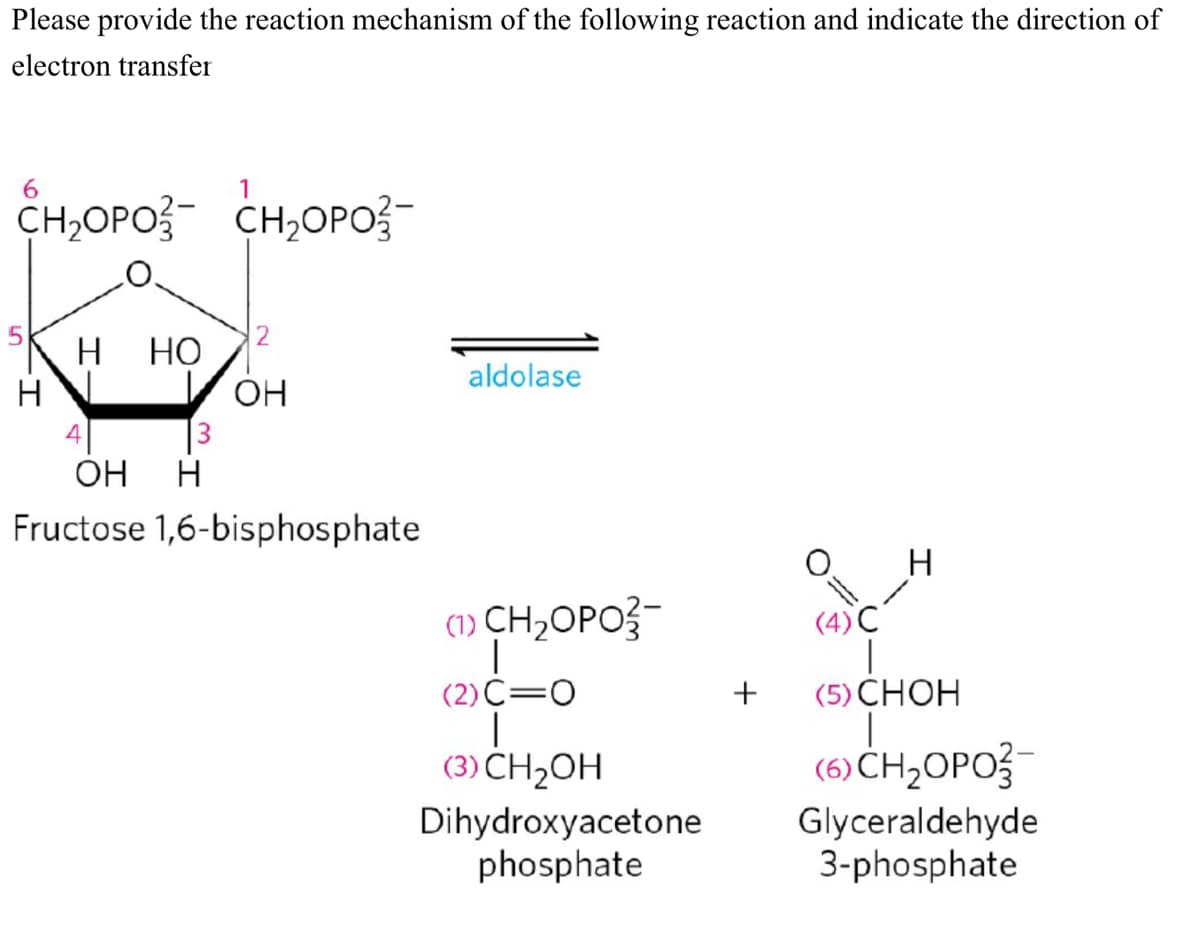
Please provide the reaction mechanism of the following reaction and indicate the direction of electron transfer 6 CH,OPO- CH,OPO- H HO aldolase OH 3 OH H 1,6-bisphosphate H (4) C (1) CH₂OPO3- (2) C=O T (5) CHOH | (3) CH₂OH (6)CH,OPO3 Dihydroxyacetone phosphate Glyceraldehyde 3-phosphate H Fructose +
Q: Question #9 In the kidney the malate:aspartate shuttle is the only system that transports the…
A: The main function of the malate aspartate shuttle is to perform the trasnportation of NADH. This…
Q: The first process in breaking down glucose is * A) the citric acid cycle. B) glycolysis. OC) the…
A: Breaksdown of glucose occurs by three stages in cellular respiration . They are Glycolysis Krebs…
Q: Using good details, compare and contrast the pairs of different biochemical reactions. Create your…
A: Introduction: The term metabolism describes the interconversion of chemical compounds in the body…
Q: 1. An aquaponics system is a system in which fish and plants are grown together. Shane Ahrens wanted…
A: "Since you have posted a question with multiple sub-parts, we will solve the first two subparts for…
Q: What is the similarity among eicosanoids, cholesterol and phospholipids in terms of their…
A: Eicosanoids are biologically active molecules, which act as mediators of inflammation, regulators of…
Q: Adenylylation is a post-translational modification that can regulate the Adenylyation involves the…
A: Introduction: In living organisms, hundreds of different enzyme-catalyzed reactions occur…
Q: f the following statements is/are FALSE? aramagnetic metal ions can have an odd number of electrons.…
A: The crystal field theory is used for giving the description of the metal-ligand bond. The…
Q: During each cycle of B-oxidation of fatty acid, all the following compounds are generated except…
A: Beta-oxidation is the process by which long chain fatty acyl CoA is degraded. Fatty acid oxidation…
Q: 10 8 6 4 2 0 -2 -4 -6- -6 -4 -2 0 2 4 log [L] (nM) 6 8 10
A: Introduction: The interaction of proteins with proteins, small molecules, lipids, nucleic acids, and…
Q: نقطة واحدة * intrinsic factor is serected by intrinsic factor Parietal cells ΟΟΟ Chief cells O G…
A: Structure of Intrinsic factor: Genuine Cbl is identified by an inherent component detected in…
Q: Which items or conditions improve iron absorption in the mammalian gut O vitamin C O compounds in…
A: Iron is the most vital component of Hemoglobin which is present in blood. Hemoglobin is a type of…
Q: Which of the following is an aldo-tetrose?* НО H НО Н Н OH -OH H -OH -ОН н OH CH2OH CH₂OH (a) Н Н НО…
A: Tetroses are monosacharide sugars that contain 4 carbon atoms. Aldoses are the sugars that contain…
Q: For i) fresh milk casein, ii) raw egg protein, explain from the protein structure why it can…
A: Proteins are classified as a very large types of biomolecules. These are the ones that are made up…
Q: A patient of 28 years old complains of pains in the spine, persistent arterial hypertension. On…
A: Hi! Since you have posted a multiple subparts questions and haven't mentioned which subparts to be…
Q: The free energy difference going from the unfolded state to the folded state in most proteins is…
A: The three dimensional structure of proteins ca b e destroyed by denaturating the protein. This…
Q: Modify isoleucine to show the predominant forms at pH 1, 7, and 13. Isoleucine has pK, values of 2.4…
A: Proteins are polymer of amino acids , and each of the amino acid residue is linked to its…
Q: Describe the chemistry involved in the processes of Lipid Interesterification and Lipid…
A: Lipid interesterification is a process where the structure of a lipid is changed by altering the…
Q: Which of the following statements is true? a) High insulin/glucagon ratio activates lipolysis in…
A: Lipolysis is the process in which the lipid, triacylglycerol is broken down into its components…
Q: What drives the transport of fatty acids for b-oxidation into the mitochondrial matrix? The…
A: The transport of fatty acids from Cytosol into the mitochondrial matrix occurs by carnitine shuttle…
Q: What glycolytic intermediate is fructose converted to in the muscle, such that it can be utilized in…
A: Fructose is an abundant dietary monosaccharide that is present naturally in fruits and vegetables…
Q: Which of the following is true about the T (tense) -->R (relaxed) transition of hemoglobin? A. The…
A: Haemoglobin is described as a very important protein that is present in our RBCs. They have the role…
Q: You apply a new drug to a different batch of neurons and record membrane potential changes in the…
A: Permeability barrier and semi permeability of cell membrane are both maintained by lipids. only…
Q: Every amino acid that undergoes catabolic breakdown give rise to an ammonia molecule that must be…
A: There are 20 naturally occurring amino acids that are found in proteins. Amino acids can be…
Q: A peptide has the following sequence: Gly-Ala-Lys-Phe-Asp-Met-Val-Pro-Arg-Ala-Leu. What is the net…
A: The amino acids contain ionizable groups. The ionic form and charge of the amino acids depend upon…
Q: Modify the amino acid by adding or removing atoms or bonds and by adding charges where appropriate.…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: 1.0 0 0.5 0 5 Ligand A Ligand B 10 15 20 25 Ligand Ligand D 40 45 50 55 60 30 35 [Ligand] (MM)
A: Kd is dissociation constant which is ligand concentration at half of saturation or at 0.5 theta…
Q: create figures using the following information and explain the results effect of the…
A: Time(mins) 15° 37° 65° 0 >3 >3 >3 2 >3 >3 >3 4 >3 1.831 >3 6 >3…
Q: Give the base sequence of the complementary DNA strand of the DNA chain with the following base…
A: DNA is a macromolecule composed of individual subunits known as nucleotides. Each nucleotide…
Q: What is the relation between GMO crops and the four of the principles of bioethics?
A: The word GMO stands for Genetically Modified Organism. GM crops or Genetically Modified crops have…
Q: How much ATP is produced from 1-stearoyl-2-arachidonyl-3-tridecanoyl glyceride? Show complete…
A: The given triglyceride molecule will be converted to stearic acid, arachidonic acid, tridecanoic…
Q: The image below shows the different interactions responsible for the spontaneous folding of a…
A: Protein protein interactions are of different kinds, they are responsible for holding the protein…
Q: Select all criterion for classifying a substance as a neurotransmitter. (select all that apply)…
A: Neurotransmitters are chemical messengers that are employed by the body to transmit nerve impulses…
Q: Describe the use of simple paths to determine the change in these by more complicated paths (ie…
A: Hess's Law states that the change in enthalpy of a chemical reaction will be the same , no matter…
Q: For effective metabolic pathway regulation, a(an) ______ binds to the enzyme catalyzing the first…
A: The enzyme acts as a catalyst in various metabolic reaction that takes place in our body.
Q: A student Hussein woke up early and then went quickly - without having breakfast - to Al-Maaqal…
A: Glucose is a very important source of energy & it is required by our body to carry out normal…
Q: 2. The kinetics of an enzyme are measured as a function of substrate concentration in the presence…
A: The enzymes catalysts that facilitate biochemical reactions. The enzymes are inhibited in presence…
Q: Predict whether the entropy change of the system in each of the following is positive or negative.…
A: Introduction: Entropy is defined as the property of a system that measures the degree of disorder or…
Q: ou are subsequently instructed to improve the biochemical function of pyruvate kinase, which aids in…
A: The enzyme Pyruvate Kinase is involved in glycolysis. Pyruvate kinase catalyzes the final phase of…
Q: Type I diabetes is caused by autoimmune destruction of the pancreatic beta cells True O False
A: Diabetes is a very common disease in the world. It is associated with metabolism of glucose. It can…
Q: The intense acidification observed in the stomach lumen is energetically driven by O a parietal cell…
A: The acidic nature of gastric secretions have been observed in many animals including humans. The…
Q: What is the preparation condition for culturing a mould sample?
A: Introduction: Mold is a fungal specimen which is grown in filamentous structure called hyphae. They…
Q: Explain in detail the condensation reactions to form Maltose and Sucrose from their monosaccharides.
A: Sugars are simple carbohydrates which are classified based on the number of monomeric units present…
Q: Explain polygenic inheritance (EC)
A: Polygenic inheritance is a condition where the inheritance of a particular trait is controlled by…
Q: 11D The T-state structure of hemoglobin is an example of a whereas its alpha helices are an example…
A: Introduction: Hemoglobin is a respiratory pigment found in red blood corpuscles. It is made up of…
Q: Which of the following mutagens results in the deamination of nitrogenous bases? O nitrous acid base…
A: The amino bases adenine and cytosine lose one amino group when they are oxidised. As a result, in…
Q: A B C E D FO
A: Omega-6 and omega-3 fatty acids are polyunsaturated fatty acids also called PUFA. The scientific…
Q: ILLUSTRATIONS. For each of the given proteins: ● Draw the final location of the following proteins…
A: The process of transcription occurs in the nucleus following which the mRNA is translated in the…
Q: What characterizes the C5 amino acids? These amino acids are converted to glutamate then deaminated…
A: Amino acid metabolism is divided into pathways based on the lengths of carbon structures involved.…
Q: A new virus has recently been discovered that infects human lymphocytes. The virus can be grown in…
A: Viruses are of two types based on their genetic material present inside their protein capsid. DNA…
Q: All of the following are considered “weak” interactions in proteins, except: a. hydrogen bonds. b.…
A: Proteins are biomolecules with different levels of conformation. Primary, secondary, tertiary, and…
Step by step
Solved in 2 steps with 1 images

- The high phosphoryl transfer potential of 1,3-bisphosphoglycerate is explained by the resonance stability of the product, 3-phosphoglycerate. The same explanation applies to the reaction that generates resonance stabilized pyruvate from phosphoenolpyruvate. Show the resonance structures for both products, 3-phosphoglycerate and pyruvate, and state why the reactant in both reactions is not resonance stabilized.If you were to determine the P/O ratio for oxidation of α-ketoglutarate,you would probably include some malonate in your reaction system. Why?Under these conditions, what P/O ratio would you expect to observe?The mechanism involved in the reaction catalyzed by phosphoglyceromutase is known to involve a phosphorylatedenzyme intermediate. If 3-phosphoglycerate is radioactively labeledwith 32P, the product of the reaction, 2-phosphoglycerate, does nothave any radioactive label. Design a mechanism to explain these facts.
- Given each set of information which may include common name(s) and the reaction catalyzed, you are required to identify the main class of the specific enzyme described. _____________________1. Name: alkaline phosphataseReaction: a phosphate monoester + H2O = an alcohol + phosphate_____________________2. Reaction: L-threonine = D-threonine.Other information: Inverts both chiral centers, a racemase. _____________________3. Name: glycine-N-acylaseReaction: acyl-COA + glycine = CoA + N-acylglycine_____________________ 4. Name: lysine decarboxylaseReaction: L-lysine = cadaverine + CO2 _____________________5. Name: methanol dehydrogenaseReaction: methanol + NAD+ = formaldehyde + NADH + H+ _____________________6. Name: citryl-CoA synthetaseReaction: ATP + citrate + CoA = ADP + phosphate + (3S)-citryl-CoA_____________________7. Name: D-xylulose reductaseReaction: xylitol + NAD+ = D-xylulose + NADH + H+ _____________________8. Name: cellobiose phosphorylaseReaction: cellobiose phosphate =…If you were to determine the P/O ratio for oxidation of a-ketoglutarate, you would probably include some malonate in your reaction system. Why? Under these conditions, what P/O ratio would you expect to observe?The isoelectric point (pI) of 6-phosphogluconate dehydrogenase is 6.0. Explain why the buffer used in DEAE cellulose chromatography must have a pH greater than 6 but less than 9 in order for the enzyme to bind to the DEAE resin.
- Draw the structure of two different aldohexoses that yield the followingaldaric acid when oxidized with HNO3. Use Figure to name eachaldohexose5-phosphate) and an aldopentose (ribose-5-phosphate) to an aldotriose (glyceraldehyde-3-phosphate) and a ketoheptose (sedoheptulose-7-phosphate). Notice that the total number of carbons in the reactants andproducts is the same (5 + 5 = 3 + 7). Propose a mechanism for this reaction. xylulose-5-PCH2OH CH2OPO32−C OHOHHOH sedoheptulose-7-PIn order to function as an oxidative phosphorylation uncoupler, 2,4-dinitrophenol must act catalytically, not stoichiometrically. What does this mean? Identify and discuss an important implication of this conclusion.
- The equilibrium constant Kc for the reaction fructose-1,6-diphosphate ⇋ glyceraldehyde-3-phosphate + dihydroxyacetone phosphate is 8.9 x 10-5 M at 250C and the behavior is assumed to be ideal. Calculate .for the process (standard state: 1M) Suppose that we have a mixture that is initially 0.010 M in fructose-1,6-diphosphate and 1.0 x 10-5 M in both glyceraldehyde-3-phosphate and dihydroxyacetone phosphate. What is ?Assume that the reaction Arabinose +Pi <> Arabinose-6-P has a delta G of +13.0 kJ/mol and the reaction XTP <> XDP + Pi has a delta G of -25.7 kJ/mol. Calculate the delta G for the combined reaction Arabinose + XTP <> Arabinose-6-P + ADP, in kJ/mol to four significant figures.The equilibrium constant Kc for the reaction fructose-1,6-diphosphate ⇋ glyceraldehyde-3-phosphate + dihydroxyacetone phosphate is 8.9 x 10-5 M at 250C and the behavior is assumed to be ideal. Calculate the delta G for the process Suppose that we have a mixture that is initially 0.010 M in fructose-1,6-diphosphate and 1.0 x 10-5 M in both glyceraldehyde-3-phosphate and dihydroxyacetone phosphate. What is the delta G

