Exactly 175 mL of a 0.32 M solution of Na₂SO3 were treated with 250 mL of 0.40 M HCIO4 and boiled to remove SO₂ formed. Na2SO3 + HCIO4 → NACIO4 + H₂O+ SO₂ (g) (reaction is not balanced !) What was the the unreacted reagent ( Na2SO3 or HCIO4) after the reaction was complete ? concentration of Select one: O a. 0.0343 M O b. 0.0141 M OC. 0.118 M O d. 0.104 M O e. 0.176 M
Exactly 175 mL of a 0.32 M solution of Na₂SO3 were treated with 250 mL of 0.40 M HCIO4 and boiled to remove SO₂ formed. Na2SO3 + HCIO4 → NACIO4 + H₂O+ SO₂ (g) (reaction is not balanced !) What was the the unreacted reagent ( Na2SO3 or HCIO4) after the reaction was complete ? concentration of Select one: O a. 0.0343 M O b. 0.0141 M OC. 0.118 M O d. 0.104 M O e. 0.176 M
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter21: The Chemistry Of The Main Group Elements
Section: Chapter Questions
Problem 120IL
Related questions
Question
Subject: chemistry
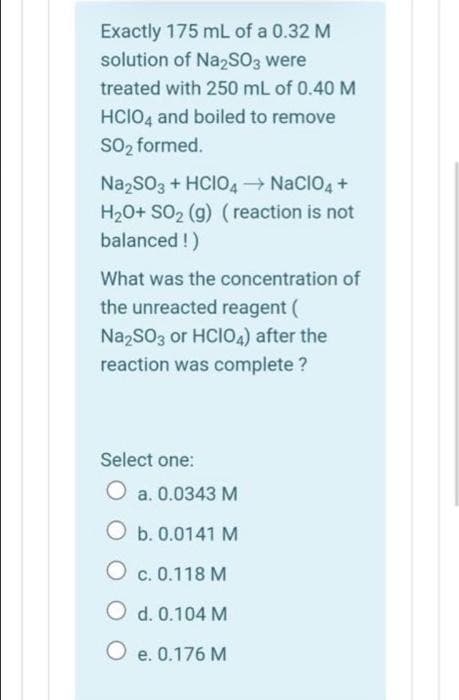
Transcribed Image Text:Exactly 175 mL of a 0.32 M
solution of Na2SO3 were
treated with 250 mL of 0.40 M
HCIO4 and boiled to remove
SO₂ formed.
Na₂SO3 + HCIO4 → NaCIO4 +
H₂O+ SO₂ (g) (reaction is not
balanced !)
What was the concentration of
the unreacted reagent (
Na2SO3 or HCIO4) after the
reaction was complete ?
Select one:
O a. 0.0343 M
O b. 0.0141 M
OC. 0.118 M
O d. 0.104 M
O e. 0.176 M
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 21 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning