Q: Draw all the possible stereoisomers for each compound, and label pairs of enantiomers and…
A:
Q: 4.2 Consider the following Haworth projection of monosaccharide C: OH OH OH OH он monosaccharide C…
A: Conversion of Haworth to fisher projection 1. For the carbon number (C2 , C3 and c4) if the OH…
Q: Ephedrine (2-methylamino-1-phenylpropanol) is a drug that is used as a decongestant and…
A:
Q: How is each compound related to the simple sugar D-erythrose? Is it an enantiomer, a diastereomer,…
A:
Q: Which two Fischer projections represent a pair of enantiomers? CH3 CH3 CH3 HOH H- H- CI но- C₂H5 A O…
A:
Q: NH H H;C H
A:
Q: 4. Are these two compounds: a) the same b) enantiomers OR c) diastereomers? HÖ +-OH Explain how you…
A:
Q: Label each pair of stereoisomers below as: a. enantiomers b. diastereomers C. identical Place the…
A: We have to find the Absolute configurations in the two stereogenic centres (by Flying Wedge as…
Q: -82 Identify each of the following Haworth projection formulas as an a-D-monosaccharide or a…
A: Note: According to our guidelines we are supposed to answer only first three subpart. Kindly repost…
Q: Draw enantiomers for each of the following using a. perspective formulas. b. Fischer projections. 1.…
A: Enantiomers- These are the stereoisomers that are non-superimposable mirror images of each other…
Q: C. For the given structure, draw its enantiomer and assign the absolute configuration (R or S) H…
A: An absolute configuration of a molecule is the spatial arrangement of atoms in a chiral molecule. It…
Q: Which of the following is the C3 epimer of L-idose? но но Но но H- он H- OH но но но H. OH OH H- он…
A: The C-3 epimer of L-idose is given below
Q: Choose the set of letters corresponding to the BEST answer. сно CHO CH,OH Fo HO H H OH CHOH CHOH CHO…
A: Hexose are classified as aldohexose and ketohexose. This classification is based on the finctional…
Q: 5. Assign the R and S configurations for all stereogenic centers in the following compounds. H.…
A: Note: According to our guidelines we are supposed to answer only first three subpart of question.…
Q: Define each stereocenter as either (R) or (S). If the molecule does not have a stereocenter select…
A: ->Each chiral center has specific configuration R or S.
Q: 1) Locate the stereogenic centers in each drug by placing an asterisk on top of it. a) Albuterol…
A: center is also political centre assymetric centre centre. Chiral center is the center where the…
Q: CH3CH(OH)COOH has a stereogenic center and is achiral Select the correct response: True False
A: Note : Carbon is attached four different groups is called asymmetic carbon or chiral carbon.…
Q: QI: Assign all the stereocenters present in each of the following compound and give their R\S…
A:
Q: CH; 13. H OH CH,CH, The Fischer projection above is of a. a D enantiomer b. an L enantiomer c. both…
A: If the OH on the bottom chiral center is at right, the sugar has D-configuration and if the OH on…
Q: The following is a terpene. How many stereocenters will there be after the compound reacts with…
A: Stereocenters are those points or atoms present in a Molecule, which contain different type of…
Q: Locate the stereogenic centers in telaprevir, a drug used to treat hepatitis C, and label each…
A: Given compound,
Q: Draw enantiomers for each of the following using a. perspective formulas. b. Fischer projections.…
A: The number of stereoisomers of an organic molecule depends upon the number of chiral carbon atoms…
Q: Identify whether each of the following pairs of compounds are enantiomers or diastereomers, and…
A:
Q: 11) Answer each of the following questions using the Fischer projections below. CH2CH3 CH,CH3 CH2CH3…
A: Since you have asked multiple subpart of a question, we will solve the first three subpart for you.…
Q: What is the relationship between the following molecules? Enantiomers, Diastereomers, Identical, or…
A:
Q: Define each stereocenter as either (R) or (S). If the molecule does not have a stereocenter select…
A: Assign priorities to the groups attached to the chiral centre as per CIP rule(Cahn-Ingold-Prelog) If…
Q: 7. Draw Fischer projections of two meso diastereomers of the following stereoisomer. CH3 CI CI H H…
A:
Q: Identify the type of stereoisomerism in each of the following molecules and draw the stereoisomer…
A: There are various isomers present. Compounds with double bond shows geometric isomers. Trans/E and…
Q: Following are several stereorepresentations for lactic acid. Use (a) as a reference structure. Which…
A: A tetrahedral atom that is bonded to four different groups is called as chiral center. The most…
Q: НО HO OH OH OH OH QH НО
A:
Q: Identify all chirality centers in each of the following compounds HO HO HO HO OH Ascorbic acid…
A: Answer: In the molecule, chirality centers are those carbons(C) where the carbon atom makes a bond…
Q: Locate the prochirality center in the following molecule, and label the H. hydrogen. Br H C C-H H.…
A: Prochiral center: The atom which is going to be chiral after the reaction.
Q: Following are four Newman projection formulas for tartaric add. (a) Which represent the same…
A:
Q: Draw the structure of a meso compound that has five carbons and three chirality centers. 5-53 5-5…
A:
Q: Identify whether each of the following pairs of compounds are enantiomers or diastereomers, and…
A:
Q: Which two Fischer projections represent a pair of enantiomers? CH3 CH3 C₂H5 H # H CI C₂H5 B HO H- H…
A: A question based on stereochemistry of organic reactions that is to be accomplished.
Q: d) Draw the structure of the expected product when monosaccharide C undergo mutarotation upon…
A:
Q: Which of the following is the alpha Haworth projection of the compound shown below? * CH2OH C=0 HO-H…
A:
Q: 1. Label each of the following pairs as identical, unrelated, or conformational or constitutional…
A: (a) These are constitutional isomer since connectivity of both Cl with C is different in both. (c)…
Q: 5. Three of the following projection formulas are the same compound. The other is the enantiomer.…
A: The absolute configuration for the given molecules is;
Q: How many chirality centers are present in the cormpound shown? Cl- OA1 OB. 3 OC The compound does…
A: How many chiral centre are present in the compound in cis-1,4-dichlorocyclohexane ?
Q: Assign R or S configuration to the chirality center in each of the following molecules: (a) CH3 (b)…
A: Some points should kept in mind before R and S nomenclature. CIP rule i.e. Chain Ingold Prelog Rule…
Q: PQ-28. How are these molecules related? H. OH HO Br CH3 Br constitutional (D) isomers (A) identical…
A: Detail description is given below
Q: 9. Assign each of the following stereocenters as either R or S. Circle your answer below. н он Br. H…
A:
Q: Locate the stereogenic centers in telaprevir, a drug used to treat hepatitis C, and label each…
A: Stereogenic centers are those where there are 4 different substituents attached to the C.
Q: Identify the relation between the following compounds. C2H5 НО -H- Br Br OH (A) Enantiomers (B) Same…
A:
Q: Please draw the fischer projections for the following compound and determine whether it is (R) or…
A: The name of the given compound is 2-chlorobutane which is an organic compound that is made up of…
Q: Zanamivir is a drug prescribed to patients with Influenza virus. Zanamivir inhibits the viral…
A:
Q: Please draw the fischer projections for the following compounds and determine whether they are (R)…
A: Fischer projection - Fischer projection is a two dimensional representation showing the…
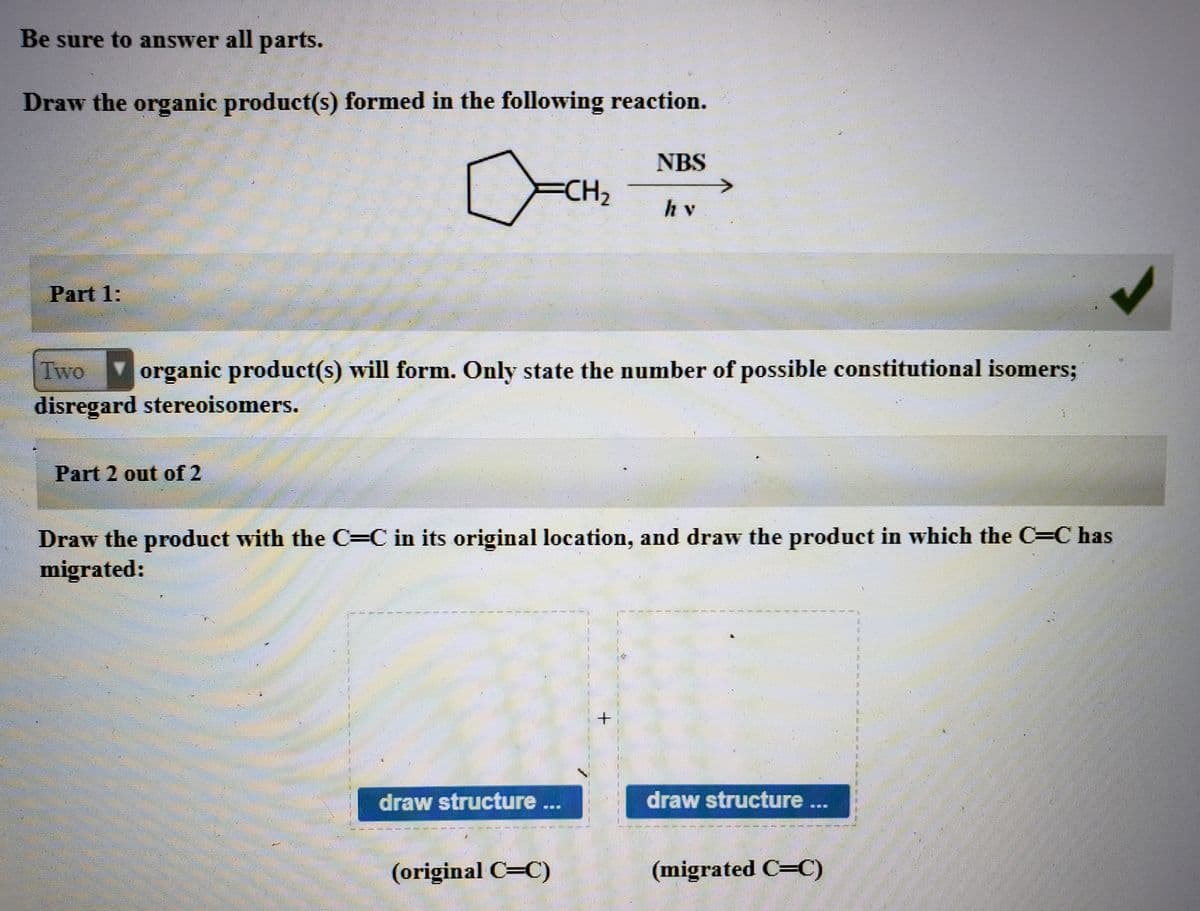
Part 2 needed
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- A hydrocarbon of unknown structure has the formula C8H10. On catalytichydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. Onhydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.(a) How rnany degrees of unsaturation are present in the unknown?(b) How many triple bonds are present?(c) How many double bonds are present?(d) How many rings ar e present?(e) Draw a structure that fits the data.Draw the structure(s) of the major organic product(s) obtained after workup of the following reaction. Use the wedge/hash bond tools to indicate stereochemistry where it exists. If no reaction occurs, draw the organic starting material. Draw structure(s) of product(s) after aqueous workup. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.Draw the organic product(s) of the following reaction. You do not have to consider stereochemistry. Include cationic counter-ions, e.g., Na+ in your answer, but draw them in their own sketcher. If no reaction occurs, draw the organic starting material. Separate multiple products using the + sign from the drop-down menu.
- Draw the organic product(s) of the following reaction. You do not have to consider stereochemistry. The aromatic ring, when present, is unreactive in all cases. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.Rearrangements can occur during the dehydration of 1° alcohols even though no 1° carbocation is formed—that is, a 1,2-shift occurs as the C— OH2+ bond is broken, forming a more stable 2° or 3° carbocation, as shown in Equation [1]. Using this information, draw a stepwise mechanism for the reaction shown in Equation [2]. We will see another example of this type of rearrangement in Section 16.5C.Draw the major organic product(s) of the following reaction. You do not have to consider stereochemistry. If no reaction occurs, draw the organic starting material. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.
- Please explain question 1 and 8 1. How many degrees of unsaturation are present in your unknown monosubstituted benzene starting material? 8. Based on all of the data provided, what is the identity of the “G” group? (Please NEATLY & CLEARLY draw its full Lewis structure in the space provided) Thank you!a) Write out the 3-step arrow pushing mechanism showing how 1-pentene is hydrated to make 2-pentanol. b) Draw the other 2 alkenes(don’t forget cis/trans isomers!) that could also be hydrated to make 2-pentanol. Briefly explain why1-pentene is the best choice.Please help illustrate the stereochemistry with dashes and wedges. Question: Draw a diastereomer for each of the following compounds: Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. The single bond is active by default. Show the appropriate stereochemistry by choosing the dashed or wedged buttons and then clicking a bond on the canvas.
- Ignoring stereoisomers, draw the two possible enols for butan-2-one (CH3COCH2CH3), and predict which one is more stable.Please solve correctly: Optio for Q1. a.)2-hexyne b.)3-methyl-1-pentyne c.)3-hexyne d.)1-hexyne e.)4-methy-1-pentyne Q2) a.)Alkenes react with nucleophiles and bases. b.)Alkenes react with electrophiles. c.)In the reactions of alkenes, the p bond is not always broken. d.)Alkenes contain double bonds from two sp-hybridized carbon atoms.1. What type of TAG is given above?2. How many kinds of products will be formed from the saponification of this TAG?

