Q: Predict the major product for the following reaction: a [H+] (-H₂O) CH3 NH₂ ?
A: The given reaction is shown below.We have to give the major product of the given reaction.
Q: Draw the major organic product(s) of the following reaction. CI + NaOCH3 CH₂OH • You do not have to…
A: Organic reaction is given.We have to find out the products and draw the mechanism for both…
Q: HCI, MeOH reflux, 3 days 62% yield I
A: The synthetic transformation described involves the reaction under acidic conditions in the presence…
Q: Predict the products of this organic reaction: 'N' +H₂O + HCI Specifically, in the drawing area…
A: this is an acid hydrolysis reaction. amide reaction with water and strong acid (HCl) to produce…
Q: A typical vitamin C tablet (containing pure ascorbic acid, H₂C6H606) weighs 500 mg. One vitamin C…
A: The objective of this question is to calculate the pH of a solution made by dissolving a vitamin C…
Q: What volume of oxygen gas is produced when 0.682 mol of hydrogen peroxide (H2O2) reacts completely…
A: For the given reaction,temperature, T = = (0 + 273.15) K…
Q: Predict the product of the following organic reaction: 0 0 0 0 0=0 CH2–O–C—(CH2)3—CH=CH–CH2–CH3…
A:
Q: b) CHI NH-CH-CH g О -NH₂ + + но но->
A: According to Lewis acid-base theory, electron pair donor is base and electron pair acceptor is…
Q: What is the relationship between the parent structure on the left and the structure on the right?…
A: Same molecule ( identical compound ) has same configuration and same connectivity of…
Q: CH3CH2CH2C( O)CH2CH2CH3 pH 4-5 Draw Enamine
A:
Q: To preview image click here For the following cation choose the correct options for the following…
A:
Q: Consider these reactions, where M represents a generic metal. 2M(s) + 6 HCI(aq) 2 MCI, (aq) + 3H₂(g)…
A: The enthalpy change is a state function and the Hess law states that the change of enthalpy in a…
Q: Draw the product obtained from the reaction of cellobiose (drawn below) with each of the following…
A: 1) NaBH4 is a reducing reagent.It converts the hemiacetal to diol to the reducing end of the…
Q: Draw the expected product of the reaction. NIH NaNO, HC1, 0°C Select Draw Templates /m C Mare H N 0
A: The given reaction is .We have to give the product of the above reaction.
Q: The conductivity of the dilute NaCl solution should be substantially higher than that of DI water…
A: The dissociation of NaCl into its ions are given by,From this balanced equation, it is well clear…
Q: 1.) A sample of oxygen had an initial volume of 4.0L and was at a standard pressure of latm, what…
A: As per our guidelines we are supposed to do three subparts of a question. Kindly repost other parts…
Q: 4. Show the steps and reagents needed to transform 1-propyne into 2-methylheptane. You may use any…
A: Given is organic synthesis reaction. In this reaction 1-propyne is converted into 2-methylheptane.
Q: 100 grams of sodium is reacted with 100 grams of chlorine gas to give sodium chloride. Assuming a…
A: Mass of sodium = 100 gmass of chlorine gas = 100 gBalanced Chemical equation is,2 Na + Cl2 ---->…
Q: A. B. 2. Propose an efficient synthesis for each of the following transformations using any…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: What is the relationship between the parent structure on the left and the structure on the right?…
A: Enantiomers are a pair of molecules that exist in two forms that are mirror images of one another…
Q: . Na-24 has a half-life of 15 hours. How much of a 20.0 g sample would remai fter decaying for 60…
A:
Q: Specify a synthetic scheme that would produce the compound shown above in the fewest steps possible.…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Draw resonance forms of the compound below formed after the reaction of an amino acid with ninhydrin…
A: Amino acids are the organic compound contains both acid -COOH and amine -NH2 functional…
Q: Draw the product of this hydrogenation reaction. Ignore inorganic byproducts. I H₂ Pd/C Drawing " 1…
A: Given,The reactions:
Q: Choose a reagent from the table to bring about the following conversion: OHO (If more than one step…
A: N-Bromo succinimide (NBS) is the best reagent for allylic bromination. The reaction proceeds via a…
Q: 2. Fill in the missing product(s), reactant(s), and/or reagents. 1. KMnO4 H₂O 2. HCI Br₂ A KOIBu,…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Using the equation E = (hcRH) ()=(-2.18 x Xb calculate the energy of an electron in the hydrogen…
A: From given equation there is need to find energy of electron when it is in orbit (n) = 4
Q: pollution generated in automobile engines. The decomposition of nitric oxide (NO) to N₂…
A: Answer:Here:k1 and k2 are the rate constants at temperature T1 and T2 respectively.
Q: A solution contains 5.54x10-3 M sodium cyanide and 1.23x10-2 M potassium hydroxide. Solid nickel(II)…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: Please give me a technique on how to do these. Should I just remove the halide (e.g. Cl) then attach…
A: When the benzene reacts with an alkyl halide in the presence of a Lewis acid catalyst alkyl benzene…
Q: Which statements are true about amines? NH3+ is the form found in our blood amines can react with…
A: NH4+ is the form found in blood.1° Amine reacts with aldehyde to form imine and 2° amine reacts with…
Q: 100 grams of sodium is reacted with 100 grams of chlorine gas to give sodium chloride. How many…
A: Mass of Na = 100gMass of Cl2 = 100gMolar mass of Na = 22.99 g/molMolar mass of Cl2 = 71 g/mol
Q: Which of the following represent the strongest nucleophile? Options: NaSH in water NaSH in…
A:
Q: Which of the following take place when CH₂Cl₂ dissolves in water? [Select all that apply.] Some, but…
A: Methylene chloride is a volatile solvent that is used in industry. It is a colorless liquid.
Q: CIO, (aq) + The reaction 2 CIO₂(aq) + 2 OH(aq) CIO₂ (aq) + H₂O(1) was studied with the following…
A:
Q: For the molecule below: 1. Identify all CHIRAL centers if any are present: 2. Name each CHIRAL…
A: we have to find chiral centres and name the chiral centres.
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1 I excess CH3NH2 I I CI I I I…
A: Acid chloride reacts with a amine to form amide.
Q: What is the relationship between the parent structure on the left and the structure on the right? Me…
A:
Q: Hi, I am in chemistry 2 and am working on acids and bases. However im struggling with identifying a…
A: The question is based on the concept of salt hydrolysis. We need to identify the nature of given…
Q: Draw the major E2 reaction product formed when cis-1-chloro-2-ethylcyclohexane (shown) reacts with…
A:
Q: Draw the skeletal ("line") structure of dipropyl ketone. Click and drag to start drawing a…
A: Functional group : ) - CO - ( ketone ) Common names. In case of mixed ketones, name the alkyl…
Q: Draw the major product of this reaction. Ignore inorganic byproducts.
A: Enamines are formed by condensing alkenes and enamines in acidic conditions. The word enamine…
Q: Draw the Lewis structures for CH4, BeF2, PCI5, SO4^2-, C2H2, and SBr4
A:
Q: A solution contains 1.11x102 M ammonium carbonate and 1.23x10-2 M potassium phosphate. Solid barium…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: All of its output energy is absorbed in a detector that measures a total energy of 0.54 J over a…
A: Q. Given λ =988 nm = (wavelength emit by laser diode)ET = 0.54 J (total energy over a period of 34…
Q: Sodium carbonate is often added to laundry detergents to soften hard water and make the detergent…
A: Molarity of Ca2+ = = Molarity of Mg2+ = = Volume of water = 19.5 gallons = 73.8 L (1 gallon…
Q: 9) A COOH H₂N-C-H CH₂ CH₂ CH₂ CH₂ NH₂ 2.18 Coo H₂N-C-H CH₂ CH₂ CH₂ 8.95 Coo HN-C-H CH₂ CH₂ CH₂ 1 CH₂…
A: If the pH is greater than pKa then deprotonation occurs and it exists in deprotonation form. While…
Q: Write the Ksp expression for the sparingly soluble compound barium phosphate, Ba3(PO4)2- Kap A₂ A³…
A: Answer:Ionic compounds that have low solubility in water are called as sparingly soluble compounds…
Q: The cyanide ion is the conjugate base of the weak acid hydrocyanic acid. The value of K for CN", is…
A: A weak acid is defined as the acid that partially dissociates into its constituent ions in a…
Q: 11. The following tetrahedral intermediate a. propanoyl chloride and methanol b. propanoic acid and…
A:
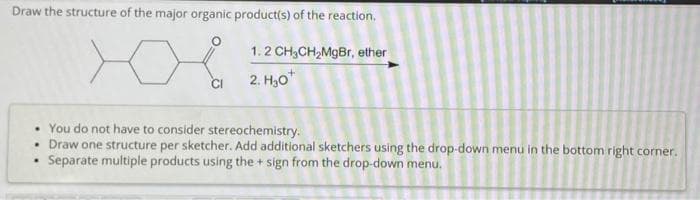
Step by step
Solved in 3 steps with 3 images

- Draw the structure of the major organic product(s) of the reaction. You do not have to consider stereochemistry. All carboxyl and amino groups should be drawn in the neutral form. If no reaction occurs, draw the organic starting material. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.This question has multiple parts. Work all the parts to get the most points. a Draw all of the monochlorination products that you might obtain from the free-radical chlorination of 2,2-dimethylbutane. You do not have to consider stereochemistry. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu.Draw the organic product(s) of the following reaction. You do not have to consider stereochemistry. Include cationic counter-ions, e.g., Na+ in your answer, but draw them in their own sketcher. If no reaction occurs, draw the organic starting material. Separate multiple products using the + sign from the drop-down menu.
- please help with part B:Draw the structure(s) of the major organic product(s) obtained after workup of the following reaction. You do not have to consider stereochemistry. If no reaction occurs, draw the organic starting material. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.Draw the structure(s) of the major organic product(s) obtained after workup of the following reaction. You do not have to consider stereochemistry. If no reaction occurs, draw the organic starting material. Draw structure(s) of product(s) after workup to neutralize acid. Include counter-ions, e.g., Na+, I-, in your submission, but draw them in their own separate sketcher. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
- See attached photo. Please show stereochemistry and regiochemistry.1) What is the major product for the reaction of 3-bromo1-butene and HBr? Give the name of the product. 2) For this product, how many chiral carbon(s) are there in the structure?3) How many stereoisomers could we write for this product? 4) Write the Fischer Projections of all stereoisomers. (attach a file or other ways you could show your work)a) Free radical bromination is more selective than free radical chlorination. Draw a reaction coordinate diagram for the specific step in the radical chain mechanism that illustrates the source of this selectivity, and explain your reasoning. b) Explain why the bond dissociate energy (BDE) of tert-butane is 95 kcal/mol while the BDE for propane is 99 kcal/mol.
- Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by the catalytic conversion of acetylene to benzene: 3C2 H2(g) ⟶ C6 H6(g). Which value of Kc would make thisreaction most useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answer.Which reagent is required to complete the reaction in Figure 11? [Generate a SMILES notation: https://jsme-editor.github.io/dist/JSME_test.html. Copy the notation (CTRL+C) and paste it as the answer to this question (CTRL+V).] * Your answerC6H5CO2CH2C6H5 can be synthesized by an SN2 reaction. Draw the structures of the alkyl chloride and nucleophile that will give this compound in highest yield. Use the wedge/hash bond tools to indicate stereochemistry where it exists. Include all valence lone pairs in your answer. Separate multiple reactants using the + sign from the drop-down menu. When a metal counterion is needed, use Na+, but draw it in its own sketcher.

