Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
100%
What is number 5?
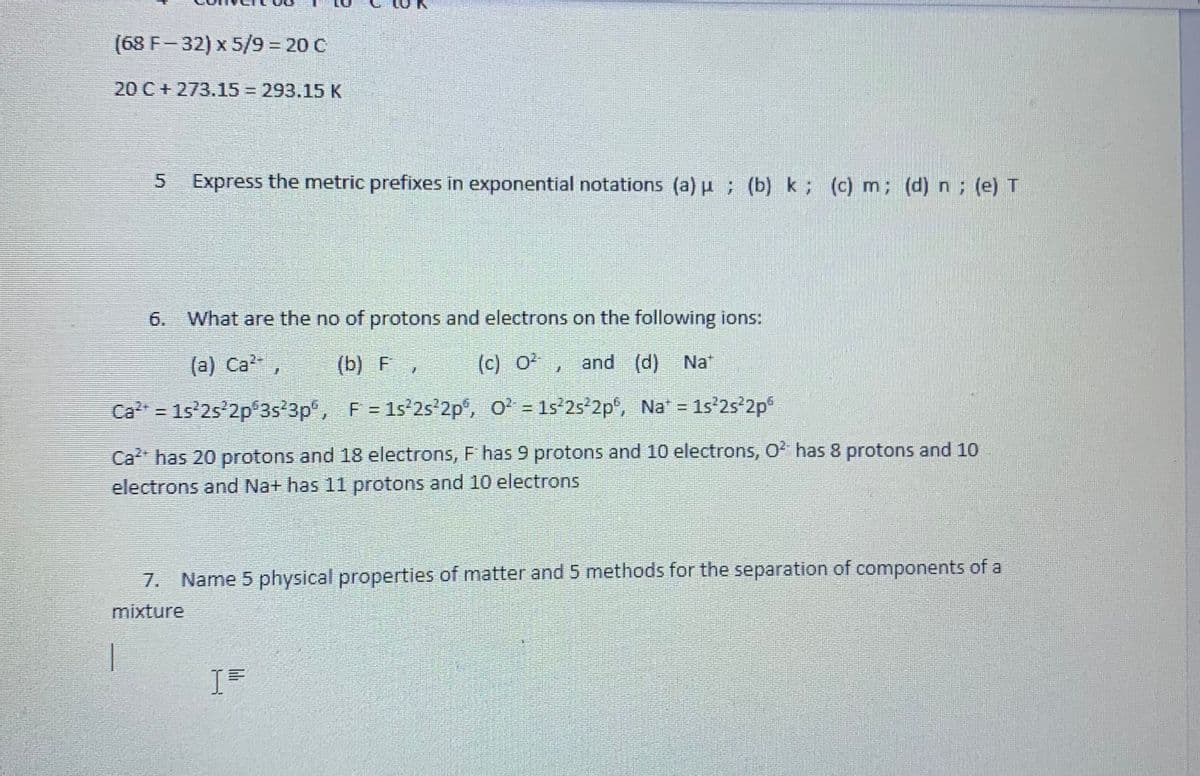
Transcribed Image Text:(68 F-32) x 5/93D20 C
20 C+ 273.15 = 293.15 K
Express the metric prefixes in exponential notations (a) u ; (b) k; (c) m; (d) n; (e) T
6.
What are the no of protons and electrons on the following ions:
(a) Ca? ,
(b) F ,
(c) 0 , and (d)
Nat
Ca = 1s 2s 2p 3s 3p, F = 1s 2s2p, O = 1s 25 2p, Na" = 1s 2s 2p
Ca has 20 protons and 18 electrons, F has 9 protons and 10 electrons, O' has 8 protons and 10
electrons and Na+ has 11 protons and 10 electrons
7. Name 5 physical properties of matter and 5 methods for the separation of components of a
mixture
%23
5.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



