Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter12: Alkenes And Alkynes
Section: Chapter Questions
Problem 12.32P: Arachidonic acid is a naturally occurring C„o polyunsaturated fatty acid. Draw a line-angle formula...
Related questions
Question
- Explain the purpose of Baeyer’s Test?
- Write the chemical equations for the reaction of potassium permanganate with
unsaturated hydrocarbons. What is the name of the precipitate?
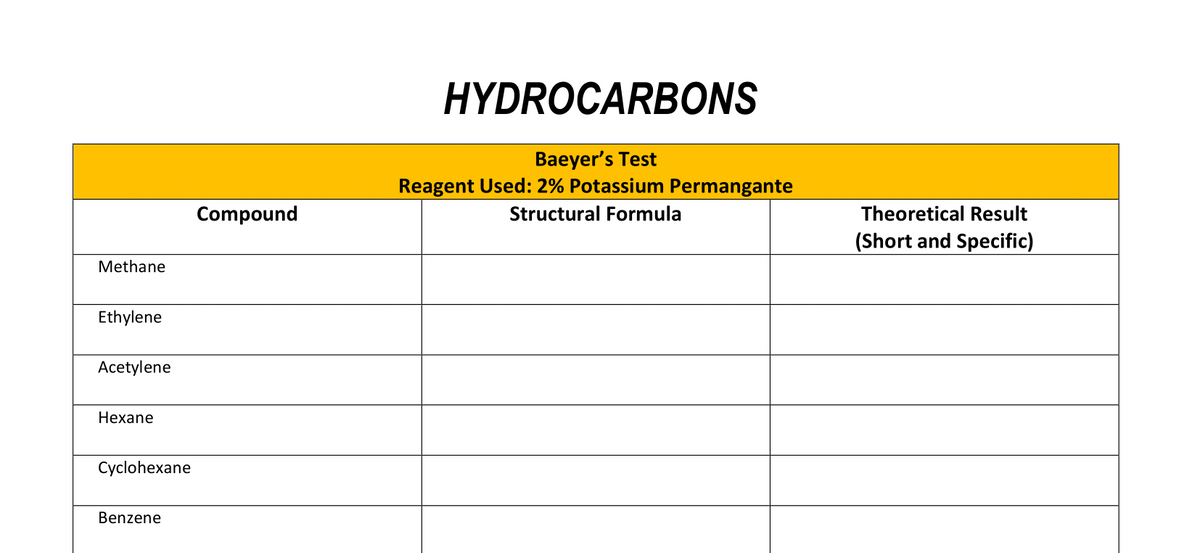
Transcribed Image Text:HYDROCARBONS
Baeyer's Test
Reagent Used: 2% Potassium Permangante
Compound
Structural Formula
Theoretical Result
(Short and Specific)
Methane
Ethylene
Acetylene
Нехаne
Cyclohexane
Benzene
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning