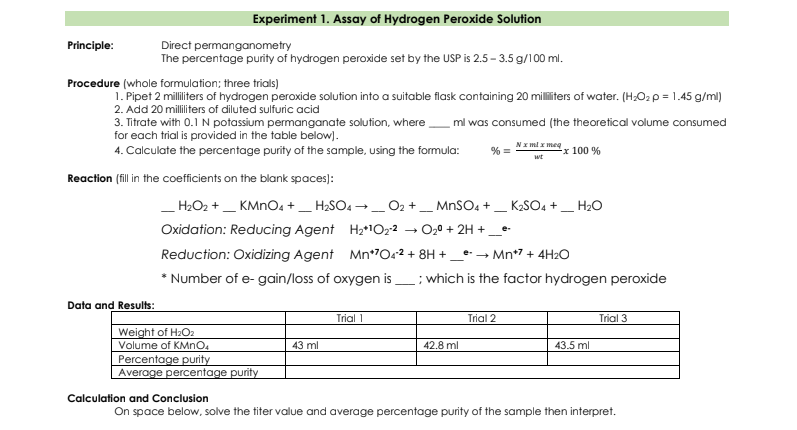
Principle: Direct permanganometry The percentage purity of hydrogen peroxide set by the USP is 2.5 – 3.5 g/100 ml. Procedure (whole formulation; three trials) 1. Pipet 2 milliters of hydrogen peroxide solution into a suitable flask containing 20 milliters of water. (H;O2 p = 1.45 g/ml) 2. Add 20 milliters of diluted sulfuric acid 3. Titrate with 0.1 N potassium permanganate solution, wheremi was consumed (the theoretical volume consumed for each trial is provided in the table below). 4. Calculate the percentage purity of the sample, using the formula: NI mi x meg 100 % % = Reaction (fil in the coefficients on the blank spaces): _H2O2 +_ KMNO4 + _ H;SO4 →_ O2 + _ MNSO, + _ K2SO4 + _ H20 Oxidation: Reducing Agent H2*1022 → 0,0 + 2H +_• Reduction: Oxidizing Agent Mn*70r² + 8H + _* → Mn*7 + 4H2O * Number of e- gain/loss of oxygen is, ; which is the factor hydrogen peroxide Data and Results: Trial 1 Trial 2 Trial 3 Weight of H:Oa Volume of KMNO, Percentage purity Average percentage purity 43 ml 42.8 ml 43.5 ml Calculation and Conclusion
Principle: Direct permanganometry The percentage purity of hydrogen peroxide set by the USP is 2.5 – 3.5 g/100 ml. Procedure (whole formulation; three trials) 1. Pipet 2 milliters of hydrogen peroxide solution into a suitable flask containing 20 milliters of water. (H;O2 p = 1.45 g/ml) 2. Add 20 milliters of diluted sulfuric acid 3. Titrate with 0.1 N potassium permanganate solution, wheremi was consumed (the theoretical volume consumed for each trial is provided in the table below). 4. Calculate the percentage purity of the sample, using the formula: NI mi x meg 100 % % = Reaction (fil in the coefficients on the blank spaces): _H2O2 +_ KMNO4 + _ H;SO4 →_ O2 + _ MNSO, + _ K2SO4 + _ H20 Oxidation: Reducing Agent H2*1022 → 0,0 + 2H +_• Reduction: Oxidizing Agent Mn*70r² + 8H + _* → Mn*7 + 4H2O * Number of e- gain/loss of oxygen is, ; which is the factor hydrogen peroxide Data and Results: Trial 1 Trial 2 Trial 3 Weight of H:Oa Volume of KMNO, Percentage purity Average percentage purity 43 ml 42.8 ml 43.5 ml Calculation and Conclusion
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.19QAP
Related questions
Question
This is a practice problem.
Transcribed Image Text:Experiment 1. Assay of Hydrogen Peroxide Solution
Principle:
Direct permanganometry
The percentage purity of hydrogen peroxide set by the USP is 2.5 – 3.5 g/100 ml.
Procedure (whole formulation; three trials)
1. Pipet 2 milliters of hydrogen peroxide solution into a suitable flask containing 20 militers of water. (H;O2 p = 1.45 g/ml)
2. Add 20 milliliters of diluted sulfuric acid
3. Titrate with 0.1 N potassium permanganate solution, where
for each trial is provided in the table below).
ml was consumed (the theoretical volume consumed
NI ml x meg
4. Calculate the percentage purity of the sample, using the formula:
% =
x 100 %
wt
Reaction (fill in the coefficients on the blank spaces):
_H2O2 +_ KMNO4 +_ H2SO4 → _O2 +_ MNSO4 + _ K2SO4 + _ H2o
Oxidation: Reducing Agent H2*1022
O20 + 2H +_e-
Reduction: Oxidizing Agent Mn*O2 + 8H +.
e- Mn*7 + 4H2O
Number of e- gain/loss of oxygen is
; which is the factor hydrogen peroxide
-
Data and Results:
Trial 1
Trial 2
Trial 3
Weight of H2O2
Volume of KMNO.
Percentage purity
Average percentage purity
43 ml
42.8 ml
43.5 ml
Calculation and Conclusion
On space below, solve the titer value and average percentage purity of the sample then interpret.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
