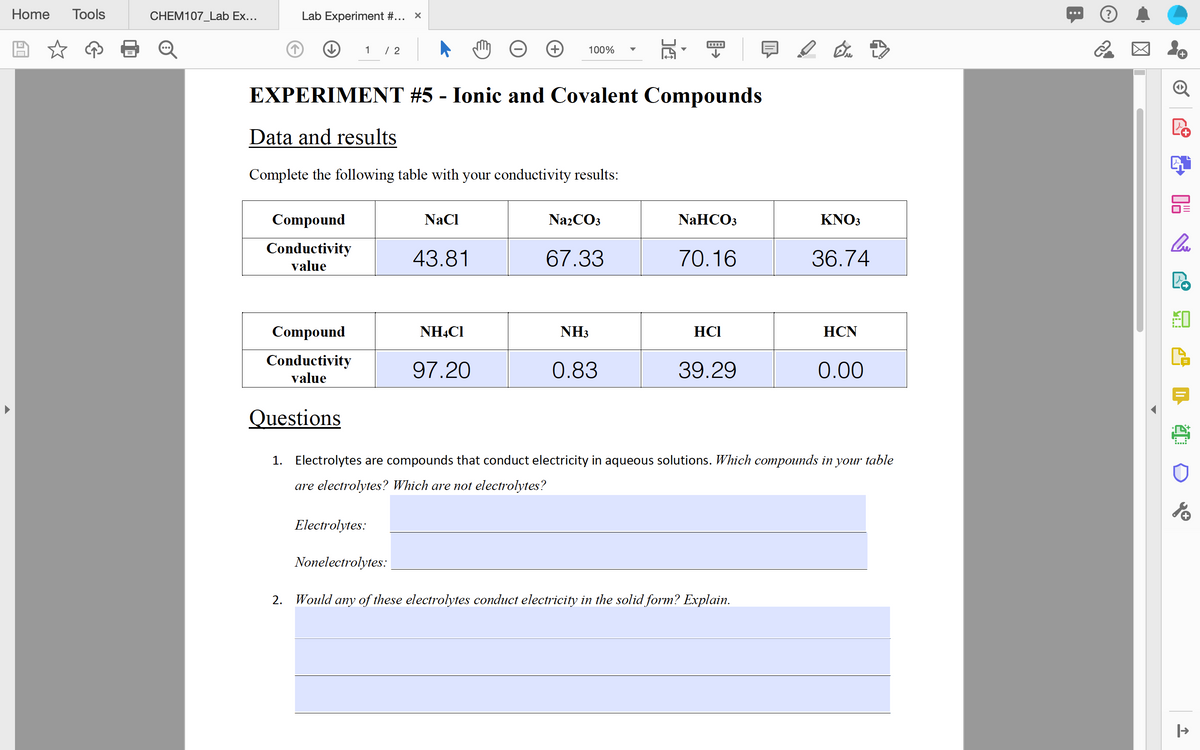
EXPERIMENT #5 - Ionic and Covalent Compounds Data and results Complete the following table with your conductivity results: Compound NaCI NazCO3 NaHCO3 KNO3 Conductivity value 43.81 67.33 70.16 36.74 Compound NHẠCI NH3 HCI HCN Conductivity value 97.20 0.83 39.29 0.00 Questions 1. Electrolytes are compounds that conduct electricity in aqueous solutions. Which compounds in your table are electrolytes? Which are not electrolytes? Electrolytes: Nonelectrolytes: 2. Would any of these electrolytes conduct electricity in the solid form? Explain.
EXPERIMENT #5 - Ionic and Covalent Compounds Data and results Complete the following table with your conductivity results: Compound NaCI NazCO3 NaHCO3 KNO3 Conductivity value 43.81 67.33 70.16 36.74 Compound NHẠCI NH3 HCI HCN Conductivity value 97.20 0.83 39.29 0.00 Questions 1. Electrolytes are compounds that conduct electricity in aqueous solutions. Which compounds in your table are electrolytes? Which are not electrolytes? Electrolytes: Nonelectrolytes: 2. Would any of these electrolytes conduct electricity in the solid form? Explain.
Chapter10: Effect Of Electrolytes On Chemical Equilibria
Section: Chapter Questions
Problem 10.11QAP
Related questions
Question
Transcribed Image Text:Home
Tools
CHEM107_Lab Ex...
Lab Experiment #... x
...
1
| 2
100%
EXPERIMENT #5 - Ionic and Covalent Compounds
Data and results
Complete the following table with your conductivity results:
Compound
NaCl
Na2CO3
NaHCO3
ΚNO
Conductivity
43.81
67.33
70.16
36.74
value
Compound
NHẠC1
NH3
HCI
НCN
Conductivity
97.20
0.83
39.29
0.00
value
Questions
....
1. Electrolytes are compounds that conduct electricity in aqueous solutions. Which compounds in your table
are electrolytes? Which are not electrolytes?
Electrolytes:
Nonelectrolytes:
2. Would any of these electrolytes conduct electricity in the solid form? Explain.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

