Q: Would the following compound be optically active or inactive?
A:
Q: Curved arrows are used to illustrate the flow of electrons. Follow the arrows to predict the interme...
A: Protonation of epoxide Attack of nucleophile at more substituted carbon Deprotonation leading to t...
Q: Which intermolecular forces are present in SeO3
A: Intermolecular force is present in molecules.
Q: The rate of a certain reaction is given by the following rate law: rate=k[N,][H,] Use this informati...
A: Chemical kinetics can be defined as the branch of chemistry that deals with rates of chemical reacti...
Q: You react 10.0 g of nitrogen gas with hydrogen gas according to the following reaction: N2(g) + 3 H2...
A: Given reaction: N2(g) + 3 H2(g) → 2 NH3(g) We have to find the mass of hydrogen gas that is require...
Q: meniscus of the liquid following situations: in the a. Cohesive force is greater than the adhesive f...
A: Here we have to predict the meniscus of liquid in different condition of cohesive and adhesive force...
Q: At active muscle I. PCO2 is high II. acidity is low III. carbamate forms IV. hemoglobin is stabilize...
A: 1. True During exercise, CO2 production is increased and thus pCO2 is high. 2. False pH of a normal ...
Q: 2) Ninety thousand grams of a gas (180 kg/mol) is stored in a tank at 120 kPa and 600 K. If the gas ...
A: Given that, The mass of the gas is m = 90000 g = 90 kg. The molar mass of the gas is M = 180 kg/mol...
Q: The rate of a certain reaction is given by the following rate law: rate =k[H,[NH,] Use this informat...
A: This question is related to chemical kinetics. Chemical kinetics is the branch of chemistry that dea...
Q: 1. If the limiting reactant was found to be K2C2O4⋅H2O, how many grams of the limiting reactant are ...
A: Mass of salt mixture = 2.64 g Mass...
Q: Suppose the average distance between the nucleus and the electron in a newly discovered 3s state is ...
A:
Q: How many significant figures are in each of these measurements? 71. 93 g, 0.420210 kg
A: The significant figure refers to particular digits in a number. According to the measurement rule, o...
Q: What are the splitting and integrations of these two structures?
A:
Q: Titrating with 0.1 M NAOH we red 270 Buffer 1 Buffer 2 DI water 0.1 M acetic acid (mL) 10 35 0.5 M s...
A:
Q: 2. A 0.4682 g sample of a metal is dissolved in a total volume of 50.0 mL. a. If the metal was pure ...
A: a) The Ag and KSCN reacts in following way: Ag+KSCN→Ag(SCN) + K Mass of Ag =0.4682 g Moles of Ag is...
Q: Consider the same reaction taking place in three vessels. The vessels all contain the same amount of...
A: Rate of reaction
Q: Ringer's solution (not lactated Ringers) is a mixture of three salts in water. It contains 8.6 g of ...
A:
Q: Predict the 13C NMR spectra of the 2-chloro-2-methylpropan-1-ol. CH3 HO-CH2 -CI 4CH3 1. How many 1°C...
A: Given, 13C- NMR spectra of 2-chloro-2-methylpropan-1-ol. we have to find out the nmr signal, chemica...
Q: Which of the following are correctly ranked in order of decreasing acidity? The least acidic compoun...
A: The compound in which the congugate base is more stable is more acidic in nature.
Q: c) Compound 3 with molecular formula of C,H,O, 3H ΙΗ ΝMR 3H 2H 15
A:
Q: created Is I, being created or destroyed by the chemical reaction? O destroyed neither created nor d...
A:
Q: In the given three-dimensional molecular structure, the IC OH N differently colored spheres represen...
A:
Q: the mass of OO is approximately 16 gg. What is the whole number multiple that describes the relation...
A: Given as, Mass of Sulphur in SO = 32 gms Mass of Oxygen in SO = 16 gms Mass of Sulphur in SO2 = 32 g...
Q: A 25.0 mL sample of 0.200 M AgNO3(aq) was allowed to react with an excess of NaCI(aq). The AgCl prec...
A: Given, A 25.0 mL sample of 0.100 M AgNO3(aq) was allowed to react with an excess of NaCl(aq). Volum...
Q: Needed is 900 cc of 1/3 (v/v) alcohol in water. How many cc of each component are needed to prepare ...
A: Concentration of solution = 1/3 (v/v) Volume of solution = 900 cc We need to find volume of each co...
Q: Sodium only has one naturally occuring isotope, 23Na, with a relative atomic mass of 22.9898 u. A sy...
A: Given, 22Na has a relative atomic mass of 21.9944 u. 23Na, with a relative atomic mass of 22.9898 u....
Q: The rate of a certain reaction is given by the following rate law: rate=k[H,][NH,] rate = Use this i...
A: "To get remaining sub-parts solved please repost the complete question and mention the sub-parts to ...
Q: 3. The following compound forms a colorless solution in ether, but when a very strong base is added,...
A:
Q: A buffer is prepared by mixing 204 mL of 0.452 M HCI and 0.50 L of 0.400 M sodium acetate. (See Appe...
A:
Q: Example (2). 1 g of water at 273°K is converted into steam at the same temperature. The volume of wa...
A:
Q: A reaction mixture initially has [SO3] = 0.150 M. What is the equilibrium concentration of SO2? ...
A:
Q: grams
A:
Q: Why are the chiral carbons (S) and (R)? Shouldn't they be both (S)
A: The answer is as follows:
Q: Attempt 3 Some of the formulas could be either molecular or empirical formulas; however, some can on...
A: A molecular formula can be defined as the chemical formula that represents the total number of atoms...
Q: Locke-Ringer's solution contains 9 g of sodium chloride, 0.24 g of calcium chloride, 0.42 g of potas...
A: Locke-Ringer Solution An isotonic solution used for the treatment of dehydration. It contains diffe...
Q: A physician has ordered 250 cc of 2.5 % (w/v) tincture of iodine. Iodine crystals and alcohol are av...
A:
Q: A chemistry student weighs out 0.234 g of chloroacetic acid (HCH,CICO,) into a 250. mL volumetric fl...
A:
Q: How are the column efficiency in a GC separation affected by the carrier gas flow?
A: How are the column efficiency in a GC separation affected by the carrier gas flow? explanation is gi...
Q: Which of the following reactions is an addition reaction? a. C,Hg + Cl2→ C,H;Cl2 b. C,H6 + Cl2→ C,H;...
A:
Q: A certain solution consists of isopropyl alcohol diluted in water. If a 500 mL bottle contains 75 mL...
A: The volume of solute in milliliter present in 100 mL solution is known as the volume percent concent...
Q: Draw a product that could be formed when 1,3-butadiene and (E)-2-butenedial. Include any relative st...
A:
Q: What is An for the following equation in relating K c to K p? LICIO 3( s) = LICIO( s) + O 2( 8) 0. 3...
A: Solid lithium chlorate decomposes into solid lithium hypochlorite and oxygen gas. The equation for t...
Q: According to Riippi et al. (1998), briefly discuss the effects of increased compression force to the...
A: When compression force increased drug material will suffer some deformation that causes the average...
Q: The rate of the reaction NO2 + CO NO + CO2 depends only on the concentration of nitrogen dioxide bel...
A: NO2 + CO -----> NO + CO2 Time (s) [NO2] mol/L 1/ [NO2] 0 0.500 2 1.20 x 103 0.444 2.25 3...
Q: Degradation of an airport's aging fuel tanks is suspected, resulting in both rust and water contamin...
A: A question based on properties of liquids that is to be accomplished.
Q: QUESTI UN 15 . Which of the following approximate pka ranges for the following acids is correct? You...
A:
Q: 4. For the following compounds determine i) HDI (degrees of unsaturation), ii) diagnostic IR bands (...
A: To find: i) HDI (degrees of unsaturation), ii) diagnostic IR bands (approx. wavenumber (cm-¹) with t...
Q: 0.800 grams of solid barium hydroxide, Ba(OH)2, is added to enough water to prepare 250.0 mL of solu...
A:
Q: What is/are the major product(s)_of the following reaction? + Br2 FeBr3 Br Br Br Br II III IV O|| O ...
A:
Q: Based on the following equilibrium H++ HCO3- ↔ H2CO3 A)State what would happen if we added HCl to t...
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on ...
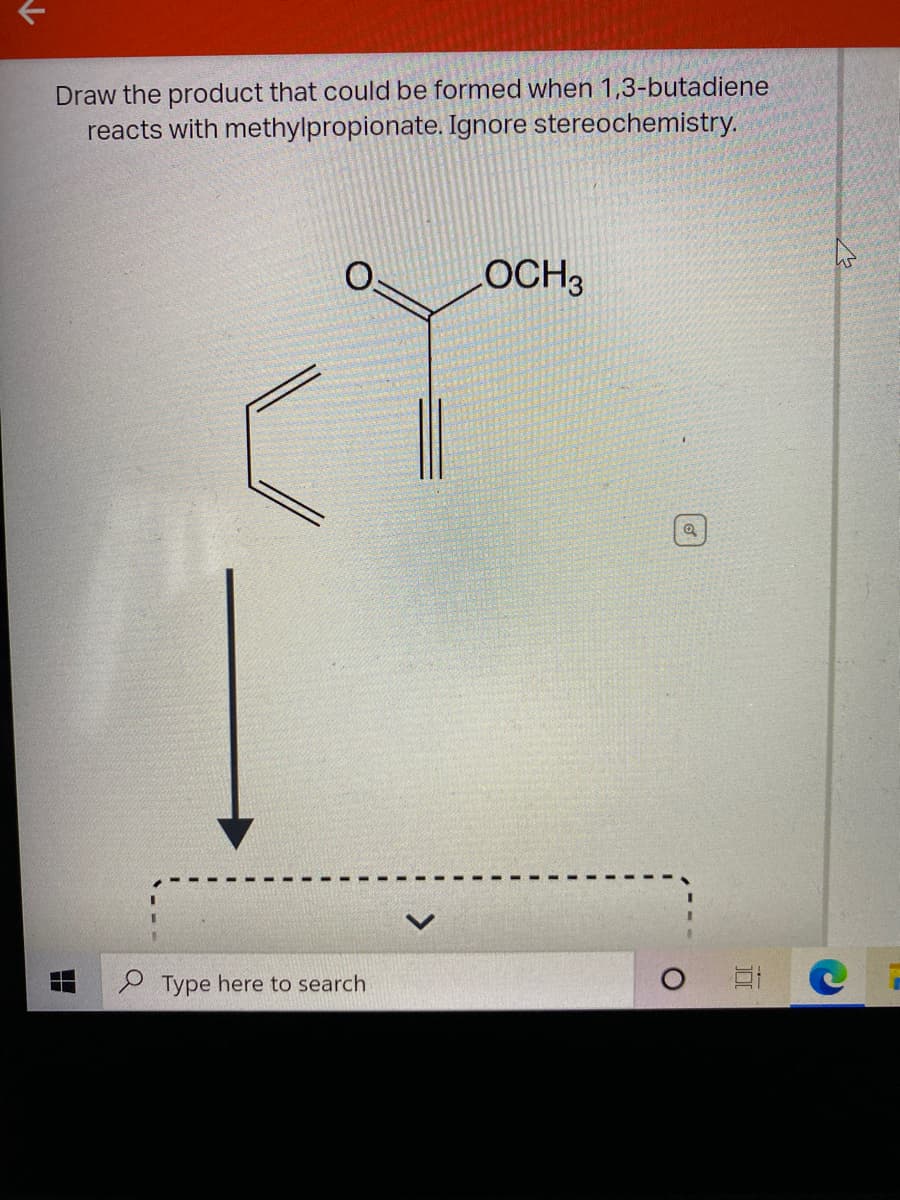
Diels alder reaction is the reaction between diene and dienophile. This is also known as 4+2 cycloaddition reaction.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw the products of the reaction. Be sure to specify stereo- and regiochemistry when appropriateo chem - please show stereochemistry and break down each step as much as possibleDraw the (pericyclic) reaction (reaction 1), including all mechanistic steps, that lead from the starting materials to the intermediate product. Show all isomers that can be formed and provide an explanation as to why that is. Determine the configuration of all chiral centers. I included the molecule, the retrosynthesis of the molecule, and reaction 1 to help solve this question.
- Please show major products for these nucelophilic substitution and elimiation reactions. explain if there is any stereochemistry, and axial and equatorial.1.Draw the product, please shows step by step in details 2.Explain if (CH3)2CH in this reaction can attach in ortho positon in Friedel Craft Alkylation?? why or why not???What are products formed from the below reaction? Draw the stereoisomers and name them. Please solve the problem in detail.
- Be sure to answer all parts. Draw the products for the reaction of 1−hexyne with ethylmagnesium bromide as if it involved ethyl anion instead of (CH3CH2MgBr) and state the correct flow of electrons.For each section, circle the mechanism from the two options given (SN1 or SN2) and draw the main organic product resulting from that mechanism. Indicate the stereochemistry and if two configurational isomers form, draw both.The molecules below react in a displacement reaction. Determine the products) and assign configuration (R/S) to any stereogenic centers in the product. The Cahn-Ingold - Prelog rules can be used to assign priority to the four different groups on a stereogenic center.
- draw the major organic product generated in the reaction below. Pay attention to Regio and stereochemical detailPlease help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)Draw all the major products resulting from the reactions below, taking into consideration both the regioselectivity and stereoselectivity of the reactions. If pairs of enantiomers form, draw both enantiomers. (NOTE: for the second reaction, draw out the hydrogen.)

