Fig. 1 shows the possible effect of pH on recovery and impurity removal at the selection of a setpoint that allows for process control and enables a robust method. At what pH the recovery is at its highest and the impurities near their lowest. Also explains the intersection of two lines at pH 5.5. PROTEIN PURIFICATION METHODS 90 80 70 60 - Recovery (%) 50 -Impurity Concentration (ng/ml) 40 30 20 10 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 pH Recovery (%) or [Impurity] (ng/mL)
Fig. 1 shows the possible effect of pH on recovery and impurity removal at the selection of a setpoint that allows for process control and enables a robust method. At what pH the recovery is at its highest and the impurities near their lowest. Also explains the intersection of two lines at pH 5.5. PROTEIN PURIFICATION METHODS 90 80 70 60 - Recovery (%) 50 -Impurity Concentration (ng/ml) 40 30 20 10 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 pH Recovery (%) or [Impurity] (ng/mL)
Chapter28: Atomic Spectroscopy
Section: Chapter Questions
Problem 28.13QAP
Related questions
Question
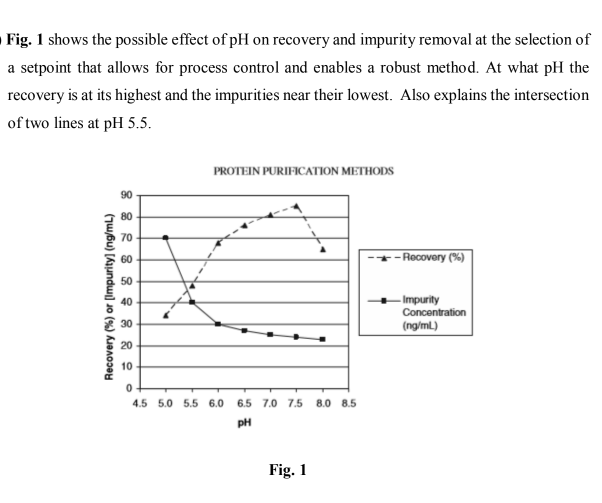
Transcribed Image Text:Fig. 1 shows the possible effect of pH on recovery and impurity removal at the selection of
a setpoint that allows for process control and enables a robust method. At what pH the
recovery is at its highest and the impurities near their lowest. Also explains the intersection
of two lines at pH 5.5.
PROTEIN PURIFICATION METHODS
90
80
70
– Recovery (%)
60
50
- Impurity
Concentration
40
30
(ng/mL)
20
10
4.5 5.0 5.5 6.0
6.5 7.0 7.5
8.0 8.5
PH
Fig. 1
(jwbu) (Aaundu) Jo (%) kanos
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

