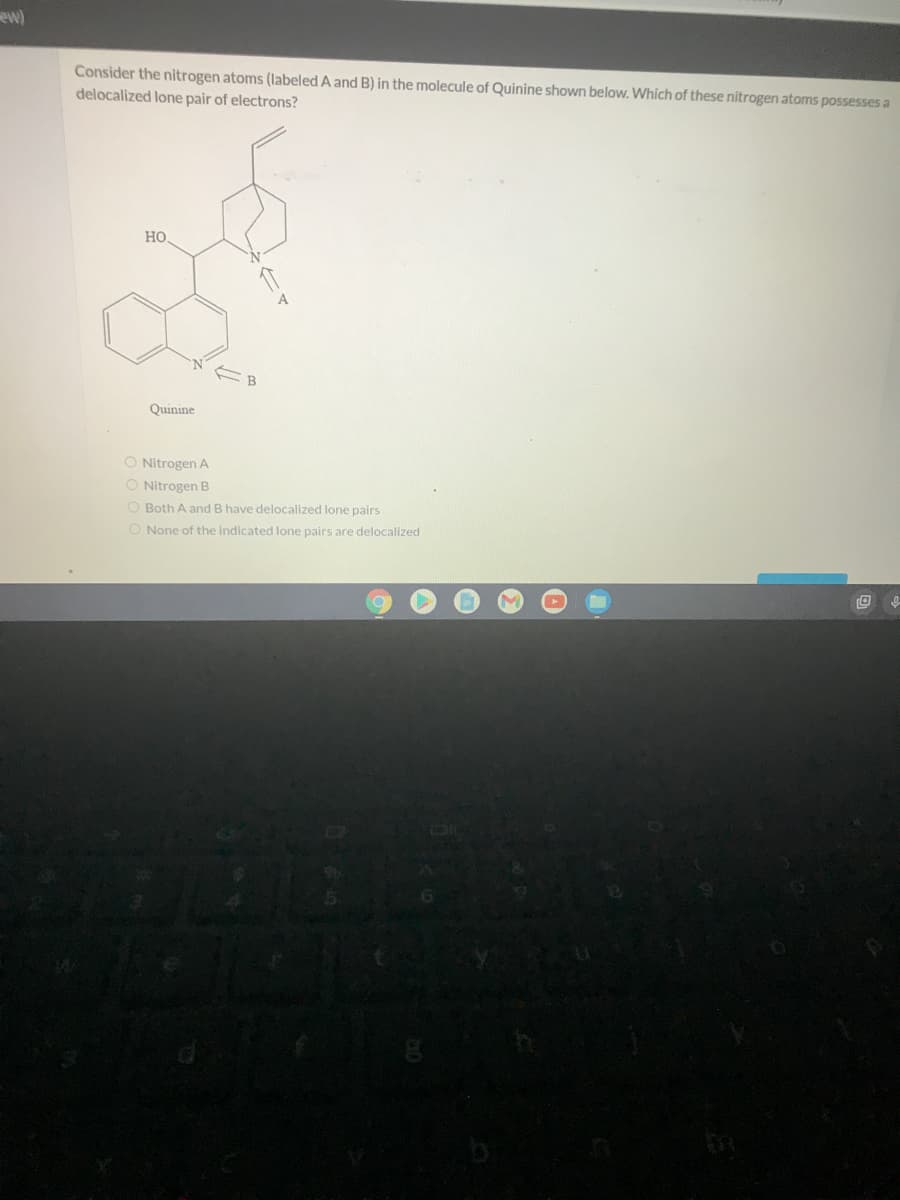
ew) Consider the nitrogen atoms (labeled A and B) in the molecule of Quinine shown below. Which of these nitrogen atoms possesses a delocalized lone pair of electrons? HO. 4 Quinine O Nitrogen A Nitrogen B Both A and B have delocalized lone pairs O None of the indicated lone pairs are delocalized OR
ew) Consider the nitrogen atoms (labeled A and B) in the molecule of Quinine shown below. Which of these nitrogen atoms possesses a delocalized lone pair of electrons? HO. 4 Quinine O Nitrogen A Nitrogen B Both A and B have delocalized lone pairs O None of the indicated lone pairs are delocalized OR
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter7: Cycloalkanes
Section: Chapter Questions
Problem 13E
Related questions
Question
Transcribed Image Text:ew)
Consider the nitrogen atoms (labeled A and B) in the molecule of Quinine shown below. Which of these nitrogen atoms possesses a
delocalized lone pair of electrons?
Но
Quinine
Nitrogen A
O Nitrogen B
O Both A and B have delocalized lone pairs
O None of the indicated lone pairs are delocalized
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning



Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

