Q: Draw the energy levels of the compounds (acetald ehyde methyl utny lethen) and mark HOMO, LUMO.
A: Acetaldehyde can be represented as:
Q: is a generalized structural representation which can be used for all of the following, except: A)…
A:
Q: Consider 2-methylpropane (isobutane). Sighting along the C2-C1 bond: (a) Draw a Newman projection of…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: Directions: Name the following hydrocarbo 1. CH3 CH3 H3C-CH-CH-CH2-CH3 CH3 CH3 2. CHз — с- CH2 CH…
A: Rules for IUPAC naming :- Select the longest chain which contains carbon-carbon single bonds.…
Q: Jse names from the table below. List each class of functional group only once. If there are fewer…
A: Functional group is a substituent because of which chemical reaction takes place.
Q: 1. Circle all of the molecules below that have 2 or more double bonds in resonance.
A: Resonance explains the fact that electrons are delocalized. The delocalisation of electrons…
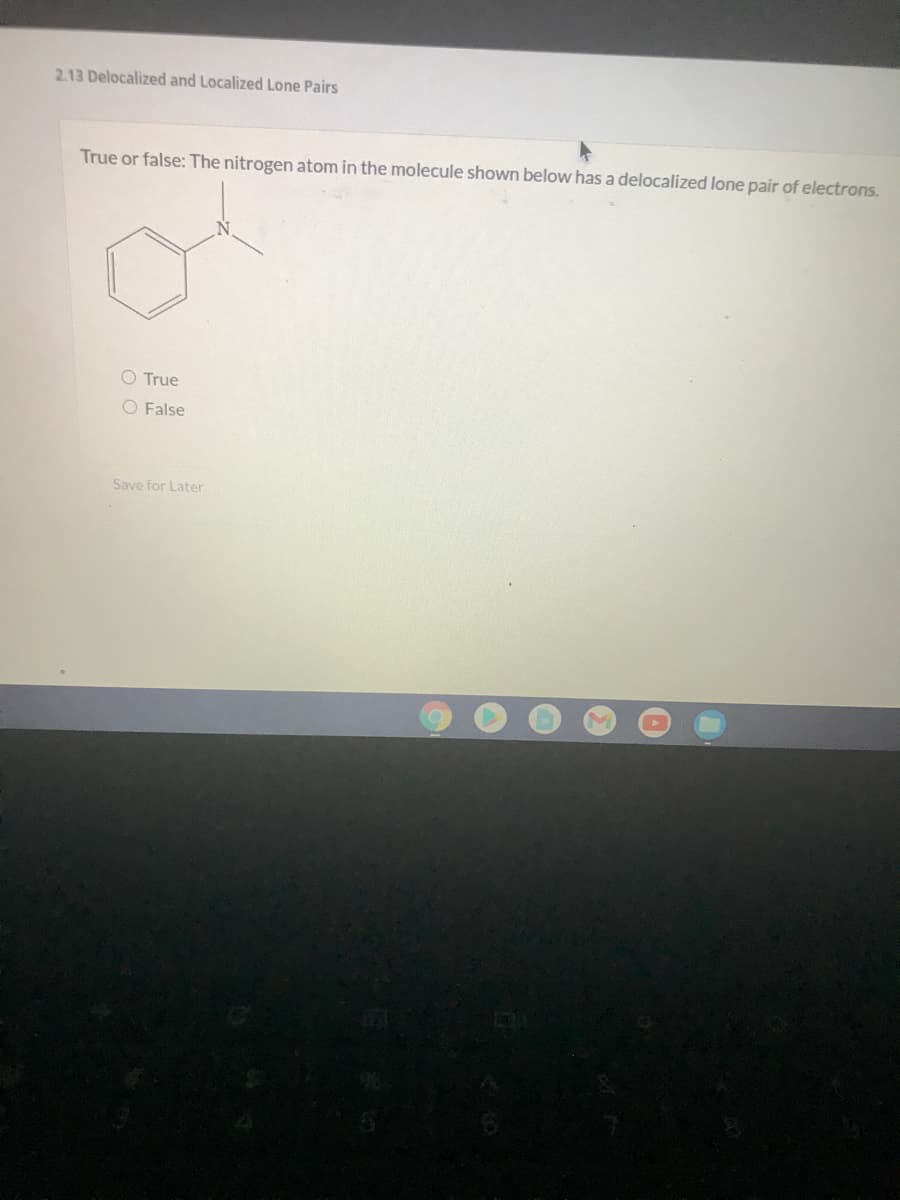
Q: If the compound has delocalized electrons, drag the word "YES" before the number. Otherwise drag the…
A: Delocalization of electron density beyond a fixed point, such as a single atom, lone pair, or…
Q: Draw the Fisher Projection of following compounds. (I) (2R,35)-2,3-dibromohen9ne (II)2R,…
A: Fischer projection is just a two dimensional representation of a three dimensional molecule. In…
Q: the wedges are supposed to be up, but why the both chair comformations point down? CH3 CH3 H3C CH3…
A: Two types of bonds exist in cyclohexane chair conformer. Axial and equatorial. When only a single…
Q: Question attached
A: Introduction: Molecular structure shows the arrangement of atoms in a space. The organic structures…
Q: How many degrees of unsaturation does this molecule have?
A: The degree of unsaturation is known as Double bond equivalent. It can be calculated by the…
Q: Can someone help me draw the structural formula of this SS configuration?
A: wedge-dash structure is drawn below having SS configuration
Q: nitrobenzene You do not have to consider stereochemistry. • Include all valence lone pairs in your…
A: Nitro group being electron withdrawing group, pull electrons from benzene ring and form resonating…
Q: How many π electrons are delocalized in the ring?
A: The given comound C has 16pi electrons from the 8pi electron due to teh double bonds
Q: Question: How many C-types do each of the following compounds have? Label them a, b,c ... And…
A: If two carbon have different environment, it means they are connected to each other and also they…
Q: Anti-Markovnokov’s rule is followed in the presence of? a. Ni b. CCl4 c. H2O2
A:
Q: Classify the following compounds as E/Z- structures CH3 HO-CH2-CH2 CH2CHCH2OH H2N CH2-CH3 H2N CH=CH2…
A: For abC=Ca'b' system E/Z nomenclature occurs and E/Z nomenclature is based on CIP rule which states…
Q: Which of the following Newman projections correctly represents the structure of (2R,35)-2-chloro-3-…
A: Newman projection visualizes the conformation of a chemical bond from front to back, with the back…
Q: 1. How many unsaturation elements each of the following molecules have? CI, HO C3H12
A: Degree of unsaturation is a measure of total number of pi bonds and rings in an organic molecule. It…
Q: In which Newman projection below the chlorine atom is gauche to a hydroxyl group? ОН Н. I I Н CI Н Н…
A: The isomers that are converted into each other by rotation around a single bond are known as…
Q: CH3 H CH3
A: Newman projection of (2R,3S)-2,3-butanediol
Q: a. Convert the following Newman projection of compound G to a three dimensional line structure in…
A:
Q: 2D Representations Conformation Newman Sawhorse projection projection 39. 40. preferred (most…
A:
Q: CH3 CH CH CH3 `CH3 CH3 CH-CH-CH2-CH3-CH-CH3 CH3 CH2 ÇH2 TH CH2 CH2 CH CI
A: Cycloalkanes are organic compounds containing carbon and hydrogen atoms. The carbon and hydrogen…
Q: Convert into a line-angle formula CH3(CH2)3CH(CH₂CH3)CH₂CH3
A:
Q: I Which structure is the ring-flipped (chair-flipped) version of this molecule? A. B. E. Ö لسلام D.
A: On chair flipping, the atom or group on equatorial position will come to the axial position and the…
Q: (a)(2R,3S)-2,3-dibromohexane, draw a three-dimensional representation.
A: Stereochemistry is the study of how molecules are affected by the way their atoms are arranged in…
Q: 4) The pair of electrons labeled “A” (at the end of the arrow) are localized 5) The pair of…
A: Firstly hear it needs to know the concepts about localized and delocalized electrons. Localised…
Q: A Newman projection and wedge-and-dash picture of a molecule are shown. The front carbon in the…
A: The Newman's projection and the wedge-dash picture of a molecule are interconconverible.
Q: H H CH3 H;C-Ċ-Ċ-Ċ-CH3 ČH3
A:
Q: S H-O O I'l" S H
A:
Q: Write each of the following condensed structural formulas as a bond-line formula (that is, using…
A: The bond-line formula of the organic compounds is the way of representing the large organic…
Q: Write the IUPAC name of the following molecule: "Lowercase letters only and D NOT put space in…
A: The given compound is a cyclohexane containing one ethyl group and two methyl groups.
Q: 3 Molecular Structures (Sawhorse basic) 3.1 Do not worry about conformation. I just want to see you…
A:
Q: Which molecule would have the greatest delocalization energy? O Phosphoric Acid (H3PO4) Carbonate…
A:
Q: a. Convert the following Newman projection of compound G to a three dimensional line structure in…
A:
Q: Draw the simplest possible set of curved arrows that shows how the structure on the left could be…
A:
Q: Convert the following Newman projections to skeletal structures and name them.
A: The conversion of Newman projection (a) into skeletal structure and its IUPAC name are shown below.
Q: Present or Absent? a. CH hyperconjugation b. Lone-pair delocalization
A:
Q: What does the most stable Newman Projection of 2,2 dimethyl hexane look like?
A:
Q: Which of the following Newman projections correctly represents the structure of (2R,3S)-2-chloro-…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: CH3 H3C- H2 Нас — с—снз H3C-c-CH Нас —с —сн i. ii. CH3 iii.
A:
Q: In C6H6OH, the bond that undergoes heterolytical change most readily is * С-С С-О С-Н O-H None of…
A:
Q: Predict whether each of these 3 substituents owill be axial or equatorial: t-butyl "CH,CH3 CH3
A: Bulkiest group is kept equitorially and with respect to that orientations of other groups are…
Q: How do you create a Newman projection that looks down both the c3-c4 and the c1-c6 bonds at the same…
A: Newman projections: The new conformations of compounds can be drawn and analyzed by Newman…
Q: Starting from the Newman projection below, rotate the back carbon to provide the structure in a…
A: Rotation of Back carbon will produce gauche and eclipsed conformation.
Q: The following molecule has how many degrees of unsaturation? 6. 4 3
A:
Q: nsed structural formula shown in the box? „CHCO(CH,),CH, A)
A: As the condensed structure has ketone functional group. So, (d) is correct bond line notation.
Q: On the propane skeleton below, drawn in the necessary substituents using wedge and dash notation to…
A: R and S configuration are stereochemistry of chiral center which can be assigned on the basis of CIP…
Q: Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Draw the structures of all…
A: We know that, Constitutional Isomer :- Constitutional Isomer also called as structural isomer where…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Write line-angle formulas for the five constitutional isomers with the molecular formula C6H14.Question: A Newman Projection of a tetrahydropyran is drawn below. Draw a bond line-angle representation of the substituted tetrahydropyran in the following figure. Be sure to show realtive positions in the bond-line structure by using either dashes or wedges.Make a continuous model for C4H10 by using 4 black 4 hole carbon atoms, 10 white one hole hydrogen atoms, and 13 pink bonds. Then write in wedge dash notation C4H10 and add in missing hydrogen atoms.
- Draw a complete structure for a molecule with the molecular formula N2H2 and CH2Cl2. Include all Valence lone pairs in your answer.Select all the partially positive carbon atoms in the following set of molecules. If there is more than one partially positive carbon in a molecule, then choose the most positive carbon. If there is not a partially positive atom, then do not select a carbon in that molecule.1. Add nonbonding electron pairs to each atom where it is implied by the line-angle drawings shown below. 2. Use the curved arrow formalism to generate four additional, valid resonance structures for each compound. 3. Rank your resonance structures in order of increasing contribution to the overall nature of the molecule.
- Convert the following 6 molecular formulae into line structures. Be mindful of bond angles and properly align/distribute all molecules appearing in the same row.a. list the polar bonds in acetone with their corresponding dipole vectors. Is acetone a polar molecule? b. What is valence bond hybridization of each carbon atom in acetone? c. What functional group is present in acetone?Draw a line angle formula of a compound that has a ring of 6 carbons and the composition C7H14
- Choose the letter of the correct answer based on the given pictures 1 and 2 1. The molecular structure of thymol, a fungicide from oil of thyme, is shown below. Based on this information, which region is the expected direction of the dipole moment? (Refer to the orientation of the molecule) a. Towards the aromatic ring b. Away from the aromatic ring c. Away from the oxygen atom d. Towards the oxygen atom 2. Based on the illustration, what is the classification of the given structure? a. Alcohol b. Ether c. Aldehyde d. Ketone e. Carboxylic AcidWrite a molecular formula corresponding to the systematic n propane. Then provide a new main projection that illustrates the highest energy confirmation of n propane How long any C-C bond axis of the same.Draw a structure of oxindole, with an aromatic ring. Make sure the diagram contains sp3 carbon atom, sp2 carbon atoms, and sp hybridised carbon atoms. The structure should also include a sp2 hybridised heteroatom and a hybridised sp3.

