Dipole Electron- # Molecule domain Hybridization of Central moment? Yes or Geometry Atom Part F No. 1 CO2 Complete the third row of the table. sp3 yes GaH3 no sp3 trigonal planar O NH3 no Molecule O CH4 5 SF4 BRF3 octahedral no sp2 yes Нао trigonal bipyramidal no 8. Submit Request Answer 9 XeF2 P Pearson Copyright © 2020 Pearson Education Inc. All rights reserved. I Terms of Use Privacy Policy / P 100% earch 2. 4. Electron- Molecule domain Hybridization of Central Dipole moment? Geometry Atom Yes or No. Complete the fourth row of the table. 1 CO2 12 sp3 3. yes О НаСО sp³ O PF3 no Molecule O BH3 trigonal planar no BRF3 5 SF4 PC33 octahedral no 7. sp2 yes Submit Request Answer trigonal bipyramidal no 8. 9 XEF2 Part I P Pearson Copyright © 2020 Pearson Education Inc. All rights reserved. | Terms of Use Privacy Policy / Pe 100% search 4.
Dipole Electron- # Molecule domain Hybridization of Central moment? Yes or Geometry Atom Part F No. 1 CO2 Complete the third row of the table. sp3 yes GaH3 no sp3 trigonal planar O NH3 no Molecule O CH4 5 SF4 BRF3 octahedral no sp2 yes Нао trigonal bipyramidal no 8. Submit Request Answer 9 XeF2 P Pearson Copyright © 2020 Pearson Education Inc. All rights reserved. I Terms of Use Privacy Policy / P 100% earch 2. 4. Electron- Molecule domain Hybridization of Central Dipole moment? Geometry Atom Yes or No. Complete the fourth row of the table. 1 CO2 12 sp3 3. yes О НаСО sp³ O PF3 no Molecule O BH3 trigonal planar no BRF3 5 SF4 PC33 octahedral no 7. sp2 yes Submit Request Answer trigonal bipyramidal no 8. 9 XEF2 Part I P Pearson Copyright © 2020 Pearson Education Inc. All rights reserved. | Terms of Use Privacy Policy / Pe 100% search 4.
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter9: Bonding And Molecular Structure: Orbital Hybridization And Molecular Orbitals
Section: Chapter Questions
Problem 71SCQ: Bromine forms a number of oxides of varying stability. (a) One oxide has 90.90% Br and 9.10% O....
Related questions
Question
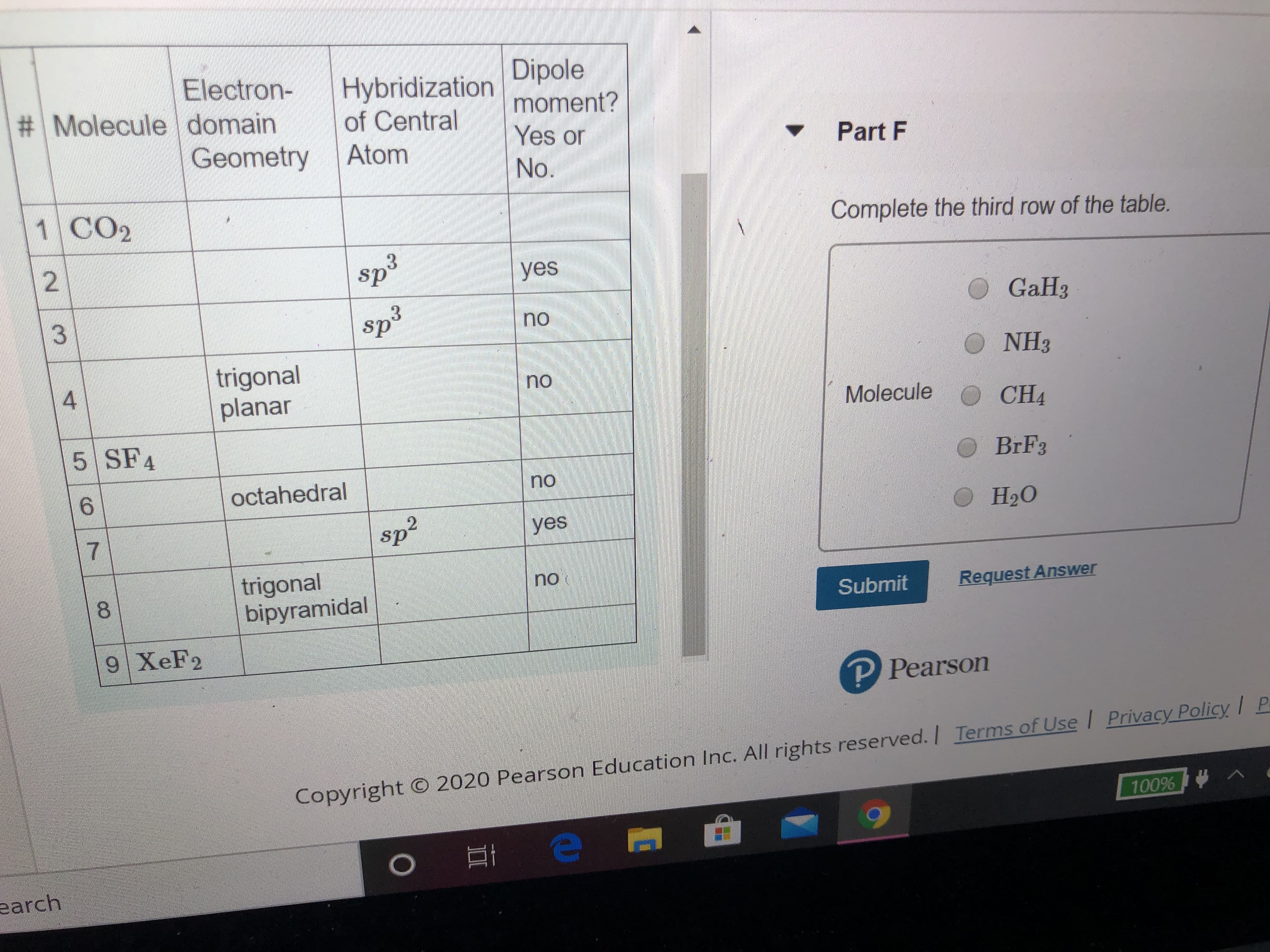
Transcribed Image Text:Dipole
Electron-
# Molecule domain
Hybridization
of Central
moment?
Yes or
Geometry
Atom
Part F
No.
1 CO2
Complete the third row of the table.
sp3
yes
GaH3
no
sp3
trigonal
planar
O NH3
no
Molecule O CH4
5 SF4
BRF3
octahedral
no
sp2
yes
Нао
trigonal
bipyramidal
no
8.
Submit
Request Answer
9 XeF2
P Pearson
Copyright © 2020 Pearson Education Inc. All rights reserved. I Terms of Use Privacy Policy / P
100%
earch
2.
4.
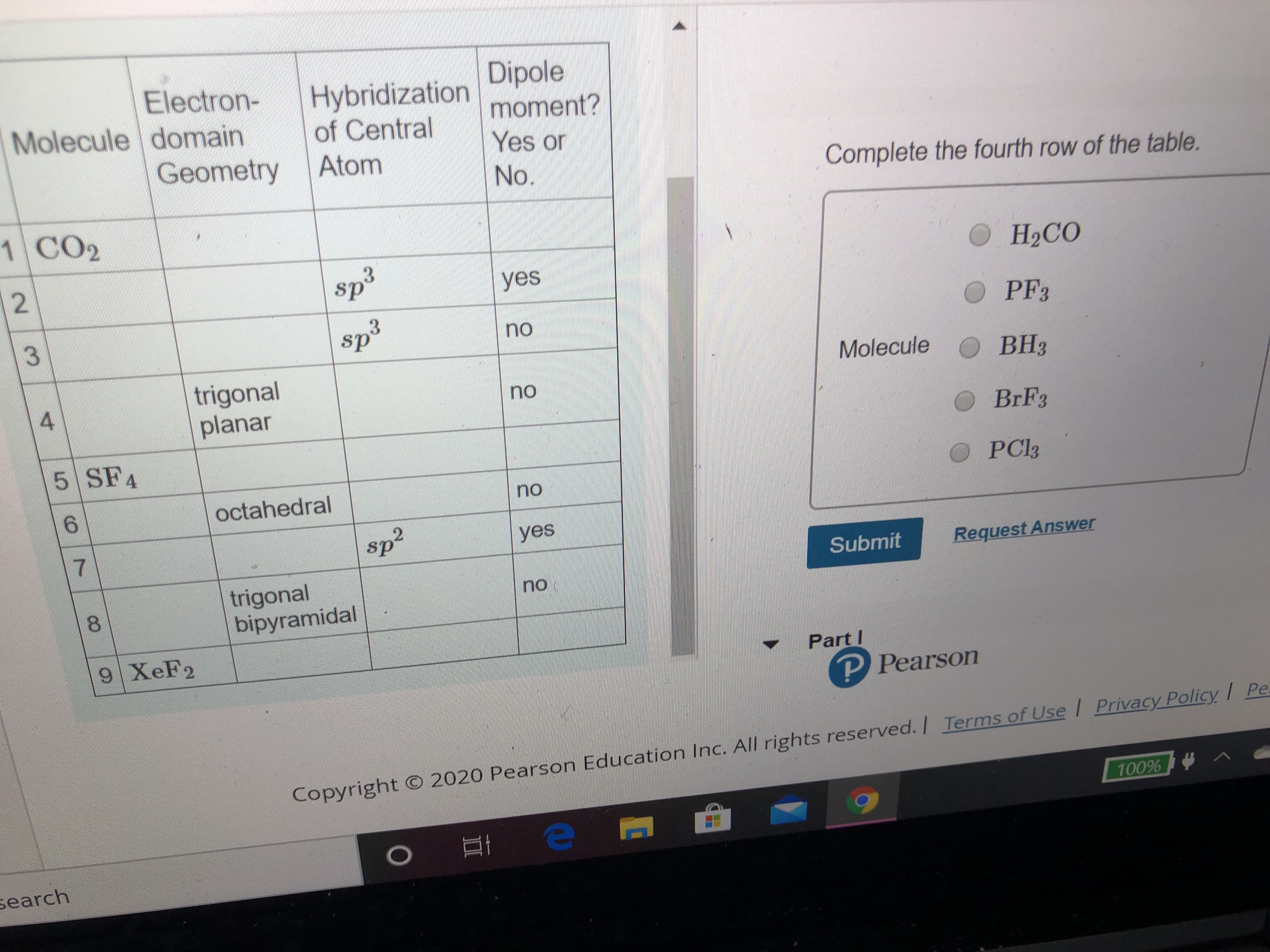
Transcribed Image Text:Electron-
Molecule domain
Hybridization
of Central
Dipole
moment?
Geometry
Atom
Yes or
No.
Complete the fourth row of the table.
1 CO2
12
sp3
3.
yes
О НаСО
sp³
O PF3
no
Molecule O BH3
trigonal
planar
no
BRF3
5 SF4
PC33
octahedral
no
7.
sp2
yes
Submit
Request Answer
trigonal
bipyramidal
no
8.
9 XEF2
Part I
P Pearson
Copyright © 2020 Pearson Education Inc. All rights reserved. | Terms of Use Privacy Policy / Pe
100%
search
4.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 5 images

Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning