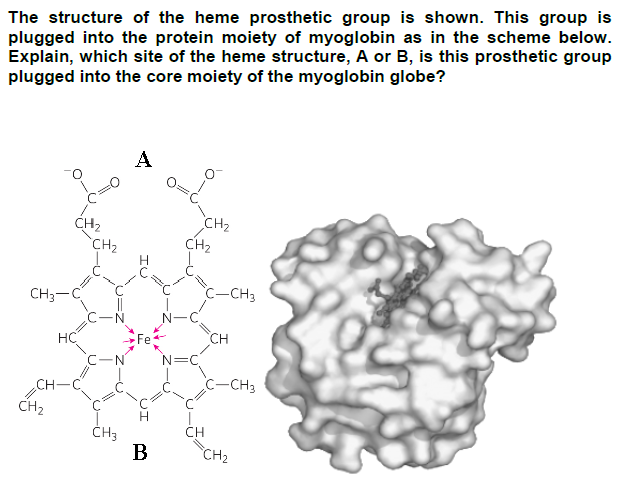
Explain, which site of the heme structure, A or B, is this prosthetic group plugged into the core moiety of the myoglobin globe? CH3- CH₂ CH₂ HC CH-C CH₂ C-N CH3 A B CH₂ CH₂ N=C CH C-CH3 CH C-CH3 CH₂
Q: Calculate how many ATP molecules will be generated from the catabolism (from b-oxidation to ETC) of…
A: Introduction: The fatty acid that is produced through diet or through degradation of triglycerides…
Q: Choose the correct properties for the given fatty acid: 0 can participate in esterification…
A: Fatty acids are important micromolecules which combine together to form lipids in plants, animals…
Q: Which of the following mechanisms of reversible enzyme inhibition describe the mode of action of…
A: Inhibition are of different type. They are mainly divided two; Reversible and Irreversible…
Q: the maximum amount of ATP that could be generated by the full oxidation of the compound…
A: In the given question, the fatty acid mentioned is CH3(CH2)4COOH. Here full oxidation means…
Q: 3-day -old female newborn with a deficiency of mitochondrial carbamoyl phosphate synthetase develops…
A: Carbamoyl phosphate synthetase is an enzyme which catalyzes the first reaction of the urea cycle.…
Q: Explain the fate of amino acids
A: Digestion of dietary proteins and degradation of cellular proteins generates free amino acids. They…
Q: Ketohexose sugars can form 8 different stereoisomers. How many of those isomers can be distinguished…
A: Carbohydrates are organic molecules arranged in form of aldehyde or ketones with multiple…
Q: Do you think obesity is a choice? What are the influences of lipids/fats mechanism in the body to…
A: Lipids are vital micronutrient that helps our body to absorbs nutrients , made hormones, even cell…
Q: What constitutes the backbone of a nucleic acid?
A: Introduction: A nucleic acid is a biological large molecule composed of nucleotide chains. These…
Q: The doctor ordered Claforan 1400mg IM every 12 hours. The directions for the 2 gm vial state: Add…
A: The doctor ordered Claforan 1400mg IM every 12 hours. The directions for the 2 gm vial state: Add…
Q: -120 2 1, 2, 4 1,3,4 60 2,3 3 180 All of the answers are correct
A: Ramchandran plot is a two dimensional plot used to visualize energetically allowed regions for…
Q: one calorie equal
A: Energy is defined as the capacity to do work. A unit is a standard of measurement. In SI system the…
Q: Which out of the following statements is true about the regulation of metabolic pathway? a) Most of…
A: The metabolic pathway involves a series of interconnected chemical reactions occurring in the cell.…
Q: Please provide the reaction mechanism of the following reaction and indicate the direction of…
A: Introduction: Glycolysis is the series of enzyme-catalyzed reaction that converts glucose into…
Q: 7.0, each into e 2 in ne ding 1. Du kan 8 10 6 itions. TABLE 2 Effect of pH on the Hydrolysis of…
A: Hydrolysis of starch is carried out in the presence of strach digesting Enzyme called amylase.…
Q: 3. What will the flow rate be in milliliters per hour for vancomycin 1g/500 mL IV, if it is to be…
A: Given Values: Total IV = 1g /500 ml Total time for infusion = 90 minutes
Q: Propose the gene for phosphoglycerate kinase is mutated and lose its function, which of the…
A: Correct answer is a- Glucose cannot be converted to pyruvate in all tissues and organs
Q: create figures using the following information and explain the results effect of the…
A: Time(mins) 15° 37° 65° 0 >3 >3 >3 2 >3 >3 >3 4 >3 1.831 >3 6 >3…
Q: Statement Analysis: Statement 1: The first law of thermodynamics states that the amount of energy in…
A: We will discuss both the statements and check if they are true or false.
Q: True or False In the presence of enzymes, the value of free energy of activiation (delta G°‡) for…
A: Enzyme: It is a biocatalyst that increases the rate of chemical reaction by lowering the the…
Q: Treatment of a polypeptide with 2-mercaptoethanol yields two polypeptides 1.…
A: Given that treatment of a polypetide with 2 mercaptoethanol yielded 2 polypeptides: 1.…
Q: List reaction or pathways of fatty acid oxidation and biosynthesis affected by insulin and glucagon.
A: Insulin is the hormone synthesized by the β cells of pancreas. Whereas glucagon is synthesized by…
Q: The nucleotide is adenine adenosine adenosine monophosphate cytidine prion
A: A nucleotide is the basic building block of nucleic acids like RNA and DNA. The nucleic acids act as…
Q: pMDawn is digested with EcoR1, and BamHI. Resulting in fragments shown below: EcoRI: 20 kb BamHI:…
A: Given: Digestion of a plasmid PMDawn with EcoRI, BamHI, and EcoRI+BamHI are as follows: EcoRI: 20 kb…
Q: Calculate the energy(Kcal.) produced from full oxidation of (3) mole of glucose (glucose --> CO2+…
A: Glucose undergoes complete oxidation via the glycolysis and the TCA cycle. When a mole of glucose is…
Q: UDP-glucuronosyltransferase enzymes bind the organic compound UDP-glucuronic acid (UDP-GA) in order…
A: Introduction: The major site for drug metabolism is the liver. It can be metabolized by oxidation,…
Q: Given the active site below, which best describes the mechanism(s) of catalysis? 5 NH 2 *H₂N. HN O…
A: Active site: The area of an enzyme's active site is where substrate molecules interact and engage…
Q: S/what is the difference between SPM and AFM² // what is polymerase chain reaction (PCR) ? Show…
A: Introduction: The polymerase chain reaction was developed by Karry Mullis in the year 1983. It is…
Q: Protein modifications occurring in the Golgi apparatus include: removal of sialic acid…
A: Introduction: Golgi apparatus is membrane-bound organelles that are sac-like present in the…
Q: During a study of digestion, a health 25-year-old woman volunteers to eat a large meal consisting of…
A: Introduction: The presence of gastric glands in the stomach undergoes gastric secretion which is…
Q: Explain why eating celery is considered ‘negative calories’ or negative net energy. Be sure to…
A: Celery is a leafy vegetable that is commonly consumed to burn calories. Most of the diets humans…
Q: Which nonvalent interaction is based on partial charges of the molecules, which are non-induced?
A: A type of chemical bond known as a noncovalent bond is most frequently seen between macromolecules.…
Q: Why is serum beta glucan testing used for diagnosing an invasive candidiasis?
A: Yeast a type of fungus. Candida is a genus of yeast. When any member of the genus Candida causes a…
Q: Based on the Hill Plot below, this protein-ligand interaction displays log Y 1-Y 2.5 0 -2.5 -2.5…
A: Hill equation developed by Archibald Hill in 1910 quantitatively describing cooperative ligand…
Q: H CH2OH ОН A CH₂OH B C Но E CH₂OH
A: Anomeric carbon is the carbon which bears a hemiacetal or acetal or hemiketal or ketal. In the…
Q: the maximum amount of ATP that could be generated by the full oxidation of the compound…
A: The given compound is a saturated fatty acid known as caproic acid. It has six carbon atoms and zero…
Q: Double-stranded regions of RNA: a. are less stable than double-stranded regions of DNA. b. can be…
A: RNA (ribonucleic acid) is a type of nucleic acid that is composed of ribose sugars, nitrogenous…
Q: TWIKKUNJUN Bacampicillin is a pro-drug of ampicillin. Esterases take function in its conversion.…
A: Bacampicillin is a pro-drug of ampicillin. Esterases take function in its conversion. Beside the…
Q: Metalloproteases can contain a catalytic triad made up of: D(H₂O)D E(Ln*)S
A: In transferase and hydrolyses enzymes catalytic triad is found. In the active site of the enzymes…
Q: P9-11 The following data on baker's yeast in a particular medium at 23.4°C were obtained in the…
A: "Since you have posted a question with multiple sub-parts, we will solve the first two subparts for…
Q: Which statement is TRUE regarding the glycolytic pathway? a. It includes five phosphate transfer…
A: The metabolic process known as glycolysis is where organisms obtain ATP energy from the breakdown of…
Q: Authophagy refers to naturally regulated mechanisms of degradation and removal of dysfunctional…
A: Denaturation is the phenomenon through which proteins or nucleic acids loses their native…
Q: On the diagram above, draw a line across each peptide bond in oligopeptide 3a. Write the…
A: Since you have asked multiple questions, we will solve the first three question for you. If you want…
Q: The key to biopolymer self-replication is: O Natural selection O Intramolecular complimentarity O…
A: Polymers refers to large molecules made up of covalently linked chain of smaller molecules known as…
Q: Avitaminosis can be caused by: O drinking red wine, but not beer O long bouts of binge gambling O…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Look at the schematic illustration of a protein (3D conformation) belpw. If the highlighted SER…
A: Proteins are the biomolecules that are made up of Aminoacids joined by Peptide bond. Proteins exist…
Q: What will happen if the enzyme renin is mixed with 1N HCL before adding milk?
A: Rennin is a proteolytic enzyme or protein digesting enzyme also know as chymosin.
Q: The diagram below shows the substrate binding cleft for a protease, providing the substrate…
A: Proteases are enzymes which digest the proteins by cleaving the peptide bonds at specific locations.…
Q: What is difference between normal primer and degenerative primer
A: Primer: These should be 18–24 nucleotides in the length and it provides for the practical annealing…
Q: Q/What are the Irreversible inhibitor ?
A: Inhibitor is an agent that slows or interferes with the chemical reaction. Enzyme inhibitor is a…
Please answer in full explaination, thank you very much.
Step by step
Solved in 2 steps

- Theoretically, the pI of myoglobin is 7.2. In phosphate buffer pH 6.0, what is the overall charge of myoglobin? How would it interact with CM Sepharose?Hemoglobin has two alpha-type chains and two beta-type chains. This would be a description of: a) primary structure b) secondary structure c) tertiary structure d) quaternary structure Myoglobin has much helix-turn-helix motif. This would be a description of: a) primary structure b) secondary structure c) tertiary structure d) quaternary structure Myoglobin folds up into a ball-like structure. This would be a description of: a) primary structure b) secondary structure c) tertiary structure d) quaternary structure Domains differ from subunits in that: a) subunits are connected by peptide bonds while domains are not b) domains are connected by peptide bonds while subunits are not c) subunits are larger than domains d) subunits are hydrophobic while domains are hydrophilicOne molecule of 2,3-BPG binds to one tetramer of hemoglobin in a central cavity of the hemoglobin molecule. Is the interaction between BPG and hemoglobin stronger or weaker than it would be if BPG bound to the surface of the protein instead? Explain your answer (hint: think about how these different situations affect the dielectric constant). (Please provide clear and sufficient explanation for the question, thank you!)
- What is inositol trisphosphate (IP3)?If instead of using 3.5 µM myoglobin (receptor) you used half of this (that is, 1.75 µM myoglobin), what would be that value of the Kd, that you calculated ( how would it change)? Please explain so I can solve on my own :) (How does changing concentration of the receptor in a ligand-receptor binding experiment affect the dissociation constant?)Sickle-cell hemoglobin (HbS) differs from normal human adult hemoglobin (HbA) by a single mutational change, Glu6 S Val, which causes the HbS molecules to aggregate under proper conditions. Under certain conditions, the HbS filaments that form at body temperature disaggregate when the temperature is lowered to 0°C. Explain.
- using examples, discuss the classification of proteins into fibrous and globular classification. - I would want to use myoglobin as an example of glubular but I don't know if that falls in the category for fibroushow do salt bridges that include amino-terminal carbamate stabilize the deoxy form of hemoglobin. Please answer it asap.... With detailed explanation...From the figure of O2 binding to myoglobin and hemoglobin (ignore the linemarked as T) as described in lecture (shown below) answer the following questions. a) Estimate the P50 for myoglobin from the plot. Show how this estimation isdetermined from the binding curve above. ( The first ghraph) b)Using YO2 = PO2/P50 + PO2 , calculate the fraction of O2 bound for myoglobin at 1 torr. (2nd graph) c)Using the binding curve on the previous page, show how you can estimate whatfraction of hemoglobin is bound near tissues at a pO2 of 30 torr and provide this value. If the pH were lowered, will the amount of O2 bound to hemoglobin at 30 torr increaseor decrease? Explain why this is so based on how this changes hemoglobin structure. If 2,3-BPG were added to the solution, will the amount of O2 bound to hemoglobin at30 torr increase or decrease? Explain why this is so based on how this changes hemoglobinstructure.
- The molecular formula for glucose is C6H12O6. What would be the molecular formula for a polymer made by linking ten glucose molecules together by dehydration reactions? Group of answer choices A. C60H120O60 B. C60H102O51 C. C60H100O50 D. C60H111O51 Use the following information to answer the following questions."The native structure of hemoglobin (HB) comprises of two α and two β subunits, each of which carries a heme group. There appear to be no previous studies that report the in-vitro folding and assembly of Hb from highly unfolded α and β globin in a 'one-pot' reaction. One difficulty that has to be overcome for studies of this kind is the tendency of Hb to aggregate during refolding. This work demonstrates that denaturation of Hb in 40% acetonitrile at pH 10.0 is reversible." (J Am Soc Mass Spectrum 2007, 18, 8-16)In sickle-cell disease, as a result of a single amino acid change, the mutant hemoglobin tetramers associate with each other and assemble into large fibers.…In a Myoglobin and azide ligand-receptor binding experiment, instead of using 3.5 µM myoglobin you used half of this (1.75 µM myoglobin), what would be that value of the Kd, that you calculated (how would the Kd change)?Magnesium blocks voltage-gated calcium channels on the membrane of smooth muscle cells. How do barium ions affect smooth muscle on the molecular level?

