Q: Use your models to determine the absolute configuration of the chiral carbon atoms in the following…
A:
Q: A stock solution will be prepared by mixing the following chemicals together: 3.0 mL of 0.00200 M…
A: Molarity of Fe(NO3)3 used, C = 0.200 M Volume of 0.200 M Fe(NO3)3 used, V = 10.0 mL
Q: Br₂ H₂O
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Consider the following reaction where Ke = 10.5 at 350 K. 2CH₂Cl2 (g) CH4 (9) + CCl4 (9) A reaction…
A:
Q: -3. (a) Provide retrosynthesis corresponding forward reaction for the following compounds : OH (i)…
A:
Q: Draw the skeletal isomers of chlorohexane, excluding those with branched carbon chains.
A: Isomers are those compounds which have same molecular formula but different position of atoms. If…
Q: Construct the (generic) molecular orbital diagram for homonuclear diatomic molecules for elements up…
A: MO diagrams predict a molecule’s physical and chemical properties such as shape, bond energy, bond…
Q: Name 20-Ne 22-Ne 35-Cl Element name 100 Notation 80 A Z P N
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: 2. Name each of the following functional Groups. H R-C-OH R-C-OH H OH HICH R-C-0-CH₂ H H R-C=CH₂…
A: Names of following functional groups will be as following :
Q: A marble weighs 4951 µg. How much does it weigh in grams? Please round your answer to the 5…
A: Consider the given information is as follows: Weight of the marble = 4951 µg Weight of marble in…
Q: 2. Cite some factors that could possibly affect the AH in this experiment. 3. Glucose, C6H12O6, is…
A:
Q: Subtract 3.58 * 10 ^ - 2 kg from 1.41 * 10 ^ 11 ng and report your answer in g
A:
Q: You are given a 27 % (w/v) solution of psicose. Molar…
A: Given, Note: 1 μmoles = 10-6 moles moles of psicose = 450 μmoles = 450 × 10-6 moles % (w/v) of the…
Q: Add or subtract the following measurements. Be sure each answer you enter contains the correct…
A: Given4.50 mL + 0.827 mL = mL11.827 mL + 8.9 mL = mL14.97 mL + 9.800 mL = mL
Q: Convert into a line-angle formula CH3(CH2)3CH(CH₂CH3)CH₂CH3
A:
Q: (b) With proper reasoning, provide the retrosynthesis and forward synthesis for the following…
A:
Q: Express the concentration of a 0.0590 M aqueous solution of fluoride, F, in mass percentage and in…
A:
Q: How many minutes are there in a solar year
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 8. Draw the Lewis structures for two resonance forms of the following moleculesions. Show any formal…
A: Lewis dot structure is diagrammatic representation of molecules in which each atoms are represented…
Q: The following are three molecular orbitals for the heptatrienyl cation: 08.0 For each MO, a.…
A: Here it is given the three π molecular orbitals for the heptatrienyl cation. There are total 7…
Q: A sprinter set a high school record in track and field, running 200.0 mm in 20.9 ss . What is the…
A:
Q: Draw resonance structures for the following compound: 요
A:
Q: Consider the electrochemical cell diagram shown below. As you observe the reaction in the cell, you…
A:
Q: What is 72.0 °F in C and K?
A: Answer: To solve this question we have to use the relation between Fahrenheit and Celsius, also in…
Q: Solve the following to the correct number of significant figures. Problem 1. (0.950m)x(6.21x10-3m)…
A:
Q: How many mL of a KOH 2,251mol/L solution do we need in order to prepare 10,0 mL of a solution that…
A: This is a problem based on general chemistry. Here we are provided with a stock KOH solution of…
Q: A chemist dissolves 592. mg of pure barium hydroxide in enough water to make up 160. mL of solution.…
A: At 25 oC, the solubility of barium hydroxide, Ba(OH)2 is approximately 4.70 g / 100 mL water. Since…
Q: Choose the name of the following functional groups: d De as AQ NH₂ gQ h NH₂ Qué
A: The special groups present in any molecule due to which the compound is classified into different…
Q: A piece of metal has a mass of 6g and it displaced 3 mL of water in a graduated cylinder. What is…
A:
Q: A mixture of NaH2PO4 and Na2HPO4 are used to form a 10mM phosphate buffer at pH 7.4. Determine the…
A:
Q: Obtained at different initial pressures of substance A in the reaction A → B + 2C in the gas phase.…
A:
Q: ▼ ▼ Part A CH3-0-CH₂-CH3 Spell out the IUPAC name of the compound. Submit Request Answer Part B…
A: IUPAC nomenclature rule for ether: 1) first you need to identify the alkyl groups on both the side…
Q: How many moles of mercury are in 17.140 grams of mercury?
A: Atomic number of mercury is ---- 80 Atomic mass of mercury is ----- 200.59 g Since atomicity of…
Q: first volume is 2.22L, which exerts a pressure of 450mmHg. If the volume changes to 4.8L, what is…
A:
Q: Ultrapure silicon is used to make solid-state devices, such as computer chips. If the volume of a…
A:
Q: [1.5] [1,4]
A: The sigmatropic shift is the movement of substituents and the loosely bound pi electrons to form…
Q: Nernst equation,
A:
Q: Determine the moles of sodium carbonate that need to be taken from the concentrated solution. (To…
A: Given data : Concentrated solution conc. (M1) = 2.80 M Diluted solution conc. (M2) = 0.200 M Diluted…
Q: The correct IUPAC name for the following molecule is CH₂ CH₂-C-CH₂-CH=CH₂ ÓH a)…
A: Here we are required to find the IUPAC name for the given organic compound.
Q: What is the mechanism of the major product in the following reaction? CI a) E2 d) SN2 D CH3-O: b)…
A: Given incomplete reaction is : What is the mechanism of the major product of this reaction ?…
Q: Which set of values is thermodynamically AG/KJ >0 >0 0 impossible? AS/J K¹ >0 0 <0 Keq MAAAB
A: Given reaction is : Which set of values is thermodynamically impossible ? Table is : Here, ΔG =…
Q: 12. A solid compound has a mass of 43.25 g. You scrape 2.5 g of the material off of the solid for…
A:
Q: raw the major organic product of the Bronsted acid-base reaction between the compounds shown below.…
A: -> In Bronsted acid base reaction base can abstract most acidic hydrogen hence, conjugate base is…
Q: 2. How many units of unsaturations does a compound with the formula C25H40Cl4 have? 3. Give the name…
A: For question 2, we have to find the number of units of unsaturation in the compound C25H40Cl4. For…
Q: Which reagents would you choose for the following reaction? ? CH₂-CH₂-CH₂-CH₂Br a) CH₂CH₂ONa d) NaOH…
A:
Q: 2. 25 red glass beads weighed 19.5 grams. How many milligrams (mg) is this? Show calculations. 3.…
A: Unit conversion factors: 1 gram = 1000 milligrams 1 centimetre = 10 millimetres 1 litre = 1000…
Q: Which of the following graphs best represents the change in the total energy vs. temperature as N₂O…
A: In the given phase diagram of N2O, The far left region shows the solid phase of N2O. The far right…
Q: Fill in the blanks. The positions of atoms on the periodic table of the ____ are determined by how…
A: The positions of atoms on the periodic table of the elements are determined by how many protons are…
Q: What is the molar mass of glycerol if a solution of 5ml glycerol in 25ml of water resulted in a…
A:
Q: (b) Provide forward reaction) compounds provided : (i) (ii) NC suitable retrosynthesis for the CN as…
A:
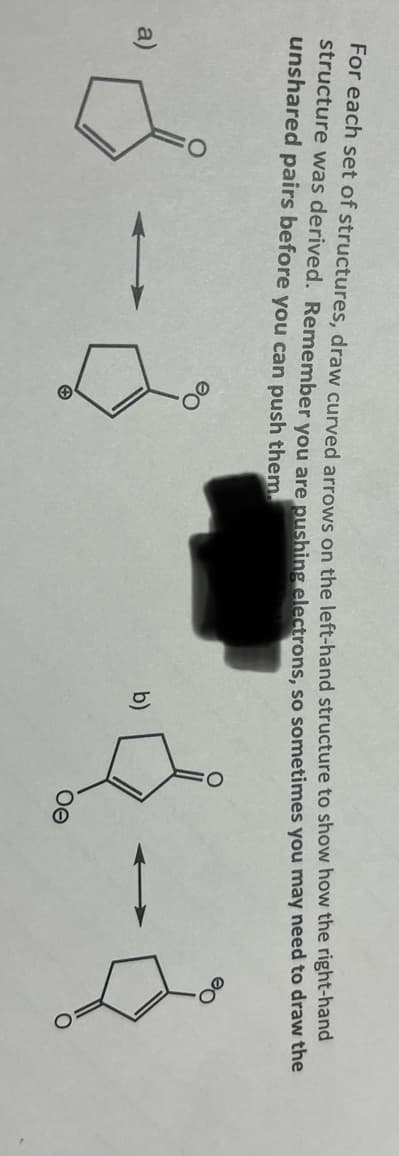
Step by step
Solved in 2 steps with 2 images

- 1. (f) Provide a name for each molecule (I narrowed it down to two subparts)Question: How many C-types do each of the following compounds have? Label them a, b,c ... And Circle the carbon type that would be the most downfield. Please answer all 4 parts of the question. Thank you!Re-draw each of the following making sure to include lone pairs and formal charges in each drawing
- Propose a curved-arrow mechanism for this reaction. Use curved arrows to show movement of electron pairs, and draw structures for all important reaction intermediates. Be sure to show all lone pairs and nonzero formal charges.Why do I number the substituents in this direction in the naming of these compounds? That is, how do we know whether to number the substituents clockwise or counterclockwise?Draw the line-angle structure of the following compounds if applicable. note: I need the answer immediately. I will send a good rate right away as well.
- Label All the functional groups present in the structures below (to the right). Note that you should choose the higher priority functional groups.Compound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-ynewhere are the head arrows. Please give me the lone pairs as well. thank you

