Q: Using line structures, give major products of each reaction
A: We have to give the major product of the below given reactions. Introduction: Dehydration reaction,…
Q: The reaction A + 2B → C has the rate law rate = K[A] [B]. By what factor does the rate of reaction…
A: Answer: Power of concentration term of a reactant in the rate law is the order of reaction with…
Q: When 2.50 g of methanol undergoes complete combustion with excess oxygen 50.8 kJ of heat energy is…
A:
Q: Part A Use oxidation states to identify the element that is being oxidized in the following redax…
A: We have to determine element oxidized element reduced in the given reaction.
Q: Predict the products of the following reactions. (a) EtO O OEt (1) EtMgBr (excess) (2) H₂O+ S Draw…
A: Grignard reagents are Alkyl Magnesium bromide(RMgX). Grignard reagents reacts with aldehydes or…
Q: Complete each of the following by supplying the missing reagents. Draw the structures of each of the…
A: The given reactions are examples of amide hydrolysis reaction. In these reactions carboxylic acid…
Q: m. cyclohexanone n. 1,4-pentanediol CH₂OH (excess) TSOH PCC
A: We have to give the product of the below given reactions. m. Cyclohexanone →TsOHCH3OH excessn.…
Q: I’m stuck in this those question To make 1L of 250mM NaCl, I would add ——-grams of NaCl to a…
A: Given question about preparation of solution 1 L of 250mM NaCl , 50 mL of 2% NaCl and 200 mL 1x…
Q: Carbonate, CO3², has three resonance structures. Draw these resonance structures and then draw the…
A:
Q: Determine the moles of PbI2 product and then usw the stoichiometric ratio to calculate teh actual no…
A:
Q: Please draw the mechanism for the formation of benzalacetone, 4-phenylbut-3-en-2-one.
A: Aldehydes with β-hydrogen condense with a diluted base to form β-hydroxy aldehydes, also known as…
Q: [Review Topics] Use the References to access important values if needed for this question. According…
A: According to the question, n =1.057 mol R = 0.0821 L atm /K molT = 275.0 K V = 1.559 L a = 3.592…
Q: Liquid hexane (CH₂(CH₂) CH3) reacts with gaseous oxygen gas (O₂) to produce gaseous carbon dioxide…
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: A tank originally contains 500 L of water with a salt concentration of 2 grams per liter. Water with…
A: the rate of change of a quantity over time. In this case, the quantity being described is the amount…
Q: What quantity in moles of NaCl are in 61.7 grams of NaCl?
A:
Q: Mass of empty 100 mL beaker Mass of beaker plus distilled water Mass of beaker plus solution of salt…
A: Molality of any solution is defined as the number of moles of solute dissolved in 1 Kg (1000 gm) of…
Q: quantum number. ): Identify electron based on its Please identify the atomic orbitals that the…
A: We know that Quantum numbers of electron in an orbital is expressed in the form of (n, l, ml)…
Q: 5. A solution is prepared by mixing 50.0 mL of 0.10 M Pb(NO3)2 with 50.0 mL of 1.0 M KCI (Ksp…
A:
Q: "Smelling salts," which are used to revive someone who has fainted, typically contain ammonium…
A:
Q: 1 LOD TRANSMETTANCEI D 4000 Но 2 3000 3 2000 4 пр HAVENUMBRI- 600 5 100 6 H ОН 7 500
A: We need to find the molecules indicated by the given IR spectra. IR spectrum provides characteristic…
Q: Convert to dash-wedge
A: Since, In the figure of the question, it is Newmann form. When change to wedge-dash keep in mind…
Q: 8. Consider the following reaction: CO(g) + H₂O(g) = CO₂(g) + H₂(g) Kc = 107 (at 500K) If a reaction…
A:
Q: What quantity in moles of Nal are in 82.3 grams of Nal ?
A:
Q: 96. The major product formed in the following reaction is Br H n-Bu3SnH, AIBN benzene reflux SnBu
A: Well, this reaction proceeds through the free radical mechanism. due to the formation of free…
Q: 2. Two substances, A and B, have the same molar extinction coefficient at 260 nm: 1.80 x 10…
A:
Q: What is the rate for the second order reaction A → Products when [A] = 0.298 M? (k = 0.761 M-¹s¹)
A: In this question, we will determine the rate of the second order reaction. Given that, A…
Q: Gaseous methane (CH4) reacts with gaseous oxygen gas (O₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: N₂ + 3H₂ → 2NH3 What is the mole ratio between Nitrogen and Ammonia in the above reaction? 4 1 moles…
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: QUESTION 4 Which of the compounds C4H10, CaCl2, Cu(NO3)2, PC13 are expected to exist as molecules? O…
A: Solution - Molecule consist of covalent bond . C4H10 is butane which consist of covalent bond . 13…
Q: For a certain reaction, the frequency factor A is 6.3 x 10's and the activation energy is 23.4…
A: Answer: This question is based on Arrhenius equation that gives us relation between rate constant,…
Q: 2. Why should anhydrous ether be used as the solvent for the Grignard reaction?
A: Anhydrous ether is used as solvent for the Grignard reaction. The main reasons of using anhydrous…
Q: 2kdoz 2kd +30₂ How m many moles of oxygen are produced when 6.74 moles of Koloz decompose completely…
A:
Q: 7985J of energy are applied to a 651 g sample of aluminum at 21.0 celcius. What is the final…
A: Answer: When energy is applied to a metal it gains that and its temperature rises and that…
Q: Question 10 OTS NaOCH₂ CH3OH pKa of CH3OH is 16 The reaction above is expected to follow a [Select ]…
A:
Q: time (s) 135 265 404 544 692 838 995 Experiment # 1 Total Moles Consumed 2.00E-04 4.00E-04 6.00E-04…
A: Solutions- Here we have to find the Rate of chemical reaction with the help of given values of time…
Q: 7. Draw the resonance structure and use the curved arrow notation to show how they can be…
A: The molecule contains one negative charge and conjugated carbonyl groups, hence several resonating…
Q: C3H9N has four constitutional isomers (structures with the same formula but a different arrangement…
A: We have to draw the structure of 4 constitutional isomers of C3H9N
Q: Two analytical methods (potentiometry and amperometry) were used to determine the concentration of…
A:
Q: Given the reaction: 2 NOF(g) ⇌ 2 NO(g) + F2(g) at a certain temperature the value for KP is 1200.…
A:
Q: Ranking the stability of carbocation intermediates Rank the carbocations in the table below in order…
A: The carbon next to the carbon with positive charge is called alpha- carbon. More the number of…
Q: naming with the following
A: IUPAC nomenclature is a systematic method of naming chemical compounds recommended by the…
Q: 1. Differentiate the following terms: a. Rate law b. Rate order
A: The terms "rate law" and "rate order" are related concepts in chemical kinetics, the study of…
Q: Complete the reaction when (NH 4) 2SO 4 is dissolved in water? (NH 4) 2SO 4 (s) →
A: Here, we have to find the complete reaction for the dissolution of (NH4)2SO4.
Q: Use IUPAC naming rules to name the following hydrocarbon compounds: a) CH3CH=CH=CH-CH=CH b) I CH₂ I…
A:
Q: 8. Give the formal charges of atoms N :0= B -C= --
A: Formal charge is a concept in chemistry that is used to predict the distribution of electrons among…
Q: Determine the energy change in kJ/mol for the following two reactions: K(g)+H(g)→K+(g)+H−(g)…
A:
Q: From this determine the identity of the IR spect. include bonds, would love to compare to my own…
A: Given that, two IR spectra. Also, the structure of the compounds are given below We have to…
Q: 8. Complete the following guided multistep synthesis indicating all appropriate missing…
A:
Q: 4. Cooking vegetables or pasta in water and pouring it through a sieve to remove the water is using…
A: When cooking vegetables or pasta, it is common to use water to help cook the food. However, once the…
Q: Two analytical methods (potentiometry and amperometry) were used to determine the concentration of…
A: Q5)1)option 2 - 1.726 Q5)2) option 2 - Yes,they produce the similar results
Need help naming with the following.
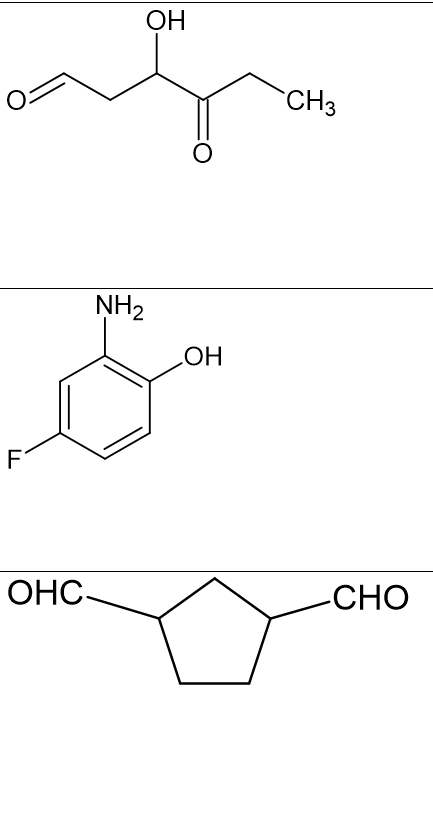
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


