Chapter21: Synthesis Of N-butyl Bromide And T-pentyl Chloride
Section: Chapter Questions
Problem 2aQ
Related questions
Question
Explain the step by step process ocrruing here.
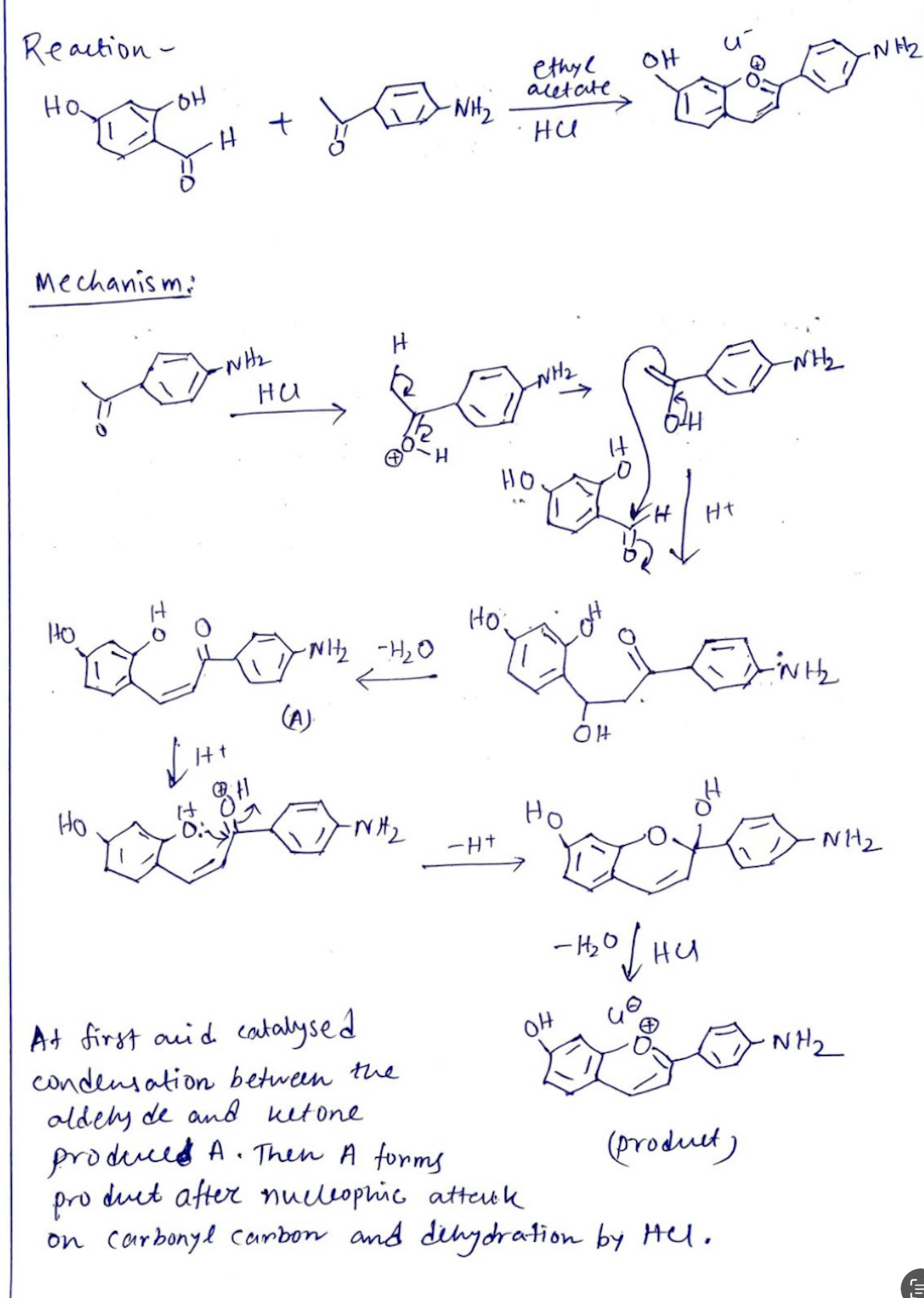
Transcribed Image Text:Reaction -
на
애
HOH
Mechanism:
HO
+ LED WHE
-NH₂
H
-NH₂
Lot for po
на
H
HO.
From
Ho
Ht
-NH2 - H2O
ethyl
acetate
ни
HO:
OH
HO
-1₂0
04
војни
Ht
gondo-
Но
-NH₂
ĐỊNH
At first and catalysed
condensation between the
aldely de and betone
produced A. Then A forms
product after nucleophic atteuck
(product)
on Carbonyl carbon and dehydration by Hel.
-NH₂
N1₂
NH₂
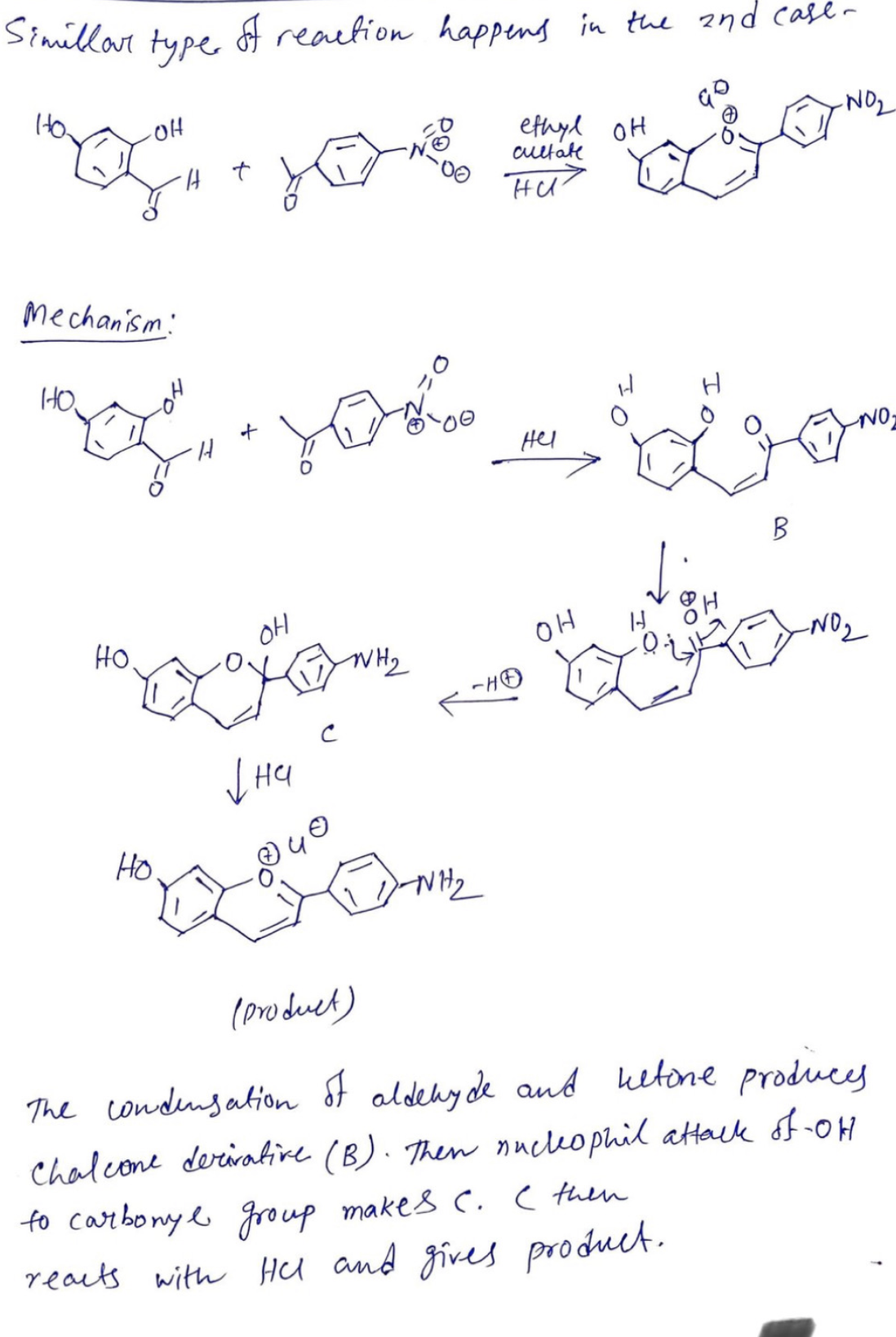
Transcribed Image Text:Simillar type
Mechanism:
HO.
OH
HO.
HO.
A
+
+
reaction happens in the 2nd case-
OH
уна
с
WH₂
-NH₂
ethyl OH
cultate
Hu
Hel
HO
OH
&
B
H
H
to carbonyl group makes C. I then
reacts with Hel and gives product.
-NO₂
-NO
(product)
The condensation of aldehyde and ketone produces
Chalcone derivative (B). Then nucleophil attack of -OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning