexadiene? O isolated diene…
A: Dienes contains two double bonds. There are following type of dienes - (1) Isolated Dienes - These…
Q: Calculate the molar solubility of Ag2CrO4 in a solution that is 0.01 M in AgNO3. Ksp of Ag2 CrO4 =…
A:
Q: An aqueous FeF₂ solution is electrolyzed under 1 bar pressure using platinum electrodes. (a) Write…
A:
Q: B. Draw structures for the products in the following reactions. CN CN CN Jo 70⁰ 1. PrMgBr 2. H₂O* 1.…
A: PrMgBr is the nucleophile here LiAlH4 is reducing agent I2, NaOH,∆=> iodoform reagent.
Q: = 5. a) Calculate the pH of a buffer solution which is 0.1 mol/L with respect to HCN (Ka 4.9 x 10-10…
A: a). pH of a buffer solution can be calculated with the help of Henderson - Hasselbalch equation : pH…
Q: IUPAC Naming Instructions: Provide proper IUPAC names. Name: O₂N. OH NO₂ NO2
A:
Q: A chemist prepares a solution of potassium dichromate (K₂Cr₂O₂) by measuring out 9.94 μmol of…
A: A homogeneous mixture of two or more substances is known as a solution. The species which present in…
Q: An aqueous FeF₂ solution is electrolyzed under 1 bar pressure using platinum electrodes. (a) Write…
A:
Q: The synthetic transformation, as indicated in the scheme below, has been attempted, and a single…
A: To identify in the given reaction whether the starting material has been retained or the product B,…
Q: 19. Provide the major organic product of the following reaction. Be sure to indicate proper…
A: Diel's Alder reaction is a [4+2] cycloaddition reaction , in which one species with higher electron…
Q: How many mols of HBr are used to neutralize 18.66 mL of a 0.2299 M Ca(OH)₂ solution?
A:
Q: balanced equation for the neutralization reaction of aqueous H₂SO4 with aqueous KOH is shown.…
A: Given Reaction :- H2SO4 + 2KOH -----> K2SO4 + 2H2O Molarity of KOH = 0.220M Molarity of H2SO4 =…
Step by step
Solved in 2 steps with 2 images

- Identify the lettered compounds in the following reaction scheme.Compounds F, G, and K are isomers of molecular formula C13H18O. Howcould 1H NMR spectroscopy distinguish these three compounds fromeach other?Resveratrol is an antioxidant found in the skin of red grapes. Itsanticancer, anti-inflammatory, and various cardiovascular effects areunder active investigation. (a) Draw all resonance structures for theradical that results from homolysis of the OH bond shown in red. (b)Explain why homolysis of this OH bond is preferred to homolysis ofeither OH bond in the other benzene ring.Tropone is an unusually basic carbonyl (C=O) compound. When it is treated with one equivalent of the strong acid HBF4, it forms A, C7H7OBF4. Draw the strucutre of A as its most stable resonance structure.
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents. f) fuming sulfuric acidSome of the following compounds show aromatic properties and orhers do not. Predict whic ones are likely to be aromativ and explain why they are aromatic or not (A, B and C)Identify the structures of D and E, isomers of molecular formulaC6H12O2, from their IR and 1H NMR data. Signals at 1.35 and 1.60 ppm inthe 1H NMR spectrum of D and 1.90 ppm in the 1H NMR spectrum of Eare multiplets.
- An unknown compound A (molecular formula C7H14O) was treated withNaBH4 in CH3OH to form compound B (molecular formula C7H16O).Compound A has a strong absorption in its IR spectrum at 1716 cm−1.Compound B has a strong absorption in its IR spectrum at 3600−3200cm−1. The 1H NMR spectra of A and B are given. What are the structuresof A and B?Compound X (molecular formula C10H12O) was treated with NH2NH2, -OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?How is compound A related to compounds B–E? Choose fromenantiomers, diastereomers, constitutional isomers, or identicalmolecules.
- Hexahelicene seems a poor candidate for optical activity because all its carbon atomsare sp2hybrids and presumably flat. Nevertheless, hexahelicene has been synthesized and separated into enantiomers. Its optical rotation is enormous: [a]D = 3700°.Explain why hexahelicene is optically active, and speculate as to why the rotation isso largeIdentify each one Which one is aromatic antiacromatic or nonaromatic ( need explaination)Under certain reaction conditions, 2,3-dibromobutane reacts with twoequivalents of base to give three products, each of which contains twonew π bonds. Product A has two sp hybridized carbon atoms, product Bhas one sp hybridized carbon atom, and product C has none. What arethe structures of A, B, and C?

