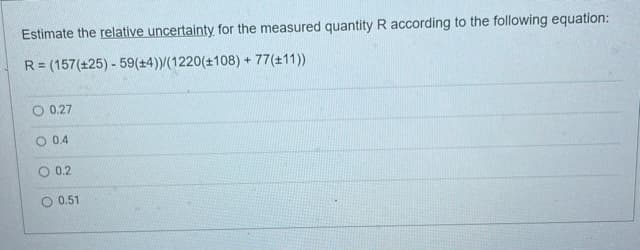
Estimate the relative uncertainty for the measured quantity R according to the following equation: R = (157(±25) - 59(+4))/(1220(±108) + 77(+11)) O 0.27 O 0.4 O 0.2 0.51
Q: 4. How would you prepare the following ethers starting with any alkyl halide and other reagents? a.…
A: Alkyl halide on reaction with alkoxides give ethers.
Q: Which of the following statements are false with respect to rate laws? Select all that apply. □…
A: Rate law is a mathematical expression that relates the rate of a chemical reaction to the…
Q: Suppose a 250. mL flask is filled with 0.60 mol of SO₂ and 1.0 mol of SO3. This reaction becomes…
A: Since, Molarity is the number of moles of solute dissolve in one litre of the solution. Thus,
Q: ORGANIC FUNCTIONAL GROUPS Understanding the names of carboxylate salts What is the systematic name…
A: We know according to Baldwin rule 5 membered ring formed faster than any other higher membered ring…
Q: Provide a complete curved arrow mechanism that accounts for the transformation below. Be sure to…
A: Answer :
Q: Question 29 of 42 Which of the following statements are false with respect to rate laws? Select all…
A: Rate law is a mathematical expression that relates the rate of a chemical reaction to the…
Q: X app.101edu.co Aktiv Chemistry X + Que What mass of precipitate (in g) is formed when 45.5
A: When stoichiometric coefficient results in the equal number of atoms of each element on both the…
Q: A saturated solution of barium fluoride, BaF₂, was prepared by dissolving solid BaF₂ in water. The…
A: Given, A. The concentration of Ba2+ in BaF2 solution = 7.52 x 10-3 M. The solubility product…
Q: I have calculated this problem various times, but every time I input the answer into the assignment…
A: Given that, the standard electrode potential (Eocell) of a cell can be calculated by: Ecell0 =…
Q: A buffer is prepared by mixing 43.2 mL of 0.196 MNaOH with 138.1 mL of 0.231 Macetic acid. What is…
A: We have to calculate the pH of solution
Q: The frequency of a photon of electromagnetic radiation with an energy of 1.2 x 10-18 Jis O 8.9 ×…
A:
Q: 6:15 Question 16 of 22 2- 3C₂² Write the basic equilibrium equation for NH3. Be sure to include the…
A: Equilibrium refers to a state where the rates of the forward and reverse reactions are equal,…
Q: A solution contains Al³+ and Co²+. The addition of 0.3923 L of 1.653 M NaOH results in the complete…
A: Number of moles is calculated by dividing given mass with molecular mass. Given that - Molarity of…
Q: 1. Write the overall reaction for an E1 reaction or the dehydration of an alcohol.
A: The dehydration of alcohol reaction follows E1- pathway where a stable carbocation is formed which…
Q: The following compound is CH3 H3CTH Br O meso O achiral but not meso O chiral Hint Is there a plane…
A: Given that, the given molecule is 2-Bromopropane. We have to tell whether the molecule is chiral or…
Q: Visited B-propiolactone Poly(beta-propiolactone) can be made by anionic polymerization (see above)…
A:
Q: A chemist titrates 80.0 mL of a 0.8669M ethylamine (C₂H₂NH₂) solution with 0.3629M HNO3 solution at…
A:
Q: 8.18) What is the base form of H30+? На о H30+ H₂O + H₂O base acid H₂O + H+
A:
Q: Write the basic equilibrium equation for HS. Be sure to include the proper phases for all species…
A: Acids when dissolve in water give H+ ions and the equation is called acidic equilibrium equation.…
Q: < Construct the expression for Kb for the weak base, CIO™. CIO (aq) + H₂O(1) OH-(aq) + HCIO (aq) Kb…
A:
Q: 4. According to Saytzeff's rule, which of the following isomers is formed during the dehydration of…
A: Answer: Given chemical reaction is the dehydration reaction of an alcohol to form alkene and this…
Q: Determine the molecular formula and molecular mass of the compound represented by the chemical…
A: Mass of a compound can be calculated from its molecular formula.
Q: Using the balanced chemical equation, determine how many moles of NaCl will be produced, if 0.907…
A: A question based on mole concept. A reaction involving formation of NaCl is given using that the…
Q: 8.17) What is the acid form of NH3? + Nhật
A: Since, From the definition, Brønsted-Lowry acid are proton donar and Brønsted-Lowry base are proton…
Q: 2880 J 3. How much heat is required to raise the temperature of 54.5g of PC13 from 18.6°C to Cp PCl3…
A: Since you have asked for question 3 only, we are providing you a solution for the same. If you need…
Q: 8.25) For each of the following, write whether the solution condition describes an acidic, basic, or…
A: pH : pH can be defined as the negative logarithm of hydrogen ion concentration pH = -log[H+] when…
Q: 2. Draw the Newman projection of the following compounds about the indicated bonds: н CH3 H F₁,, Н…
A: Place groups that are present on the wedge at right hand side and the groups that are present on the…
Q: calculate the equivalence point volume in the containter of 2.00 g Ba(OH)2 with 0.100 M HCl. molar…
A: We have to calculate the equivalence point volume
Q: How many mL of 1.25 M HCI would react fully with 15.25 mL of 0.836 M Ca(OH)2? 2 HCI + Ca(OH)2 →…
A:
Q: Question 3: Heat Capacity and Latent Heats a. Revolutionary War soldiers made their own musket balls…
A: Given :- Mass of Lead, Pb= m = 21 g Initial temperature of Pb = Ti = 25 °C Final temperature of…
Q: Indicate the majority product of each of the following reaction sequences. 1) HNO3 H₂SO4 2) Br₂…
A: We know nitro group is meta directing so forming para bromo nitro benzene followed by reduction…
Q: Another mixture of AsF3(g), AsF3(g), and F₂(g) is at equilibrium at a different temperature in a…
A: The reaction between AsF3 and F2 can be given as: AsF3(g)+F2(g)→AsF5(g) According to the Le…
Q: H Ph 1) NEt3, DMF, 40°C 2) mild H3O* workup e.s Ph OH minor product OH Ph major product
A:
Q: The reaction rate constant is determined to be 0.012 M-1 s-1. If after 27 minutes the amount of A…
A:
Q: 2.) Provide a detailed arrow pushing mechanism for this transformation. Clearly show all…
A:
Q: For each of the equations, determine the change in the number of moles of gas, Angas. (NH4)₂CO3(s)…
A: Given that, Equations- 1.(NH4)2CO3(s)---->2NH3(g)+CO2(g)+H2O(g) 2.H2(g)+Cl2(g)------>2HCl(g)…
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into…
A: Given : energy released by the reaction, ∆E = -274 KJ where negative sign indicates energy is…
Q: what is the structure of the major product with protons labeled, along with a table that lists those…
A: Given reaction is the Wittig reaction in which the aldehyde is converted into olefins. The…
Q: Which of the following errors are determinate errors? Personal errors. O Method errors. O Random…
A: Determinate errors are those errors that can be identified and corrected. They include personal…
Q: Br SMe/DMSO
A:
Q: Explain the processes of dissolution of Nacl
A: When NaCl (sodium chloride) is added to water (H2O), it dissolves and forms a solution. This process…
Q: In lab, you will be provided with a 1.8 M acetic acid solution (CH3COOH) and a 1.0 M NaOH solution.…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: A) B) 1) 9- 2) H2O2, NaOH فه C) D) you
A:
Q: 1.) The fragrance industry uses volatile molecules to add scent to perfumes. A strategy shown below…
A: (a) Imines are formed by acid catalyzed reaction between aldehyde and amines. Imines having C=N-…
Q: 3 Zn(s)3 Zn(�) +2 Cr3+(aq)+2 Cr3+(��) →3 Zn2+(aq)→3 Zn2+(��) +2 Cr(s) If the [Zn2+][Zn2+] is 4…
A: Answer: Galvanic cell is a type of cell that converts chemical energy into electrical energy via…
Q: CdF2(s)⇄Cd2+(aq)+2F−(aq) A saturated aqueous solution of CdF2 is prepared. The equilibrium in the…
A:
Q: 2 Al(s)+3CuCl,(aq)→3Cu(s)+2 AlCl, (aq) A piece of aluminum metal is placed in an aqueous solution of…
A:
Q: Why vigorous stirring is required during the oxidation of borneol to camphor, during this week's…
A: Vigorous stirring is required during the oxidation of borneol to camphor to ensure efficient mixing…
Q: At a certain temperature, the equilibrium constant K for the following reaction is 311.: H₂(g) +…
A: Please find your solution below : The given balanced reaction is : H2 (g) + Cl2 (g) → 2 HCl (g)…
Q: Solid Ca₃(PO₄)₂ is placed into 10.0 L of water. When equilibrium is established, the concentration…
A: Answer :
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- To test the quality of the work of a commercial laboratory, duplicate analyses of apurified benzoic acid (68.8% C, 4.953% H) sample were requested. It is assumed that therelative standard deviation of the method is sr → = 4 ppt for carbon and 6 ppt forhydrogen. The means of the reported results are 68.5% C and 4.882% H. At the 95%confidence level, is there any indication of systematic error in either analysis?Estimate the absolute deviation(or uncertainty) for the results of the following calculations. Round each result so that it contains only significant digits. The numbers in parentheses are absolute standard deviations. Finally, write the answer and its uncertainty.a.) Y= 6.75 (± 0.03) + 0.843(±0.001) - 5.021 (±0.001) = 2.572b.) Y= 19.97(± 0.04) + 0.0030(±0.0001) + 4.29(±0.08) = 24.263c.) Y= 143(± 6) - 64(±3) = 5.9578 x10-21249 (±1) +77 (±8)An analysis of city drinking water for total hardness was done by two students in the laboratory and produced the following results (in ppm CaCO3): Student A: 228.3, 226.4, 226.9, 227.1, and 228.6. Student B: 229.5, 226.1, 230.7, 223.8, and 227.5What is the 95% confidence interval for the mean?
- A volumetric calcium analysis on samples of the blood serum of a patient believed to be suffering from a hyperparathyroid condition produced the following data: mmol Ca/L = 3.10, 3.08, 3.28, 3.15, 3.26, 3.12, 3.14, 3.18, 3.25, 3.11, 2.95. Calculate: Mean Standard deviation Coefficient of variation What is the 95% confidence interval for the mean of the data, assuming no prior information about the precision of the analysis? Apply the Q test to the following data sets to determine whether the outlying result should be retained or rejected at the 95% confidence level. ( work on the first measurements according to your case)The following data were obtained in order to determine the density of a cylindrical metal bar. If the accepted value for the density of the bar is 6.70 g/cm^3, what is the percent relative error?What is the relative error in the ppt of the following data if the accepted value is 70.05 M? 70.24 M, 70.22 M and 70.10 M
- Chemistry The levels of an organic pollutant (P) in the groundwater at the perimeter of a plant were a cause for concern. A 10 mL sample of the water was taken and the pollutant was extracted with 95% efficiency using 25 mL of diethyl ether. GC was used to analyse the concentration of P in diethyl ether. A calibration curve was plotted for a series of standards of P which yielded the following results: Peak Area Toluene Conc. (µg/ml) 12,000 2.6 23,700 5.0 35,500 7.7 46,800 9.9 31,250 Sample Determine the concentration of P in ppb in the initial groundwater sample.A powder was prepared containing 3.00% NaCN and 97.00% NaCl. A sample obtained from that mixture containing 7.374×10^5 particles weighs 10.0 g. Determine the number and percent relative standard deviation of NaCN particles from a sample of the mixture weighing 6.30 g. I need nNaCN particles and %RSDTypes of Error; Propagation of Uncertainty from Random ErrorYou prepare an NH3 [FM 17.030 5 (±0.000 2)] solution by diluting 7.36 (±0.03) mL of a 28.00 (±0.05) wt% NH3 solution [density = 0.899 (±0.003) g/mL] to a final volume of 500.00 (±0.02) mL. Find the molarity of the NH3 solution along with its absolute uncertainty.
- What is the median of the values 7.79, 8.24, 5.81, 4.97, 1.04, 7.39, and 7.22?1) Report the results of this calculation along with both the percent relative and absolute uncertainties. 417.3 ± 0.4 × 71.8 ± 0.7(647.21 ± 0.2 − 182.3 ± 0.4) Absolute uncertainty:Percent uncertainty:Estimate the absolute standard deviation (or uncertainty) for the results of the following calculations. Round each result so that it contains only significant digits. The numbers in parentheses are absolute standard deviations. Show your solution and express the final answer and its corresponding uncertainty. a) y= 2.998(±0.002) - 3.98 (±0.15) + 9.035 (±0.002) = 8.0531 b) y= 39.2(±0.3) x 3.054 (±0.022) x 10^ -2 = 1.197 c) y= [198(±6) - 89 (±3)] / 1335 (±2) + 64 (±7)] = 7.791 x 10^-2



