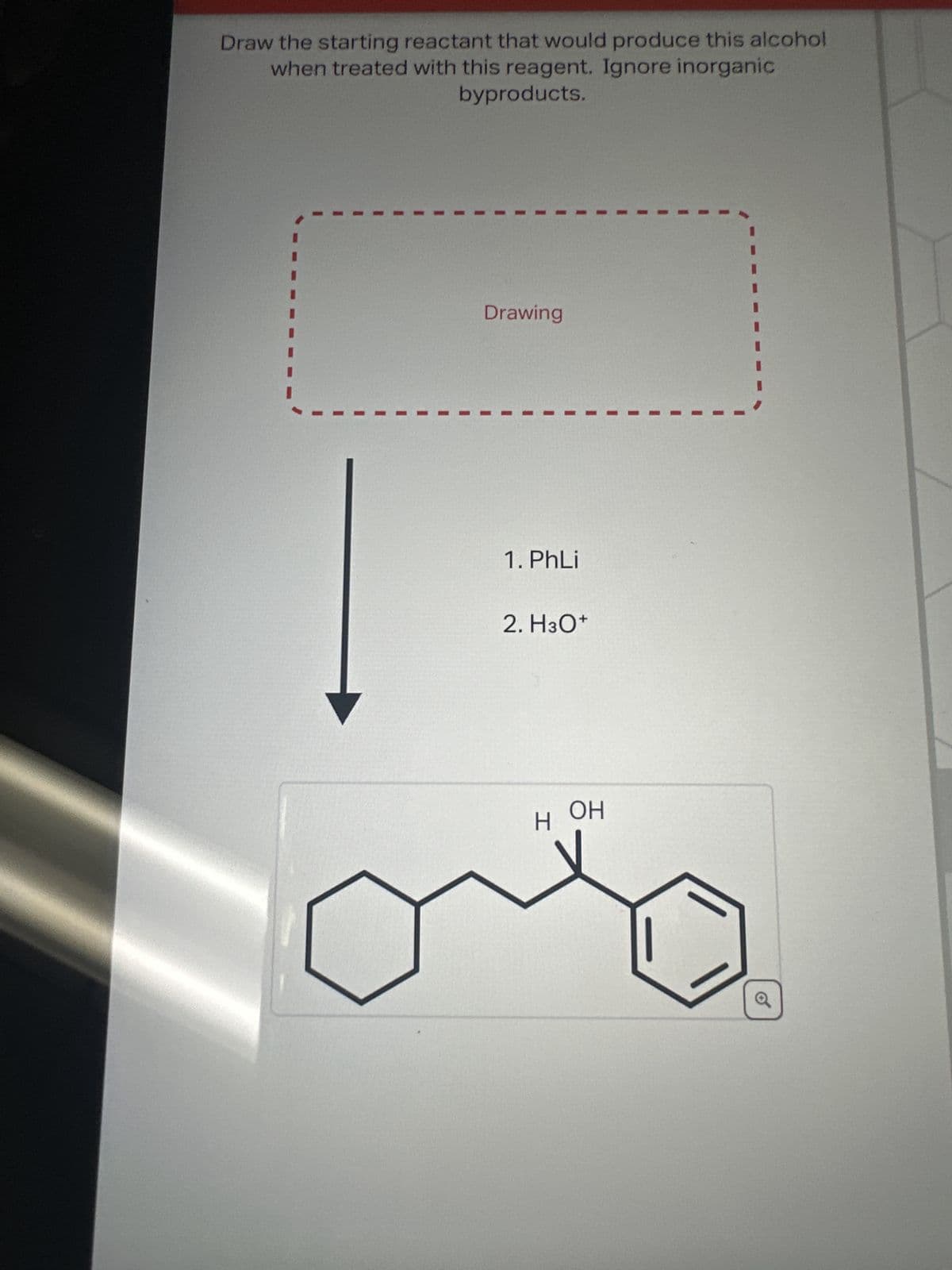
Draw the starting reactant that would produce this alcohol when treated with this reagent. Ignore inorganic byproducts. Drawing 1. PhLi 2. H3O+ HOH
Q: Learning AA prod03-cnow-owl.cengagenow.com Login Learning Learning × Online tea... y dr. marlow...…
A: Given:G°f(BrF3(g)) = -229.4KJ/molG°f(Br2(g)) = 3.1 KJ/molG°f(F2(g)) = 0.0 KJ/mol Balanced chemical…
Q: יוון UIPAC name of this compound?
A: The IUPAC nomenclature of organic compounds follows this naming format:Writing the name of organic…
Q: 5) ✓ PbS04 → PbSO3 + _ 02 6) _ Al + Fe2O3 → Fe + . Al203 7) SF6 → S + F2 8) _ C2H4 + 02 → CO2 + _ 9)…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Step 1: DecarboxylationDecarboxylation goes through a concerned six membered transition state.Normal…
Q: Suggest a structural reason why tertiary alcohols are more reactive than secondary alcohols with…
A: Reactivity with Hydrochloric Acid:Tertiary alcohols are more reactive than secondary alcohols when…
Q: The concentration at which micelles first form in solution is known as critical micelle…
A: Detailed explanation:The determination of CMC can be achieved through various experimental…
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Fe(s) + O2(g) → 2FeO(s) ΔΗ…
A:
Q: :$;$;$;:$:&;&;&&;&
A:
Q: Part II: Kinetic Energy and Speed Sketch and compare the distributions for kinetic energy and speed…
A: Step 1:
Q: an efficient synthesis for each of the following compounds using the 22.39 Propose acetoacetic ester…
A: Step 1: a)1. Acetoacetic ester with sodium ethoxide, deprotonates the acidic hydrogen forming…
Q: Mail-Giacove... J. - Outlook Account Setti... Course Hero HEM101_0W1 SHARPE - INTRO TO CHEMISTRY…
A: Final Answer (See explanation below):To draw the "box notation" electron configuration of chromium…
Q: A weak base has a K₁ of 5.6x104, determine the pK₁ of the base. 3.25 7.64 10.75 6.36
A: The pKb of a base is the negative logarithmic of Kb value of the base. pKb = - log Kb As per the…
Q: Predict the major product of the following transformation: CO₂Et H₂O+ Heat C10H100 Modify the given…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 4.00 g NaOH(s) (39.996 g mol–1) dissolves in 100.0 g of water in a constant–pressure calorimeter.…
A: Step 1:Moles of NaOH= molarmassmass= 39.996g/mol4.00g= 0.100 mol.Given,…
Q: Help
A:
Q: A rock found in the Australian Outback contains 438 g of Pb-206 for every 1.00 g of U-238. How old…
A: The objective of the question is to determine the age of a rock based on the ratio of Pb-206 to…
Q: Please answer in tipping format
A:
Q: Using thermodynamic values from Misra appendix 5, NOT R&H95, write reactions that solubilize…
A:
Q: Give the IUPAC name of the product formed in the multi-step synthesis reaction below. The first step…
A:
Q: Balance the following equation in basic solution using the lowest possible integers and give the…
A: Ag+ + I- -----> Ag + IO3- Step 1:Determine oxidation number of each element:Ag+ has…
Q: OH What is true about the following acid-base reaction? OH is the weakest base due to resonance…
A:
Q: In class we said that an approximate form for the bond stretch potential is V = k. -stretch (r-r)².…
A: The objective of the question is to derive an expression for the root mean squared (RMS) stretch…
Q: The write-up says “A C18-silica column is used in this experiment, a 0.46 x 15 cm column with 5-µm…
A: Column dimensions: Particle size: Flow rate: Buffer concentration: 0.010 MBuffer pH: 7.2
Q: Select the most appropriate reagent for the synthon shown below: A) (A) B (B) C) (c) (D) OH la you…
A: Step 1: Synthon : Synthon is a generalized fragments of a chemical compound which can be formed…
Q: Identify how you would make pentylamine from 1-hexanol: OH NH2 The transformation above can be…
A: 1-hexanol is first treated with PBr3 to replace -OH with -Br via SN2 reaction.Afterward, a strong…
Q: You want to quickly set up a temporary water bath in your lab with a volume of 10.0 L and a…
A: Step 1:Volume of water is 10LFinal temperature is 37oCInitial temperature of hot water is…
Q: What is the product of this acid base reaction assuming
A: Amides in the presence of a strong acid proceed to promote a hydrolysis reaction. When this occurs,…
Q: dont provide handwriting solution ....
A: The objective of the question is to classify the given chemical compounds into primary, secondary,…
Q: None
A: Step 1:To find out the exact molecule from the H-NMR, the following steps are used:According to the…
Q: What is the final temperature when 100.0 g of Al (s = 0.900 J g–1 °C–1) at 0.0°C is placed in…
A:
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: O Chemical Reactions Solving for a reactant in solution 0/5 Bis One way the U.S. Environmental…
A: Given is:Volume of sample = 200 mL = 0.20 Lmass of silver chloride collected = 5.9 mg = 5.9x10-3 g…
Q: None
A:
Q: OH CO₂H + NH₂ SO3H 1. Give the mechanism for the reaction 2. Discuss the reason(s) why the dye…
A: The question is asking for the mechanism of the reaction between OH CO2H and NH2 SO3H, the reason…
Q: According to the equation: Ca(OH)2(aq) + 2HCl(aq) 2H2O (l) + CaCl2 (aq) The balanced ionic equation…
A:
Q: The half-life of Ra-226 is 1.60 x 10^3 years = 5.05 x 10^10 s Find k What is the activity in…
A: Step 1: Step 2: Step 3: Step 4:
Q: Can you explain the problem?
A: Step 1: Step 2: Step 3: Step 4:
Q: 1) Provide the structure of the major product, which results from Diels-Alder reaction. +…
A:
Q: 5) 2 PbS04 → IN PbSO3 + 02 Type 6) _ Al + Fe2O3 → Fe + _Al2O3 5 Type 7) SF6 → S + F2 Typ 51 D 032 8)…
A: Step 1: Step 2: Step 3: Step 4:
Q: 3) Provide the structure of the major organic product in the following reaction. CH3 4) Classify the…
A: Step 1: Step 2: Step 3: Step 4:
Q: Table 1 Selected standard reduction potentials for half-reactions red measured in Reduction…
A: explained above
Q: 2. Starting with 2-methyl-1-phenylpropan-1-one, show how you can convert this to 2-methyl-1-…
A: The objective of the question is to convert 2-methyl-1-phenylpropan-1-one to…
Q: The lone pairs of which heteroatoms(s) contribute to the aromaticity of the following molecule? N $
A: Step 1:The heteroatoms present in the given ring structure are:Oxygen and nitrogen. Nitrogen…
Q: Gallium-67 (t1/2 = 25 hours) is used in the diagnosis of tumors. If a patient ingests a compound…
A: The objective of this question is to determine the amount of Gallium-67 remaining in the patient's…
Q: Heating D-altrose with dilute acid produces a nonreducing anhydro sugar (C6H1005). Methylation of…
A: The objective of the question is to understand the structure of the anhydro sugar formed by heating…
Q: Can you explain the problem? How do you identify them?
A: Step 1: SolutionThe removal of one proton (H+) from a given compound gives us a conjugate base. We…
Q: Please correct answer and don't use hend
A: Step 1:If there were acetal present with oxygen atoms in place of the sulphur, then the hydrolysis…
Q: Predict the product(s) for the following reaction. The answer is shown, please help with the…
A: Step 1: Step 2:MechanismStep 3: Step 4:
Q: A 50.0 mL sample of 0.0594 M AgNO3(aq) is added to 50.0 mL of 0.100 M NaIO3(aq). Calculate the [Ag+]…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Data Table Test mL of mL of pH tube HOAc NaOAc Trial Trial pH HOAC pH Average Millimoles Millimoles…
A: To calculate the average pH and average calculated pKa, we first need to determine the…
Step by step
Solved in 2 steps with 1 images

- Draw the reaction scheme. Use one arrow direct from starting materials to products.Acetal product formation requires an acid catalyst in the second step, but NOT in the first step. True or False?draw the major product of this reaction. ignore inorganic byproducts and phthalhydrazide side-product nh2nh2 koh

