Draw the products of the two step reaction sequence shown below. Ignore inorganic byproducts. HO PBr3 Select to Draw thiol DMF cyclohexane NaH
Q: What is the pOH of an aqueous solution of 2.56 × 10-² M sodium hydroxide? POH =
A: Given : [NaOH] = 2.56 × 10-2 M Tip : pOH = -log[OH-]
Q: Nuclei with the same number of neutrons but different mass numbers are called isotones. Write the…
A: Nuclei with the same number of neutrons but different mass numbers are called isotones. Mass number…
Q: A mass of 1.3 g of an unknown solute is added to 10.7 benzene (Kf=5.12°C kg/mol). The freezing point…
A:
Q: A 48.0 g sample of an unknown metal at 99.0 °C was placed in a constant-pressure calorimeter…
A:
Q: A liquid is exposed to infrared radiation with a wavelength of 2.43 × 10-4 cm. Assume that all the…
A: Answer : Given that, Wavelength =2.43 x 10-4 cm =2.43 x 10-6 m Energy= 30.03 J
Q: Show what Grignard reagent and what carbonyl compound you would start with to prepare the alcohol…
A: Grignard reagent is an alkyl magnesium halide. The general formula of Grignard reagent is RMgX. R =…
Q: 2 KClO3 2 KCl + 3O2 How many grams of O2 will be formed from 3.76 grams of KClO3
A:
Q: Bromine has two stable isotopes: 79Br and 81Br with masses of 78.9183371 amu and 80.9162906 amu. If…
A: In this question we have to tell the the Mass of 79Br in 10 gram of Br which has 79.904 amu mass.
Q: Figure 3-7 100 IT A 60 0. 100 1 0 4800 100 80 3000 W тари B 1000 20 0 4000 IR2017-91129TM 3000 2000…
A: The IR spectrum is used for the confirmation or detection of functional groups present in the…
Q: Consider the following demand function Q(P)=1509-69P Calculate the slope of this function (i.e., the…
A:
Q: When the aromatic ring attacks a strong electrophile, E*, via the mechanism shown which of the…
A: Introduction
Q: Which of the following is not a factor that controls the rate of the reaction?
A: Given : statement
Q: Consider the following scenario, would say the figure is demonstrating: OOOO The less dense nexane…
A: When one substance is completely dissolved in another substance, then it is called completely…
Q: What information is needed to determine the molarity of a solution? What are the implications of the…
A: Molarity is a measure of the concentration of a solution, expressed as the number of moles of solute…
Q: Please draw 2-propylpentane and 4-propylpentane.
A: Please find your solution below : Structural formula is the representation of a compound in which…
Q: Incorrect. Predict the major product(s) that are expected when the following compound is heated with…
A: In this question, HBr is concentrated that means it is in excess amount so that you will get…
Q: Temperature, °C Estimating from the heating curve show, what is the approximate melting point of the…
A: When a substance undergo phase change (solid to liquid or liquid to gas ) the energy involved in a…
Q: What kind of intermolecular forces act between a hydrogen iodide molecule and a chloroacetylene…
A: Introduction Intermolecular forces are the forces of attraction or repulsion that act between…
Q: A possible resonant structure for the following molecule is: a. b. C. d. NH₂ + NH₂ +NH₂ + | : NH₂…
A: Introduction to Resonance Resonance is an important concept in chemistry that is used to explain the…
Q: Select all of the true statements about melting points. a. The melting point range of a pure…
A: The melting point of a substance is the temperature at which it changes from a solid to a liquid…
Q: The solubility of O2 in water is 5.85 x 10-4 M at 25 oC and 0.45 atm of O2 pressure . What will the…
A:
Q: Identify the relationship between the following two Newman projections. H3C_ H CH3 н CH3 H and H3C…
A: Given : two Newman projection
Q: Under standard biochemical conditions, which ONE of the following relationships must be TRUE by…
A: Under the given set of conditions, a chemical reaction will be spontaneous if the change in the…
Q: Please provide an equation for the following nuclear process: 231 Pa decays via ß-emission. 91
A: This question is related to nuclear chemistry. There are mainly three types of radioactive decay-…
Q: A gas contracts to half its initial volume while releasing heat to the surroundings. What can be…
A: Given : heat is releasing to the surrounding And gas contract
Q: RECTIONS: Fill in the anks (1, 2,3, & ??) to mplete the organizer. word bank is provided your…
A:
Q: AlP Express answer as an ion.
A: Since, Ionic compound are those compound which on dissociation produced cation and anion. Thus,
Q: What is the multiplicity expected in the hydrogen NMR spectrum for the hydrogen atoms marked by a…
A: Given : structure of molecule Tip : multiplicity in NMR = n+1 n = adjacent hydrogen
Q: What will be the product or products if zinc (Zn) reacts with hydrochloric acid (HCI) Most active…
A:
Q: Una estructura resonante posible para la siguiente molécula es: a. b. C. NH₂ NH₂ +NH₂ + 1 :NH₂ :NH₂
A: In this question, we will identify the possibile resonance structure of the given compound. You can…
Q: When a small amount of water is added to t-butyl, what happens to the cooling curve for this new…
A: Colligative properties are properties of a solution that depend on the concentration of solute…
Q: How much heat must be supplied to 36 g of ice at -21°C to convert it to steam at 152°C? (Specific…
A: we have to calculate the heat need to heat ice at -21°C to steam at 152°C
Q: For each of the salts on the left, match the salts on the right that can be compared directly, using…
A: Answer: For sparingly soluble salts it is considered that all their dissolved part in aqueous…
Q: Rates of reaction can be explained by Orate theory O collision theory quantum theory O Plank theory…
A: Rate of reaction is the change in concentration of reactant or product, per unit of time. A→B…
Q: Consider the reaction: 2 C₂H₂(g) +5 O₂(g) →4 CO₂(g)+2 H₂O(1) Hrxn=-2600. kJ/mol pos90 31000d food 0…
A: Given : reaction and value of ∆Hrxn Tip : energy is given for 1 mole
Q: What is the correct name of the compound below? gl
A: Rule of IUPAC- 1) Choose the longest carbon chain as parent chain. 2) Numbering start from those…
Q: 애 CH₂OH H 애 -0 애 OH H In the box below, draw the open-chain structure (as a Fischer projection)…
A: To convert a Fischer projection to a Haworth projection, follow these steps: Locate the horizontal…
Q: 26 Write equations for the reaction of 1-butanol, a primary alcohol, with these reagents. heat (a)…
A: Given : name of reactants and reagent
Q: Ⓒ Diaw structural formulas for the major organic products for XOH SOCI₂ CH b) 2) HO Hcl -OH HBr…
A:
Q: Calculate the molarity of 110.0 grams of calcium chloride in a total of 500.0 mL of aqueous…
A: Given, mass of calcium chloride = 110.0 g we know, molar mass of calcium chloride(CaCl2) = 110.98…
Q: When a solution is made from 47.8 g of an unknown nonelectrolyte dissolved in 151.4 g of solvent,…
A: Given: mass of non-electrolyte = 47.8 g mass of solvent = 151.4 g = 0.1514 Kg Elevation in boiling…
Q: Which chemical reaction below yields the heat of formation of liquid ammonia (NH3)? 3H(g) + N(g)→→…
A: Reaction equation in which a mole of a chemical compound is formed from its constituent elements…
Q: a. The density of sorbitol 70% w/w solution is 1.285g/ml. What is the concentration of sorbitol in…
A: 70 %w/w solution means that 70 g of sorbitol is present in 100 g of solution.
Q: of NaOH MeOH
A:
Q: Which statement best describes the structure that results from the following curved arrow: |- ? O…
A:
Q: GENERAL INSTRUCTIONS: Fill the table with the necessary information. Use your MOLECUL MOLECULAR…
A:
Q: OH + Na OE a. draw the products and label the acid, base, conjugate acid and conjugate base with pk,…
A: A base is a compound which abstracts a proton from an acid and form a conjugate acid. And the acid…
Q: Calculate the molarity of a 1.03 m aqueous solution of tin(IV) nitrate with a density p= 1.082 g/mL.…
A:
Q: Which statement is the best description of carbon trifluoride, CHF33 polar bonds, non-polar molecule…
A: CHF3, also known as carbon trifluoride
Q: 3) Predict the products of the following reactions: a) Y b) c) Br Mg Ether Mg Ether (N) H₂O 1. 2x…
A: Alkyl magnesium bromide is known as a Grignard reagent. The Grignard reagent is a very useful…
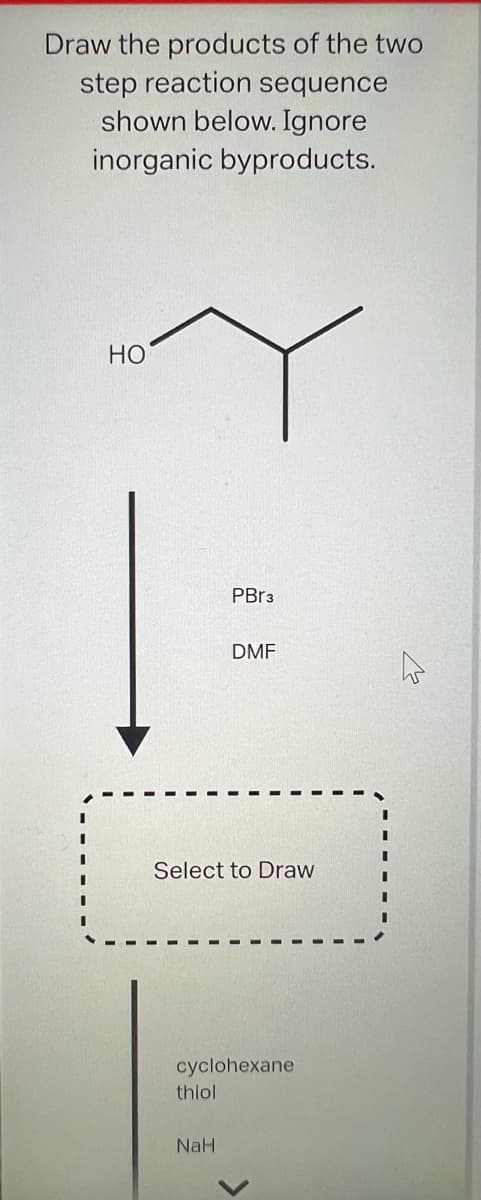
The attack of PBr3 on alcohol follows SN2 reaction.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- The Favorskii reaction involves treatment of an -bromo ketone with base to yield a ring-contracted product. For example, reaction of 2-bromocyclohexanone with aqueous NaOH yields cyclopentanecarboxylic acid. Propose a mechanism.The following molecule was formed by a Robinson annulation reaction. What reactants were used?When a 2, 6-disubstituted allyl phenyl ether is heated in an attempted Claisen rearrangement, migration occurs to give the p-allyl product as the result of two sequential pericyclic reactions. Explain.
- Draw the major organic product(s) of the following reaction. If no reaction is expected, write "N.R." Clearly show stereochemistry if appropriate.(a) What happens when CH3—O—CH<sub3 is heated with HI?(b) Explain mechanism for hydration of acid catalyzed ethene :CH2 = CH2 + HzO CH3—CH,—OHDraw the major organic product(s) of the following reactions. If no reaction is expected, write "N.R." Clearly show stereochemistry if appropriate.
- When a cyclic ketone reacts with diazomethane, the next larger cyclic ketone is formed. This is called a ring-expansion reaction. Draw a mechanism forthe following ring-expansion reaction.When 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsevproduct predominates.(a) Draw the reaction, showing the major and minor productsThe reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO- + H2O2 leads to a racemic mixture. Explain why a racemic mixture is obtained in each case.

