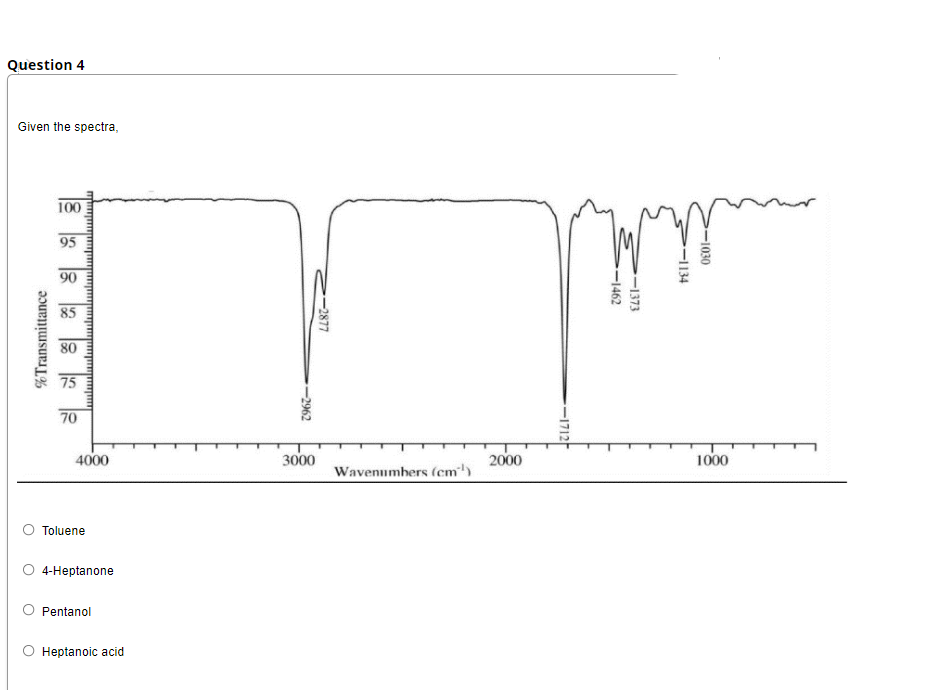
en the Spectra, 100 95 90 85 80 75 70 4000 3000 2000 1000 Wavenumhers (cm') D Toluene D 4-Heptanone D Pentanol O Heptanoic acid -1030 -1134 -1373 -1462 -1712 -2877 -2962 %Transmittance
Q: Consider the following mechanism for the reaction between hydrogen bromide and oxygen: 1.) HBr(g)+O2...
A:
Q: Consider the following reaction where K. = 7.00x10"5 at 673 K. NH4I(s) NH3(g) + HI(g) A reaction mix...
A:
Q: Give the major organic product(s) for each of the following reactions. Write NR if a reaction will n...
A:
Q: Under certain conditions the rate of this reaction is zero order in dinitrogen monoxide with a rate ...
A: Given, 2N2O (g) ➝ 2N2(g) + O2(g) Reaction is zero order Rate constant (k) = 0.0065 M.s-1 Volume of ...
Q: QUESTION 1 Based on the given data, what is the Amax of cobalt? 0.1M Cobalt 300 0.451 325 0.467 350 ...
A: Definition: The wavelength at which a sample has highest absorption.
Q: A certain substance has a heat of vaporization of 34.65 kJ/mol. At what Kelvin temperature will the ...
A:
Q: Under what conditions is the volume of one mole of a gas equal to 22.4 L? What do you call this volu...
A: Under what conditions, volume of one mole of a gas is equal to 22.4 L ? What do you call this volum...
Q: CH2=CH(CH2)4CH3 Give the Expanded Structure and IUPAC Name
A: The IUPAC name of the molecule is hept-1-ene.
Q: What is the Rf formula and is it based off the middle of the spot, top, or bottom for distacne trave...
A: Given, Retention Factor (Rf) Required, Formula of retention factor ...
Q: What is the molar absorptivity of cobalt at λmax of cobalt?
A: The data given is,
Q: Calculate the molarity of a phosphoric acid (H3PO4) solution that is 84% by mass phosphoric acid and...
A:
Q: The Atomic spectra is divided into three groups. of spectral lines which group is depicted by the il...
A:
Q: Which of the following phosphate salts are insoluble in water? A. (NH4)3PO4 B. Li3Po4 C.Na3PO4 D...
A: Given : We have to tell which one is insoluble in water.
Q: Calculate the new concentration of 1500 mL of 2.0 M solution after adding water to become 2225 mL?
A: Given :- Initial concentration of solution (M1) = 2.0 M Initial volume of solution (V1) = 1500 mL ...
Q: statement
A: When a pure substance changes phase from subcooled liquid to superheated steam, this CAN be reversed...
Q: Minor product via E1 for 2-butanol and HCl?
A: Alcohol reacts with hydrogen chloride to give substitution and elimination reactions, depending on t...
Q: The equilibrium constant, K, for the following reaction is 4.68x10-2 at 536 K. PCI5(g) PCI3(g) + Cl2...
A: We have been given equilibrium constant value along with equilibrium concentration at a particular v...
Q: In a typical AAS what is the ouput of the istrument given out as data? % composition O voltage O Abs...
A:
Q: Main group elements have valence electrons in which suborbitals
A: In the periodic table, the elements present in the same group have the same number of valance electr...
Q: Given the following data and the λmax determined in the previous questions, determine: 1) the molar ...
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only. ...
Q: What is the concentration of cobalt in mM?
A: The data given is,
Q: How to calculate grams of iron (III) carbonate in 1.15 mole of Fe2(CO3)3?
A:
Q: Calculate the number of oxygen atoms in 3moles of H2SO4
A: One mole of any compound contains Avoogadro number of particles which is equal to 6.023*1023
Q: Calculate the volume (in mL) of a 1.205M stock NaOH solution needed to prepare 250.0 mL of a 0.1157M...
A:
Q: Why is that the Organic Chemistry separated as branch of Chemistry
A: We know that Organic chemistry is the chemistry of the compounds of carbon, i.e. organic chemistry...
Q: I dentity the tollaming salts as Acidic, Basic or neutiral : HCook Naclos ,kCloy i CoH 5. CH; Nity N...
A: Salt is substance produced via reaction between acid and base. If salt produced via strong acid and...
Q: 2. Salt-in effect implies that solubility: a. Remains constant with increase in ionic strength b. In...
A:
Q: 3. In a material that can undergo a positional ordering phase transition, is an ordering transition ...
A: Positional ordering refers to the extent to which molecules shows transitional symmetry.
Q: what is 0.939g of 4-nitrobenzoyl chloride in mmol?
A:
Q: 6) An analyst meanred the con entration acid in a solution uning two diferent indicators. lis result...
A: Solution -
Q: True or False ____________7] Salivary amylase can be use to hydrolyze 1,4 glycosidic bonds of cel...
A: Given : We have to tell whether statement is true or false.
Q: The Atomic spectra is divided into three groups. of spectral lines which group is depicted by the il...
A: The electronic energy levels in an atom are quantized and when an electron undergoes a transition fr...
Q: A chemistry student is given 1.00 L of a clear aqueous solution at 17 degrees Celsius. He is told an...
A:
Q: Annotate the IR spectrum by labelling each of the major peaks as a functional group. Does this IR sp...
A: IR spectral analysis:
Q: 250. mg of an unknown protein are dissolved in enough solvent to make 5.00 mL of solution. The osmot...
A:
Q: How many calories of heat are required to lower the temperature of 550 grams of water from 64.4℉ to ...
A: Mass of water = 550 grams Initial temperature = 64.4 °F Final temperature = 53.6 °F Specific heat...
Q: Why are there multiple products produced from the halogenation of Alkanes with ultra light
A: Halogenation of alkane: Alkanes can be halogenated in the presence of light or heat. The alkane does...
Q: Draw/Illustrate both of their structural formula of C4H6 and C5H8 (both are Alkynes). Also, fin...
A: DBE = C + 1 - H/2 Where, DBE = Double Bond Equivalent C = no of carbon H = no of hydrogen
Q: C H* CH,CH,CH,OH H,C OH С - ОН H* CH,CH,OH H* CH,CH,CH,OH CH,CH,COOH
A:
Q: Give the four different types of physical states and their symbols.
A: Physical states of matter are interconvertible. one state can be converted to other by exchange of e...
Q: Develop a general equation for the standard heat of reaction as a function of temperature for one of...
A: ANSWER : (a) C2H2(g) + H2O (g)→(CH2)2O (g)Heat of reaction is as follows ;∆Hrxn= ∑products,ini∆Hf29...
Q: It can be shown mathematically that 2 or more smaller extractions (using smaller portions of a solve...
A:
Q: Draw the Lewis dot structure for CH, Cl, . Determine the electron geometry of CH,Cl,:
A:
Q: What is commonly determined using Atomic Spectroscopy? Bodn energy Elemental composition Concentrati...
A:
Q: Compare and contrast GC/MS IR spectrophotometry. TLC spectroscopy, and UV/VIS spectrophotometry Make...
A: Analytical spectroscopy:
Q: CH=C-CH2-CH2-CH3 + H2 Pt, Pd, or Ni A) + HBr в) C)
A: The product of the above organic reaction is given below
Q: 5. For the reaction, 2N2O5 (g) 4 NO2 (g) + 02 (g), the rate of formation of N02 (g) is 4.0 x 10 mol ...
A:
Q: 6. A synthesis of a molecule being explored as an antidepressant drug (J. Org. Chem 2012) utilized t...
A:
Q: c. One of your propagation steps should have created "HBr" as a product. Write the products of the r...
A:
Q: The diagram below represents the layout of a typical single-beam UV-Vis, explain the function of eac...
A:
Step by step
Solved in 2 steps with 2 images

- Expected number of signals in Styrene 13CNMR how many peaks should be seen between a. 100-150ppm b. 150-220ppmWhat is the chemical shift in ppm of a carbon that resonates at 1325 Hz on a 250 MHz instrument (Answer is not 5.3 ppm)?What is the main particularity of a MS signal for a molecule having: An enriched level of 13C atoms (up to 50/50 12C/13C)?
- Isoamyl acetate is the primary component of artificial banana flavor. Which signals will be in the positive phase, negative phase, or nonexistent in normal 13C NMR, DEPT-90, and DEPT-135 of isoamyl acetate?A solution containing two different fluorescent compounds, Ben and Jerry, were analyzed for their individual concentrations in the mixture. Standards of pure Ben and pure Jerry were prepared at a concentration of 500.0 mM and were run in a UV-Vis Spectrophotometer to determine their absorption properties. Absorbance Wavelength Compound Ben 500 mM Compound Jerry 500 mM 400 nm 0.137 0.136 450 nm 0.312 0.113 500 nm 0.154 0.078 550 nm 0.076 0.079 600 nm 0.227 0.148 650 nm 0.230 0.230 700 nm 0.151 0.357 750 nm 0.157 0.246 800 nm 0.154 0.154 A standard curve of the standards was also prepared to help determine the concentration of each component in the solution. The solution produced an absorbance reading of 0.486 at the λmax of Ben, and 0.463 at the λmax of Jerry. STD CURVE BEN Λmax Ben Λmax Jerry STD CURVE JERRY Λmax Ben Λmax Jerry CONC (mM) ABS ABS CONC (mM)…A solution containing two different fluorescent compounds, Ben and Jerry, were analyzed for their individual concentrations in the mixture. Standards of pure Ben and pure Jerry were prepared at a concentration of 500.0 mM and were run in a UV-Vis Spectrophotometer to determine their absorption properties. Absorbance Wavelength Compound Ben 500 mM Compound Jerry 500 mM 400 nm 0.137 0.136 450 nm 0.312 0.113 500 nm 0.154 0.078 550 nm 0.076 0.079 600 nm 0.227 0.148 650 nm 0.230 0.230 700 nm 0.151 0.357 750 nm 0.157 0.246 800 nm 0.154 0.154 A standard curve of the standards was also prepared to help determine the concentration of each component in the solution. The solution produced an absorbance reading of 0.486 at the λmax of Ben, and 0.463 at the λmax of Jerry. STD CURVE BEN Λmax Ben Λmax Jerry STD CURVE JERRY Λmax Ben Λmax Jerry CONC (mM) ABS ABS CONC (mM)…
- A solution containing two different fluorescent compounds, Ben and Jerry, were analyzed for their individual concentrations in the mixture. Standards of pure Ben and pure Jerry were prepared at a concentration of 500.0 mM and were run in a UV-Vis Spectrophotometer to determine their absorption properties. Absorbance Wavelength Compound Ben 500 mM Compound Jerry 500 mM 400 nm 0.137 0.136 450 nm 0.312 0.113 500 nm 0.154 0.078 550 nm 0.076 0.079 600 nm 0.227 0.148 650 nm 0.230 0.230 700 nm 0.151 0.357 750 nm 0.157 0.246 800 nm 0.154 0.154 A standard curve of the standards was also prepared to help determine the concentration of each component in the solution. The solution produced an absorbance reading of 0.486 at the λmax of Ben, and 0.463 at the λmax of Jerry. STD CURVE BEN Λmax Ben Λmax Jerry STD CURVE JERRY Λmax Ben Λmax Jerry CONC (mM) ABS ABS CONC (mM)…A carbon signal measured in a 300MHz spectrometer with an observing frequency of 75.47MHz isfound to have a chemical shift of 187.23 ppm. What is the resonance frequency of the carbon withthis chemical shift?A) 56169 HzB) 75 484 130 HzC) 14 130 HzD) 300 056 139 HzA4 The chemical shift of signed proton H at 500MHz was 5ppm. Show the coupling pattern using graphical analysis. And what Hz is the frequency? When measured in 300MHZ, what is the change in frequency and couping constant? Explain.
- In determining the concentration of a sample X, it was found that it has maximum absorbance at 275 nm. Compound X, has molar absorptivity at 275 nm of 8400 L / mol.cm. Using a spectrophotometer with a 1 cm cuvette, an absorbance of 0.70 was found. What is the concentration of compound X in the sample?You are a forensic chemist working in a crime lab. You have received a white powder from investigators that is suspected to be a drug of abuse. Based on the FTIR spectrum, make a decision and justify it based on peak assignments above 1500 cm-1 . Which of the three is it?heres the data TZ # Concentration Absorbance at 430 nmnm 1 33.6480 0.931 2 25.2360 0.757 3 16.8420 0.210 4 8.41200 0.137 5 4.20600 0.122

