Q: What is the NAME for the following ion? NO₂ H₂ H₂ A1₂ A₂ NO₂…
A:
Q: Which compound is best represented by the following IR spectrum? Major peaks (cm¹); Oa Ob Oc Od Oe…
A:
Q: A peptide bond (also called an amide bond) joins two amino acids together. What atoms are linked by…
A: Two amino acids are joined together via amide bond. The bond between carbon and nitrogen takes…
Q: What will be the freezing point of solution when 14.05 g of stearic acid (K₁ = 4.5 C/m) is mixed…
A: Given, Mass of stearic acid = 14.05 g = 0.01405 kg Freezing point depression constant of stearic…
Q: he overall reaction
A:
Q: The text states that the magnitude of ΔSvap tends to be similar for a wide variety of compounds.…
A: Entropy (S) :- Entropy is a measure of disorderness or randomness present in the system Higher is…
Q: Give the mechanism and choose correct option 0 130² A 애 @ heat 0대3 B 심 E C
A: In this question we have to tell the major organic product of the reaction.
Q: 1. Write a short paragraph that connects the following words: oxidation, reduction, anode, cathode,…
A: The words that we have to explain are, oxidation, reduction, anode, cathode and electrons.
Q: Draw a structural formula for 2-methyl-3-pentanol. • You do not have to consider stereochemistry. •…
A:
Q: What orbital(s) are the lone pairs of electrons on the O atom placed? - What orbital is the lone…
A: The given molecular structure is known as peptide where the carboxylic acid group of one amino acid…
Q: 5. If a first order isomerization reaction takes 24 hours for 20% of the compound present at the…
A:
Q: In the determination of the activation energy for the step-wise decomposition of iron (III)…
A:
Q: the shape of the model
A:
Q: = The decomposition of hypothetical molecule XY shown below has an activation energy Ea kj 100.- and…
A: Arrhenius equation says that k=Aexp(-Ea/RT) where, k=rate constant, A=pre-exponential…
Q: e pH of a 0.125 M solution of HIO3
A:
Q: 6. Complete the product(s) of the following substitution reactions. Include the stereochemistry.…
A: 6. a. OH group is converted into OTs using TsCl and py. b. 2-butanol is protonated first followed…
Q: Calculate the entropy change for the following process: H₂O (1, 298 K, 1 bar)→ H₂O (g, 398 K, 0.8…
A: The change in entropy can be calculated as follows
Q: How many moles of NH4Cl must be added to 1.0 L of a 0.050-M solution of NH3 to give a buffer…
A:
Q: Pls helo ASAP. Pls help in all asked questions.
A: The data given is,
Q: B₂H6(diborane) → 2BH3(borane) Plot this data to determine if the reaction follows first order…
A: Rate constant of first order reaction can be calculated from the equation K= (-1/t)ln {[A]/[A0]} On…
Q: None of these compounds will impact the solubility of AgBr. KOH (potassium hydroxide, a salt) Agl…
A:
Q: Explain the various factors that impact the solubility of substances in water
A: Solubility is the maximum amount of a substance that will dissolve in a given amount of solvent at a…
Q: over Short Answer 2. 3. Rank the following molecules from most acidic (1) to least acidic (4) (ень…
A:
Q: IMOLK Data, Trial 2 Ideal Gas Law-PV=nRT Molarity of HCI (M): Data, Trial 1 3.00M 3.00 M Volume of…
A: Hii there, As there are multiple question posted. we are answering first question. If you need…
Q: What is the IUPAC name for the following molecule? OH O1-ethyl-1-hydroxybutan-2-one…
A: In this question we have to tell the IUPAC name the molecule.
Q: Acetylene (C2H2) burns in oxygen (O2) to produce carbon dioxide (CO2) and water (H2O). 8.64 L of…
A: STP condition: T = 0°C P = 1atm Given that: Volume of acetylene = 8.64L
Q: pH Select 3 correct answer(s) B A B vol. added Solutions: strong base + strong acid Solutions:…
A: The given curve is shown for acid base titration . The type of solution are predicted from number of…
Q: Molecular Solid: A solid material primarily held in this phase by intermolecular forces causing…
A: Metallic solid - - > The metallic solid are the solids in which the metallic particles arey held…
Q: A chemist wants to determine the Ksp of BaC2O4 at T = 285.15 K. He prepared 0.10 L of saturated…
A: Given: Volume of aliquot taken in the titration flask = 0.01 L Concentration of HCl = 5.60 × 10-4 M…
Q: Which starting reagent would give the following as the major product? CI HCI none of these starting…
A: Note : Alkenes undergo rearrangements in the presence of acid because tertiary carbocation is more…
Q: In the reaction NH3 + H+ --> NH4+, which species acts as the Lewis acid? O NH3 O Ht O NHA 31 O
A: A lewis acid is an acid that accepts an electron pair.
Q: which of the following compounds itt have the highest molar Solubility im pare water ? KSP 4.96X10-9…
A:
Q: You need to make 50.0 mL of a 5.50 M solution of H,SO,. How many grams of H,SO, should you add to…
A:
Q: What is the relationship between pressure and temperature? as pressure decreases, temperature…
A: We would use the following diagram and check the relation between temperature and pressure.
Q: Draw a structural formula for 2-cyclopenten-1-ol. • Show stereochemistry only if given in the name.…
A: The structural formula for 2-cyclopenten-1-ol is:
Q: 7. Write the structures for A and B in the following synthesis scheme. Also, write a detail…
A: In this question we have to tell the product of the reaction and also its mechanism.
Q: 11 Iron for welding railway lines together is produced from the reaction of Po with aluminium. How…
A: 11. Given : Fe2O3 + 2Al → 2Fe + Al2O3 Mass of Al = 1.00 kg To calculate :- Mass of Fe2O3 (in…
Q: What is the strongest base on this list? i. 10 ii. 10₂ iii. 103 iv. 104
A: given molecule: IO-, IO2 -, IO3-, IO4- Base: Those species or molecules which have the tendency to…
Q: 5 3 6 18 : 20 10: For the following reaction, use the plots below and the information on them to…
A: The explanation is given below-
Q: 2. What is the molarity of hydroxide ions in a solution made by dissolving 0.2745 g Ca(OH)2 in…
A:
Q: The enthalpy changes of the following reactions can be measured: C₂H6 (g) + 7/2 O2 (g) → 2 CO₂ (g) +…
A: In this question we have to calculate the enthalpy change of the reaction.
Q: Give the mechanism and Correct regents? Br DHNO3, H₂SO4 Fe 2) BO2, 2) Alela Au OD Sell Alc13 2) BB₂…
A: The reaction given is,
Q: alt. Because the protein solubility depends on the total ion conce alts yielding doubly charged ions…
A: Mass of NaCl = 67.1 gram Moles of NaCl = mass/molar mass = 67.1 gram /58.44 g/mol = 1.148 mol NaCl…
Q: 2. Name the following molecules (a) (b) CH3C=c-up),ut=atz CH (c)/ 애 1000
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost…
Q: N'd TiPz 2 P;=P.V,Tz Pz= V2T. Al pha Particle Emission Beta Parti cle Emission Symbol Gamma Ray…
A:
Q: o determine mole ratios for
A:
Q: Shown below are three amino acids. What are their R/S configurations? HO NH₂ HO HO NH₂ NH₂ 4 S R S…
A: The atom with highest atomic number gets first priority. In case if you found same atom on moving…
Q: The following reaction can be carried out in two steps. Choose the correct reagents i) and ii) to…
A: In this question we have to tell the reagent used for the given organic transformation.
Q: Predicting Products of Chemical Reactions
A: "To get remaining sub-parts solved please repost the complete question and mention the sub-parts to…
Q: When ionic bromination of cyclohexene is carried out, the product is trans-1,2-dibromocyclohexane.…
A: When ionic bromination of cyclohexene is carried out, the product is trans-1,2-dibromocyclohexane.…
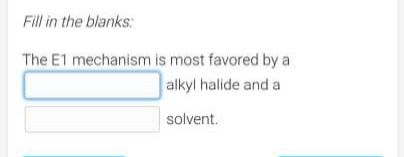
Step by step
Solved in 2 steps


