Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the aldol…
A: Step 1: Step 2: Explanation: Reaction of carbanion with aldehyde produces beta-hydroxy carbonyl…
Q: 19. the concentration of a reactant is 6.25%, how many half-lives has it gone through? A. 7 B. 6 C.…
A: Initially there was 100% reactant. After one half life the initial amount of reactant will half.…
Q: I need expert answer solutions and step by step
A: The objective of this question is to propose a multi-step synthesis for the starting materials for…
Q: None
A: Step 1:
Q: Please make sure answer is correct a lot of controversial answer on this I will trust you and take…
A: The sequence of reactions is: Double elimination from vicinal dihaloalkanes ⟶ alkyne ⟶cis-alkene ⟶…
Q: Write the systematic name of each organic molecule: structure CI CH3 CI C=CH-CH-CH3 CH3 CI CH=C CH3…
A: Step 1: Identify the longest continuous chain containing the double bond (C=C). Number the carbon…
Q: When a 22.7 mL sample of a 0.472 M aqueous hydrofluoric acid solution is titrated with a 0.493 M…
A: The objective of this question is to calculate the pH at the midpoint of a titration between…
Q: None
A:
Q: I completed an experiment "Iron contenr of fruit and vegetable through complex ion formation" How…
A: The number of moles of a substance is the amount of a substance in which there are 6.022×1023…
Q: The active ingredient of bleach such as Clorox is sodium hypochlorite (NaClO). Its conjugate acid,…
A: Given:The word pH stands for the potency of hydrogen and it is used to measure the concentration of…
Q: Please correct answer and don't use hend raiting
A: First we will Calculate the number of moles of each component using their molecular weightsthen we…
Q: None
A: To identify the oxidizing agent and the reducing agent in the given reaction:2KCl + MnO2 + H2SO4 →…
Q: An important reaction involved in the operation of vacuum tubes is the following y BaO(s) + x M(XM,…
A: The objective of the question is to calculate the partial pressure of barium in the given…
Q: Claisen Schmidt Condensation Unknown Product C19H180 Unknown IR and NMR Spectrums 100 50 D 4000 3000…
A: The objective of the question is to determine the product of a Claisen Schmidt Condensation reaction…
Q: What changes occur in the structure of a protein molecule when it becomes denatured? Ochange in…
A: Approach to solving the question:To solve the question, we need to understand the concept of protein…
Q: Show reaekson and don't use hend raiting and step by step solutions
A: I can't access the specific image you sent, but I can still explain why alkenes undergo alkene…
Q: O Hydrocarbons Naming and drawing linear alkenes with one double bond Write the systematic name of…
A: Naming Alkenes Step 1: Find the longest carbon chain Step 2: Begin numbering from the terminal end…
Q: 3 NEXT > < PREV 1 2 Based on your ICE table (Part 1) and the definition of Ka, set up the expression…
A: For Parts 1 to 3, a buffer system was prepared with HC7H5O2 and NaC7H5O2. The reaction for the…
Q: Could the following pair of compunds be seperated through recristilization or destilation and why?
A: This two are Diastereomer can't be separated by recrystallization or distillation, as both have same…
Q: A certain half-reaction has a standard reduction potential Ered = -0.08 V. An engineer proposes…
A:
Q: Which of the following describes a metallic substance? A tightly packed lattice of positively and…
A: The objective of the question is to identify the correct description of a metallic substance among…
Q: By titration, it is found that 62.5 mL of 0.176 M NaOH(aq) is needed to neutralize 25.0 mL of…
A: Step 1:To find the concentration of the HCl solution, we can use the equation for neutralization…
Q: 5. Devise concise syntheses for the following transformations. Clearly show the reagent and product…
A: Explanation : 5a. In the first step excess NaNH2 generates the alkyne. Then in the next step NaNH2…
Q: When treated with base, the following compound undergoes an intramolecular aldol reaction and…
A: Step 1:Aldol condensation reaction is a base catalyzed reaction where the acidic hydrogen at…
Q: Construct an "I-C-E" table that describes the equilibrium system of HCOOH with a nominal…
A: To create an ICE table for the dissociation of formic acid (HCOOH) initially at 0.50 M, we'll follow…
Q: If 278.8 mL of SO2 is allowed to react with 158.7 mL of O2 (both measured at 314 K and 48.0 mmHg),…
A: The objective of the question is to determine the limiting reactant in the reaction between SO2 and…
Q: The solubility of Ag,CO, in water at 25 °C is measured to be 0.035 Round your answer to 2…
A: Given:Solubility of Ag2CO3 = 0.035 g/LFind: Ksp = ?Steps to follow:Convert,g/L Ag2CO3 → mol/L…
Q: None
A: To name the provided alkene using E/Z nomenclature, we must first determine the priority of…
Q: ideal gas is taken (Cv is taken (Cv = 3R/2). Volume Vc = 8 expansion, with p₁ = 10.0 atm and V =…
A: The objective of this question is to determine the process where heat is added to the gas, Identify…
Q: A famous Italian chef wants to open a restaurant in one of the cities (Mumbai, Denver, Mexico City).…
A: At the boiling point, the vapor pressure of a liquid and the atmospheric pressure are equal. At this…
Q: Based on your ICE table (Part 1) and the definition of Ka, set up the expression for Ka in order to…
A:
Q: help 8
A:
Q: Homework: The pK values of glutamate (Glu, E) are: 2.19, 4.25 and 9.67. You have 10 mL of 1M…
A: The objective of the question is to calculate the .
Q: hello , can you solve this question with using chemical kinetics?
A: Below are the steps of how to solve this decay problem: Step 1: General Approach:This problem…
Q: (8) The cell: SCE//HA(0.250M), NaA(0.170M)/H2(1.00bar), Pt has a poten- tial of -0.721V at 298K.…
A: The objective of the question is to Sketch the cellCalculate the Potential of the CellCalculate the…
Q: What are the symmetry elements and point group of each of the following?
A:
Q: What is the molarity, M, of ions in a 0.60 M aqueous solution of sodium sulfate, Na2SO4? 0.30 M 0.20…
A: The objective of this question is to find the molarity of ions in a 0.60 M aqueous solution of…
Q: Show reaekson and don't use hend raiting and step by step solutions please
A: Step 1:Step 2:The reaction simply follows the SN2 mechanism which is a most favoured solvent that is…
Q: A buffer solution is 0.341 M in CH3COOH and 0.270 M in CH3COONa. If Ka for CH3COOH is 1.8x 10^-5 ,…
A: The objective of this question is to calculate the pH of a buffer solution. The buffer solution is a…
Q: Sn2 and N (starting concentration 10 mol/L) should be separated by fractionated precipitation using…
A:
Q: Hydrogen peroxide (34.02 g/mol) decomposes into water and oxygen gas according to the reaction 2…
A: The objective of the question is to determine the pressure of O2 gas produced from the decomposition…
Q: Which substance would you expect to have the LOWEST melting point? CH3F CH3OH Ni LiCl
A: The objective of the question is to determine which among the given substances - CH3F, CH3OH, Ni,…
Q: Complete the table below, which lists information about some diatomic molecules or molecular ions.…
A: The objective of the question is to determine the stability, magnetic nature (diamagnetic or…
Q: Draw the missing organic structures or select the missing reagents in the following multistep…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: None
A: Step 1:Explanation Step 2: Step 3: Step 4:
Q: 1d.) Draw mechanism of cationic ring-opening polymerization of monomer M initiated with propargyl…
A: Here's a breakdown of the mechanism for cationic ring-opening polymerization of monomer M initiated…
Q: How to classify the ionic compounds according to the mole ratio between the cation and anion?
A: An ionic compound is made up of ions that form charged particles when an atom gains or loses…
Q: Which statement is TRUE for an acid-base neutralization reaction? The reaction will take place if…
A: The objective of the question is to identify the correct statement about an acid-base neutralization…
Q: Draw the starting structure that would yield this product under these conditions. 1. LDA, -78 °C 2.…
A: Step 1: Step 2: Step 3: Step 4:
Q: Please answer question 3.
A: Here's why acetone spontaneously evaporates at room temperature, even without considering specific…
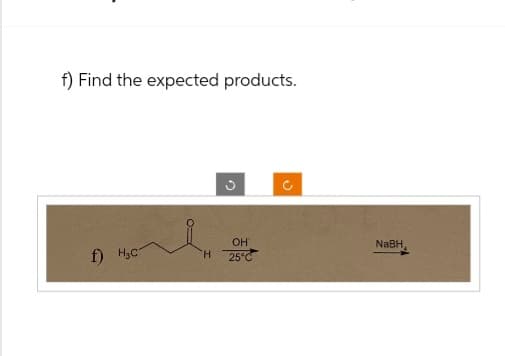
Step by step
Solved in 2 steps with 2 images

- Predict the product in the combination reaction below. Al (s) + N2 (g) Select one or more: a. Al3N b. AlN3 c. AlN d. Al3N2 e. AlN2Predict the product in the combination reaction below. Al (s) + N2 (g) → Select one or more: a. Al3N b. AlN3 c. AlN d. Al3N2 e. AlN2Predict what the product for each reaction is
- Consider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:Predict the product for each reaction and predict the products. Explain answersWhat is the correct product ?

