Electron- # Molecule domain Geometry Hybridization of Central Dipole moment? Yes or Atom No. 1 CO2 Part K yes ds sp Complete the fifth row of the table. no trigonal planar 4 no sp sp² 5 SF4 Hybridization of Central atom octahedral Osp no sp3 sp? yes Not applicable trigonal bipyramidal 8. no Submit Request Answer 9 XeF2 Pearson Copyright O 2020 Pearson Education Inc. All rights reserved. I Terms of Use Privacy Policy I Pern 100% arch 2. 3. 7, CO
Electron- # Molecule domain Geometry Hybridization of Central Dipole moment? Yes or Atom No. 1 CO2 Part K yes ds sp Complete the fifth row of the table. no trigonal planar 4 no sp sp² 5 SF4 Hybridization of Central atom octahedral Osp no sp3 sp? yes Not applicable trigonal bipyramidal 8. no Submit Request Answer 9 XeF2 Pearson Copyright O 2020 Pearson Education Inc. All rights reserved. I Terms of Use Privacy Policy I Pern 100% arch 2. 3. 7, CO
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter10: Molecular Geometry And Chemical Bonding Theory
Section: Chapter Questions
Problem 10.18QP: Which of the following molecular geometries does the XeF5+ cation exhibit? a Tetradral b T-shaped c...
Related questions
Question
The fifth row's electron domain geometry is trigonal bipyramidal.
does it have a dipole moment?
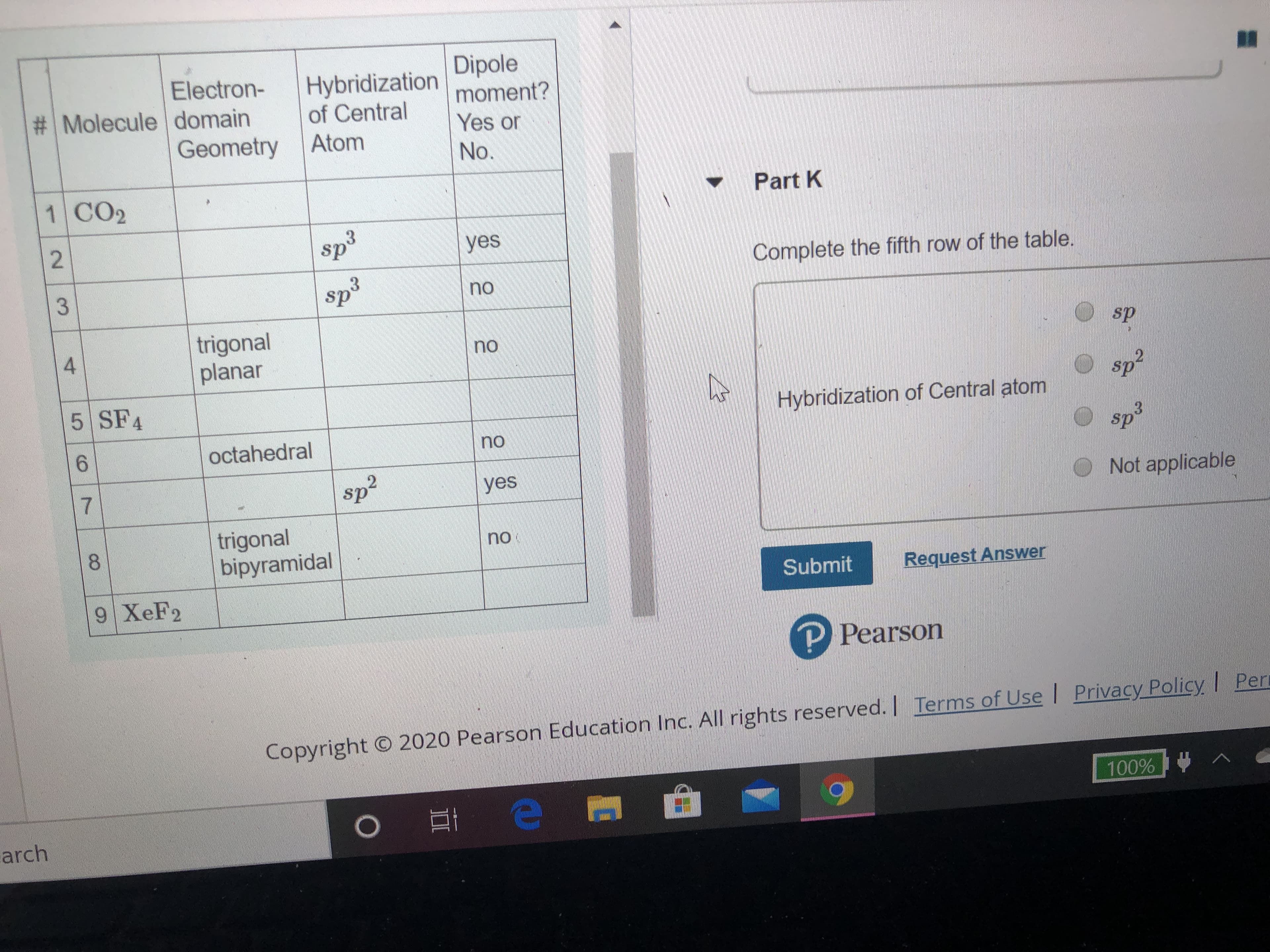
Transcribed Image Text:Electron-
# Molecule domain
Geometry
Hybridization
of Central
Dipole
moment?
Yes or
Atom
No.
1 CO2
Part K
yes
ds
sp
Complete the fifth row of the table.
no
trigonal
planar
4
no
sp
sp²
5 SF4
Hybridization of Central atom
octahedral
Osp
no
sp3
sp?
yes
Not applicable
trigonal
bipyramidal
8.
no
Submit
Request Answer
9 XeF2
Pearson
Copyright O 2020 Pearson Education Inc. All rights reserved. I Terms of Use Privacy Policy I Pern
100%
arch
2.
3.
7,
CO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax