emental carbon usually exists in one of two forms: graphite or diamond. It is generally believed et diamonds last forever. Here are the standard enthalpy of formation (ArH) and the standard olar entropy (S) values for diamond and graphite • Part A AH" (kJ mol-1) s° (J mol K-1) ubstance "raphile 5.740 What is the standard Gibbs energy for the transformation of diamond to graphite at 298 K? "alamond CaumondCyraphile 1.897 2.38 Express your answer to three significant figures and include the appropriate units. > View Available Hint(s) Tempjetes Symbols undo redo fesét keyboard shortcuts help A,G Value Units Submit
emental carbon usually exists in one of two forms: graphite or diamond. It is generally believed et diamonds last forever. Here are the standard enthalpy of formation (ArH) and the standard olar entropy (S) values for diamond and graphite • Part A AH" (kJ mol-1) s° (J mol K-1) ubstance "raphile 5.740 What is the standard Gibbs energy for the transformation of diamond to graphite at 298 K? "alamond CaumondCyraphile 1.897 2.38 Express your answer to three significant figures and include the appropriate units. > View Available Hint(s) Tempjetes Symbols undo redo fesét keyboard shortcuts help A,G Value Units Submit
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter16: Spontaneity Of Reaction
Section: Chapter Questions
Problem 62QAP: Consider the reaction NH4+(aq) H+(aq)+NH3(aq) Use G f for NH3(aq) at 25C=26.7 kJ/mol and the...
Related questions
Question
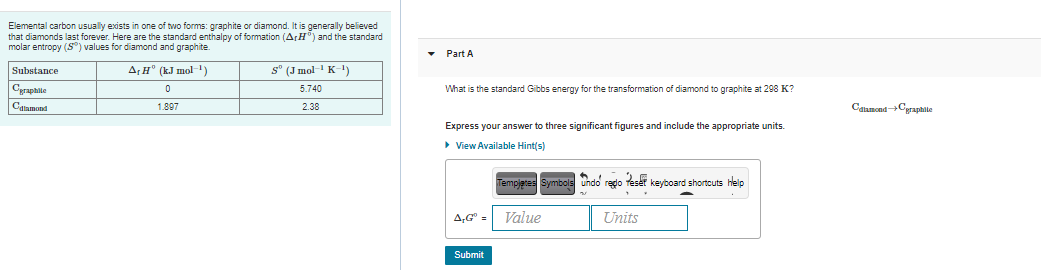
Transcribed Image Text:Elemental carbon usually exists in one of two forms: graphite or diamond. It is generally believed
that diamonds last forever. Here are the standard enthalpy of formation (ArH") and the standard
molar entropy (S) values for diamond and graphite.
Part A
Substance
A,H° (kJ mol-1)
s° (J mol K-1)
Cyrapaile
5.740
What is the standard Gibbs energy for the transformation of diamond to graphite at 298 K?
Calamond
CalamondCyraphite
1.897
2.38
Express your answer to three significant figures and include the appropriate units.
> View Available Hint(s)
Tempjetes Symbols undo redo fesét keyboard shortcuts help
A,G" =
Value
Units
Submit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning