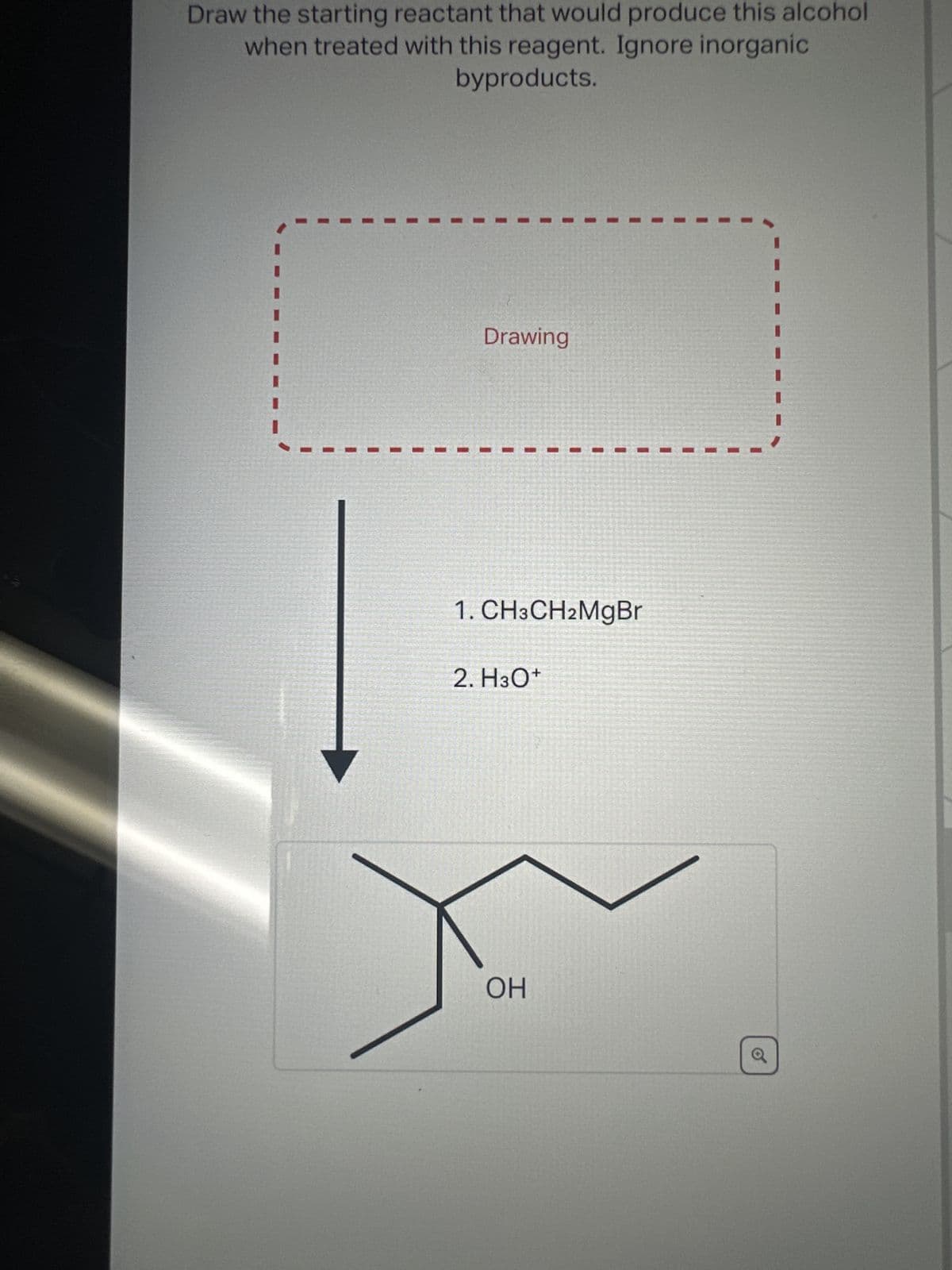
Draw the starting reactant that would produce this alcohol when treated with this reagent. Ignore inorganic byproducts. Drawing 1. CH3CH2MgBr 2. H3O+ OH
Q: calculate activity grams of ferric oxide ions in 1g ferric oxide in 20ml solution.
A: Given:1 gram of ferric oxide (Fe2O3)Volume of solution = 20 mL
Q: None
A: 1. **NaF**: Sodium fluoride (NaF) has the lowest melting temperature among the options provided.…
Q: 2. Starting with 2-methyl-1-phenylpropan-1-one, show how you can convert this to 2-methyl-1-…
A: The objective of the question is to convert 2-methyl-1-phenylpropan-1-one to…
Q: None
A: The formulas you provided are a mix of calculations and data points:a) 21.8 / 4.79 This is a simple…
Q: None
A: To create a graph showing the relationship between Concentration and Density for a sugar solution,…
Q: 2. Which of the following compounds would be considered anti-aromatic?
A: Step 1:Hückel's rule for aromaticity, antiaromatic and non aromatic compound are :-…
Q: A student artificially pumped 1 mole of each substance seen below in a piston with a moveable lid…
A: The reaction:2 C3H7OH(g) + 9 O2(g) ←→ 6 CO2(g) +8 H2O(g) The number of molecules in reactant side =…
Q: From the table below, calculate : 1. The order of A 11. The order of B 111. The Rate Law 1. The Rate…
A:
Q: Determine whether the following compound is optically active or optically inactive: O optically…
A: Step 1: Step 2: If a compound contains a chiral carbon and lacks a plane of symmetry, it's…
Q: Find the starting materials of this compound via retrosynthetic approach (4 marks) Further synthesis…
A: Step 1: Step 2: Step 3: Step 4:
Q: 1. Draw a map tracing the steps that this molecule would take to go individually through…
A: Detailed explanation:Let's break down the steps for each of the three metabolic pathways:…
Q: Propose a retrosynthetic analysis of benzocaine from the starting material shown. Your answer should…
A: Step 1: methyl group on benzene ring is an electron donating group so nitration of toluene with HNO3…
Q: A 1.00 L flask is filled with 1.45 g of argon at 25∘C. A sample of ethane vapor is added to the same…
A: The objective of this question is to find the partial pressures of argon and ethane in a flask. The…
Q: What is the pH of a 0.30 M solution of NH3? (Kb 1.8 x 10-5) a) 6.84 b) 8.32 c) 11.43 d) 7.09 e) 7.99…
A: Step 1:
Q: Select the most appropriate reagent for the synthon shown below: A) (A) B (B) C) (c) (D) OH la you…
A: Step 1: Synthon : Synthon is a generalized fragments of a chemical compound which can be formed…
Q: Select the appropriate set of reagents for the below transformation: ? NMe2 A Set A B Set B C) Set C…
A: Let's analyze the reagent sets:Set A (Me₂NPh/Cl⁻):Me₂NPh (dimethylaniline) can act as the…
Q: Gallium-67 (t1/2 = 25 hours) is used in the diagnosis of tumors. If a patient ingests a compound…
A: The objective of this question is to determine the amount of Gallium-67 remaining in the patient's…
Q: ge 2: 3: O Listen What is the type of reaction and what is the product of this reaction when…
A: This reaction undergoes an elimination reaction because the -OTs group is a good leaving group and…
Q: None
A: Step 1:The number of valence electrons in Sn is 4 as it belongs to group 14. On the other hand, F…
Q: Provide a mechanism and explain the stereochemistry at position 3 and 4 of the product. TIPS OBn 3…
A: The objective of the question is to explain the mechanism of the reaction and the stereochemistry at…
Q: יוון UIPAC name of this compound?
A: The IUPAC nomenclature of organic compounds follows this naming format:Writing the name of organic…
Q: Pleeeeeease ssssolve questiiiiion 5
A: 5. Given: Empirical Formula, EF:…
Q: Identify how you would make pentylamine from 1-hexanol: OH NH2 The transformation above can be…
A: 1-hexanol is first treated with PBr3 to replace -OH with -Br via SN2 reaction.Afterward, a strong…
Q: Draw the mechanism for the following reaction. Ο EtOH, HCI
A:
Q: 8. What is the identity of Peak 1 in the mass spectrum bellow: [Cytochrome C+HI Relative Abundance…
A: A mass spectrum is a graphical representation of the distribution of ions based on their…
Q: One of the nuclides in spent nuclear fuel is U-235 an alpha emitter with a half-life of 703 million…
A: The objective of this question is to determine the time it will take for the amount of U-235 to…
Q: Can you please set up the ice table
A: Step 1:Introduction to datas :Given ,Ka for HNO2= 6.8∗10−4Molarity of HNO2= 0.55 MMolarity of…
Q: Help please
A:
Q: 77. For the reaction SbCls(g) AG (SbCls)=-334.34 kJ/mol SbCl3(g) + Cl2(g), AG°f (SbC13)=-301.25…
A: The objective of this question is to calculate the equilibrium constant (Kp) for the given reaction…
Q: Can you explain the problem? How do you identify them?
A: Step 1: SolutionThe removal of one proton (H+) from a given compound gives us a conjugate base. We…
Q: Please fast expert answer solutions
A:
Q: Draw the products and label them as major or minor.
A: See image.
Q: Add curved arrow(s) to draw step 2 of the mechanism to form product A. Modify the given drawing of…
A:
Q: Write the complete equation for and identify the mode of decay for each of the following unstable…
A: The objective of the question is to write the complete decay equation and identify the mode of decay…
Q: How many hydrogen bond donors does the following molecule have? H Η
A: Step 1:Hydrogen bond donor : H bond donor is a strong electronegative atom (N, O or F) which is…
Q: 2. When an anhydrous solution of CrCl₂ in EtOH was treated with ligand 1 (below), a precipitate of…
A: (a) Analytical Data Interpretation and Possible Structures for X and YGiven the analytical and…
Q: From the table below, calculate : 1. The order of A 11. The order of B 111. The Rate Law 1. The Rate…
A: The objective of the question is to determine the order of reaction with respect to reactants A and…
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Fe(s) + O2(g) → 2FeO(s) ΔΗ…
A:
Q: Predict the product(s) for the following reaction. The answer is shown, please help with the…
A: Step 1: Step 2:MechanismStep 3: Step 4:
Q: Please correct answer and don't use hend raiting
A: The molecule on product of the reaction is 2-methylbutanal. It can be synthesized from no more than…
Q: You want to quickly set up a temporary water bath in your lab with a volume of 10.0 L and a…
A: Step 1:Volume of water is 10LFinal temperature is 37oCInitial temperature of hot water is…
Q: The half-life of Ra-226 is 1.60 x 10^3 years = 5.05 x 10^10 s Find k What is the activity in…
A: Step 1: Step 2: Step 3: Step 4:
Q: The lone pairs of which heteroatoms(s) contribute to the aromaticity of the following molecule? N $
A:
Q: 2.49 Write the equality and two conversion factors, and identify the numbers as exact or give the…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Solvellllllll
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculate quantity of materials produced from the electrolysis of a molten salt. A current of 17.3A…
A: To predict the products of the electrolysis of molten MgCl2 and write the reactions occurring at the…
Q: The concentration at which micelles first form in solution is known as critical micelle…
A: Detailed explanation:The determination of CMC can be achieved through various experimental…
Q: Data Table Test mL of mL of pH tube HOAc NaOAc Trial Trial pH HOAC pH Average Millimoles Millimoles…
A: To calculate the average pH and average calculated pKa, we first need to determine the…
Q: Hi, this is an Organic Chemistry problem. Please help. Thank you. Can you please…
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 1: Aromaticity a. Draw a fully labelled MO energy diagram for each (match clearly). b.…
A: Step 1: The given molecule have the following IUPAC name: • Pyrrol-3-one • 1-Methyl-1H−borepine…
Step by step
Solved in 2 steps with 1 images

- (a) What functional group is undergoing a transformation in the reaction? (b) What functional group is it being transformed into (in the final product)?Give one example of Williamson ether synthesis. Show the reactants materials and the product. Draw the reaction mechanism?Please name Andrwo the major product of this reaction

