Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter24: Nuclear Chemistry
Section24.2: Radioactive Decay
Problem 6PP
Related questions
Question
100%

Transcribed Image Text:In the table below, there are descriptions of an experiment on samples of three different chemical elements. Decide whether the element is a metal or nonmetal,
if you can. If there is not enough information to decide, choose can't decide in the third column.
element
description
metal or nonmetal?
Element 1 is a moderately soft silvery-gray solid. A 5 cm × 5 cm square
of it, only 1 mm thick, is twisted using two pairs of pliers, giving it a
slight curve.
O metal
O nonmetal
O (can't decide)
1
Element 2 is a hard silvery-gray solid. A 10. g cube of it is set on a hot
plate. After 10 minutes, the temperature of the top of the cube has risen
by less than1 °C.
O metal
O nonmetal
O (can't decide)
Element 3 is a shiny silvery-gray solid. Wires are fastened to each side of
a 2 cm slab of it, and an ordinary household 9 V battery is hooked up so
that it can feed electricity through the slab to an LED. The LED glows
brightly.
O metal
O nonmetal
O (can't decide)
3
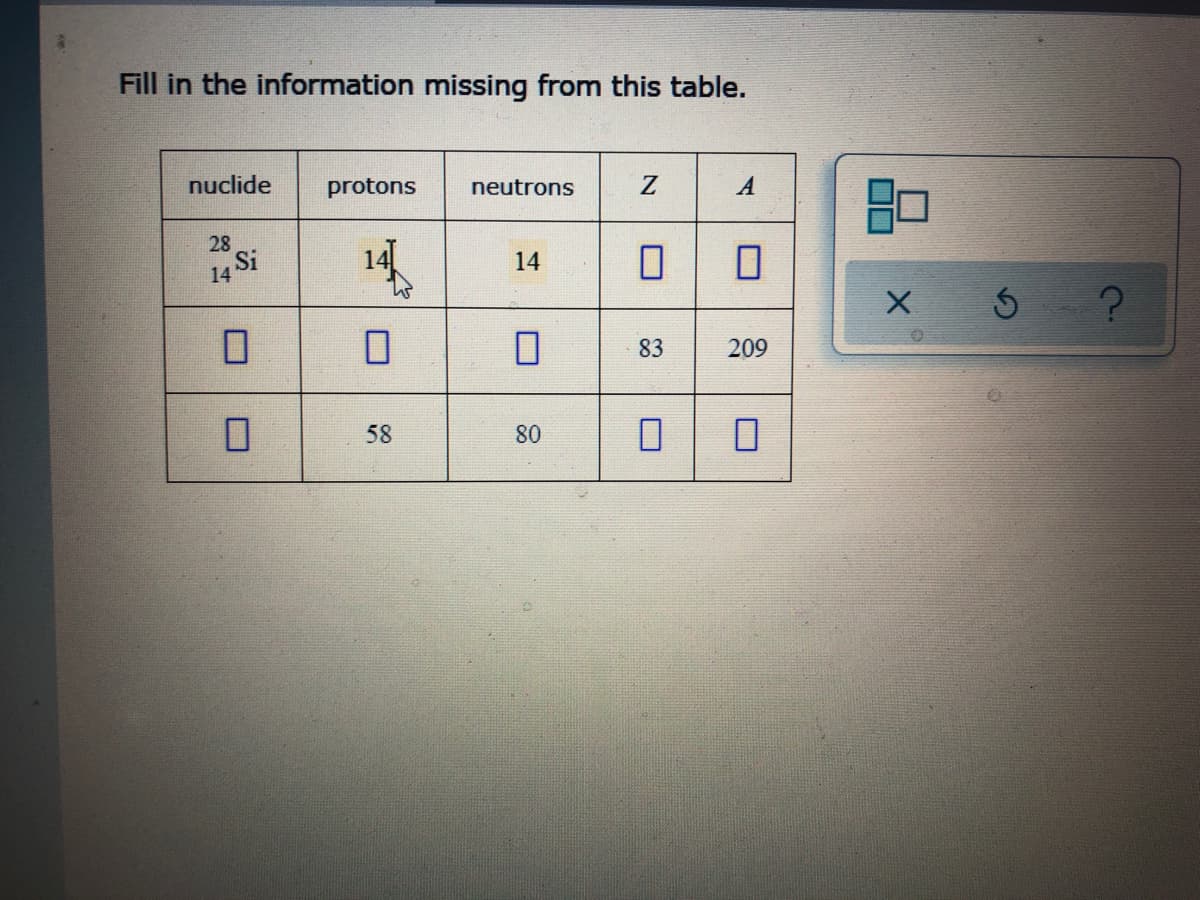
Transcribed Image Text:Fill in the information missing from this table.
nuclide
protons
neutrons
A
28
14 Si
14
83
209
58
80
口口
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning