Q: CIA Trial 1 Trial 2 Trial 3 Trial 1 Trial 2 Trial 3 Volume change(50 Volume change(50 Volume change(...
A: The above reaction is kinetic reaction of chemical bleach and dye, where all of the reactant is adde...
Q: H OH H но. NHCH, CH,NHCH, HO, CH HO (-)-ephedrine (-)-epinephrine (adrenalin)
A:
Q: 43. La method used for two immiscible liquids, that is liquids that do not mix together. O Distillat...
A:
Q: G and h
A: Given:
Q: 15.5 (Pages 650 -656), 15.7 (Pages 661-666) Suppose that a catalyst lowers the activation barrier of...
A: Given : Temperature = 298K
Q: he wavelength in
A:
Q: assuming
A: According to the question, After 53.0 min, 16.0% of a compound has decomposed. We need to determine ...
Q: What is the proper cell notation for the reaction: Cu + 2Fe^3+ = Cu^2+ + 2Fe^2+ * O Cu(s) | Cu*+2 (a...
A:
Q: To raise the temperature of aluminum which has a mass of 200 grams from 298 K to 348 K, 8400 joules ...
A: specific heat of aluminum, c= 840 J/kg.K
Q: What is the pH of the buffer that is composed of 0.10 M Na2HPO4 and 0.15 M KH2PO4? (Ka1=7.5 x 10-3, ...
A:
Q: moles
A:
Q: The specific heat of a certain type of metal is 0.128 J/(g.°C). What is the final temperature if 305...
A:
Q: A solution made up of 2.00 g of an unknown solute and 275 mL of water (d = 1.00 g/mL) has an osmotic...
A: Given: Mass of solute = 2.00 g The volume of solution = 275 mL Osmotic pressure = 0.872 atm Tempera...
Q: Using the data in the table, determine the rate constant of the reaction and select the appropriate ...
A: K is equilibrium constant
Q: A. Water and chloroform has a partition coefficient of 5.00. A 50.00mL sample of a 8M aqueous soluti...
A: The partition coefficient for water and chloroform is given to 5.00. We are required to calculate th...
Q: Q1 Complete the following table: [OH¯] pH [H,O*] Acidic, Basic, or Neutral? 2.0x10-6 9.8 3.5x10-3 Ne...
A:
Q: Determine the cell potential of the following system: a) Pb/PbSO4//Pb2+/Pb
A:
Q: 61. This process is called Paper chromatography Thin Layer Chromatography Column chromatography
A: Chromatography is a technique in which we separate a single chemical compound from a mixture. If the...
Q: 67. This compound is called Steroid Glycerin Wax
A: Steroid contain 4 ring fused ring in a perticular manner Glycerine is saturated hydrocarbon compoun...
Q: e redox reaction, us
A:
Q: The phase diagram for carbon indicates the extreme conditions that are needed to form diamonds from ...
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only. ...
Q: The equilibrium binding constant of cocaine to dopamine transporter is 10* at 25 °C and 5×107 at 40 ...
A: Given: Initial temperature (T1) = 25 oC Final temperature (T2) = 40 oC Equilibrium constant at 25 oC...
Q: - Name the following compounds. NH, CH, - сн-соон - NH - CH;-CH, NH,
A: Since you have posted a question with sub-parts , we are entitled to answer the first three sub-par...
Q: What is the wavelength (in nm) of blue light that has a frequency of 6.69 x 1014 s1? (c = 3.00 × 10®...
A:
Q: Four isomers A-D with the formula C5H12O exhibit different reactivity patterns as indicated below. ...
A: We have to determine the structure of isomer A,B,C,D.
Q: 12) Provide the major organic product of the following reaction. Br 1. Li 2. Н,СО 3. НО"
A: Solution-
Q: Consider the redox reaction: Al + MnO4^- + 2H2O → Al(OH)4^- + MnO2. The chemical species being oxidi...
A:
Q: Illustrate how electrochemical cell can be used as an alternative source of energy. Give examples.
A: Electrochemistry is a branch of chemistry that relates electricity and chemical reactions associated...
Q: What are the components of the buffer solution when 0.40 mol H3PO4 is mixed with 0.50 mol of a stron...
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The buffe...
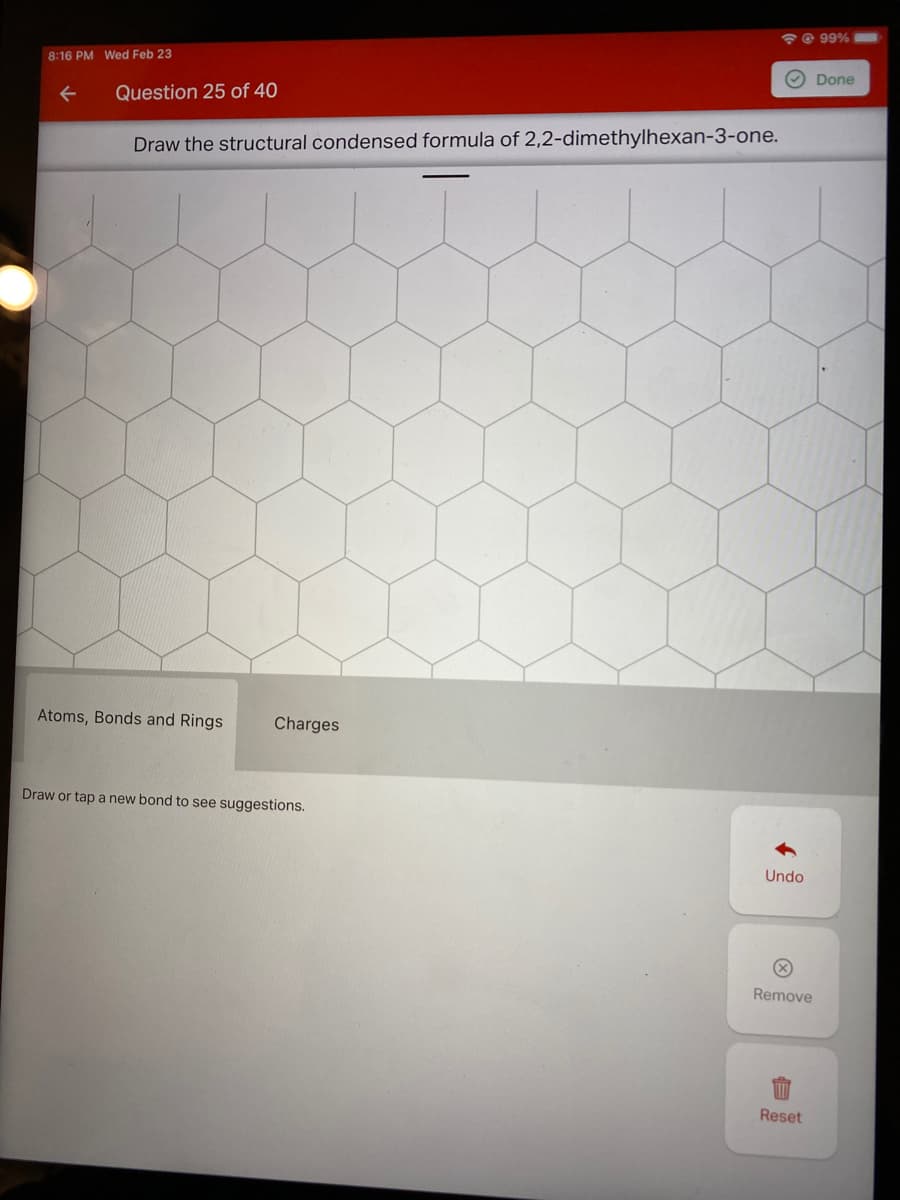
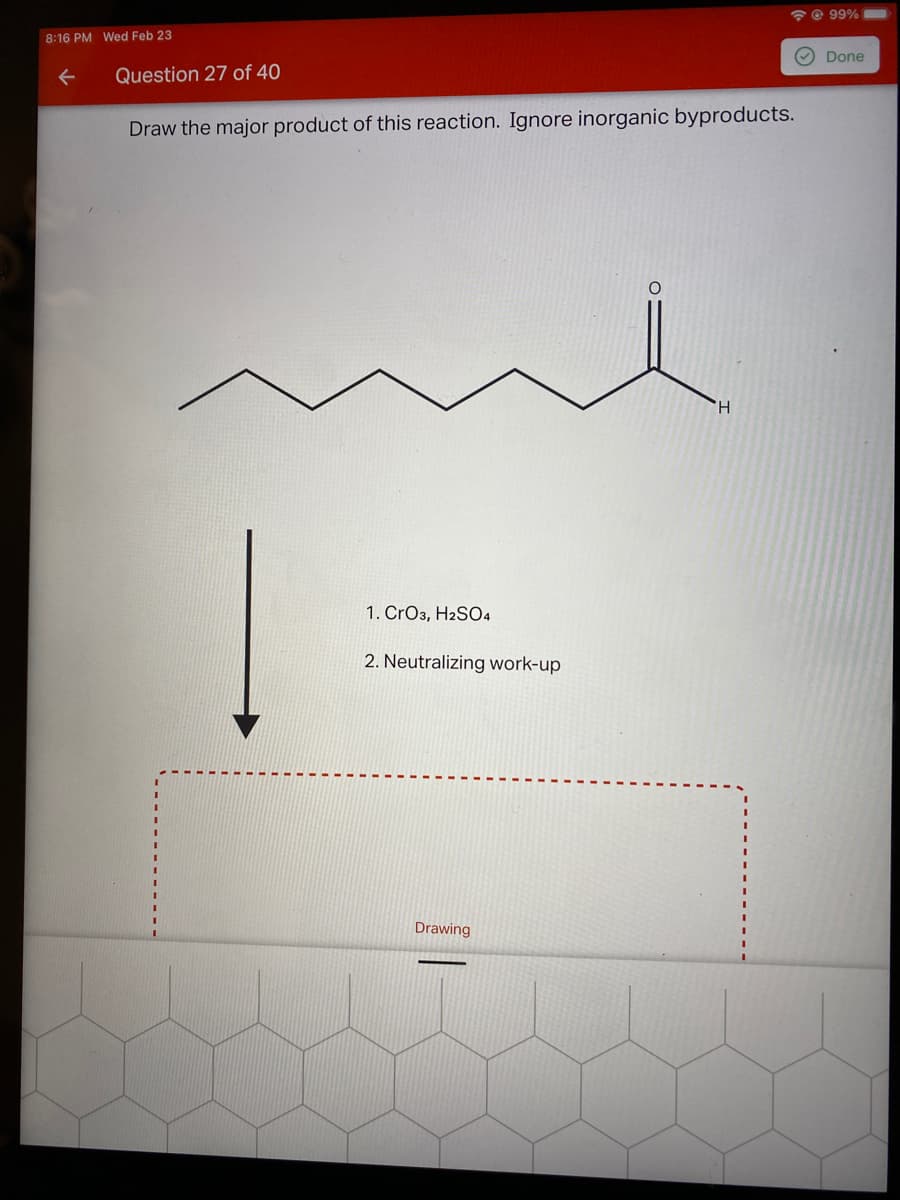
Q: Draw the major product of this reaction. Ignore inorganic byproducts. H 1. CrO3, H2SO4 2. Neutralizi...
A: In ketone and aldehyde both have carbonyl group. Due to the electronegativity difference between car...
Q: When measured at 22°C, a particular reaction has a rate constant of 5.8x10 /s and an activation ener...
A:
Q: Provide an example of a solution that contains a nonvolatile saw you dissolved in water. Statewood s...
A: Most seen example for the solution that contains non volatile solute dissolved in water is Urea diss...
Q: 58. This picture is an example of: " Vander Wals Force of Attraction O Dipole-dipole Interaction O H...
A: The attraction forces between the different molecules are known as intermolecular forces. Different ...
Q: Ph CH3 Ph-P= CHs Ph A B G) AICI3 (Give both products)
A:
Q: The conversion of ammonium cyanate into urea is a second order reaction. If the initial concentrati...
A:
Q: 45. The amount of energy required to break one mole of bonds of a type so as to separate the molecul...
A: Definition :
Q: Briefly describe and explain how to build a stable and reliable three-electrode cell using typical c...
A: The three electrode system consists of a working electrode a counter electrode and a reference elect...
Q: 65. This is an example of, hybridization." 109.5° A sp sp2 O sp3
A:
Q: The particle on a ring is a useful model for the motion of electrons around the porphine ring, the c...
A:
Q: b) 1.881x10-3g of copper (II) sulfate in 122mL of solution
A:
Q: I. Directions: Draw the lewis structures of the neutral atom and the ion of each element. Determine ...
A: Given,
Q: The following data show the rate constant of a reaction measured at several different temperatures. ...
A: The rate constant v/s temperature data given is,
Q: average rate
A:
Q: 16.85 To which of the following would the addition of an equal volume of 0.60 M NaOH lead to a solut...
A:
Q: How many grams of oxygen gas is required to completely react with 77.29 g pf ethane (C2H6) in the ba...
A:
Q: Temperature ("C)- Given the phase diagram for CO, which phase is most likely to exist at the followi...
A:
Q: The rate constant for this second‑order reaction is 0.540 M−1⋅s−1 at 300 ∘C. A⟶products How long...
A: Given: Reaction is second order. Rate constant of the reaction = 0.540 M-1s-1. And the concentration...
Q: An emulsifier: A . to control insects and vermin b. produces and stimulates the production of CO2 c....
A:
Q: What is substance examples?
A: To define: A substance.
Q: Convert your answer to scientific notation (i.e. ## x 10x mg) = Convert a 3.5% NaCl solution to µg/...
A: Given: Concentration of NaCl in %(m/v) = 3.5 %.
Step by step
Solved in 2 steps with 2 images

- 3. Label nonequivalent hydrogens in the structures below with numbers (1, 2, 3, etc.)The following molecule, the silicon analog of formaldehyde, has never been successfully prepared. H2SiO or H2Si=O The following reaction was attempted by researchers in order to synthesize this unusual compound: H2Si=S + P4O10 → H2Si=O + P4O9S None of the desired compound (H2Si=O) was obtained. Instead, the following molecule was obtained: (H2SiO)2 a molecule in which the O and Si form an …Si-O-Si-O… square. Further research indicated that although H2Si=O formed, it immediately formed (H2SiO)2 and released a substantial amount of energy according to the reaction: 2 H2Si=O → (H2SiO)2 + heat Based on the information above and your knowledge, what can be concluded about the relative strength of the Si-O and Si=O bonds? Question 24 options: A) An Si=O bond is stronger than two Si-O bonds. B) An Si=O bond is weaker than two Si-O bonds. C) An Si-O bond has the same strength as an Si=O.…I put in the answer for compound A, (dimethyl‐lambda4‐sulfanylidene)cyclopropane, and the product 9‐oxadispiro[2.0.44.13]nonane, in the boxes, but it says it's incorrect. What am I not getting?Thank you for your help!
- Give good explanation Asap Thanks calculate the empirical and molecular formula and degrees of saturation. Carbon= 65% Hydrogen= 6.1% Molar mass: 166g/mol what is the empirical formula? molecular formula: c9h__o3 fill in H, what is the molecular formula? degrees of saturation? round to the nearest whole number. is not 6.B + 2C ---> D + EI'm confused by the (CH3)2 at the final step. is there supposed to be a - connecting it? What is the name of the molecule? I've never seen a structure written like that
- 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.There are four cis-trans isomers of menthol (Problem 4-45), including the one shown. Draw the other three.
- __ Al(OH)3 + ___ HBr ------> ___ AlBr3 + ___ H2O Can yo balence this for meKindly xplain the solubility of alkanes, alkenes, and alkynes to H2SO4.a. Draw and name all of the isomeric products obtainedfrom the monobromination of propane with Br2/light. If halogenation were a completely randomreaction and had an equal probability of occurring atany of the C—H bonds in a molecule, what percentage of each of these monobromo products would beexpected?b. Answer part (a) using 2-methylpropane as the starting material.



