Q: In the pH metric titration, calomel and glass electrodes are used for measuring. What are the other…
A:
Q: ( avestion) J - What mass 15.0 of NH3 of №₂ and 12.5g of H₂ is produced ??? are reacted,
A:
Q: A 220 mL sample of a 0.500 M NaCl solution with an initial pH of 7.00 is subjected to electrolysis.…
A:
Q: 2. NH₂ SO3 CH3 H₂SO4 ?
A: In this question we have to tell the product of the reaction.
Q: I don't understand why the second formula was multiplied by 2 and why the delta G of the 1st formula…
A: To explain why the second reaction is multiplied by 2 and the sign of delta G of the first reaction…
Q: What is the major product of the following reaction? В12, но OH Br OA В с A Br В 'Br с Br B -Br D E…
A: Organic chemistry is branch of chemistry in which organic reactant react to form organic products.
Q: What weight of soda ash must be taken for analysis so that by using 0.5000N HCl for titrating, (a)…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: (b) In the pH metric titration, calomel and glass electrodes are used for measuring. What are the…
A:
Q: 1.Write the nuclear equation to represent beta particle emission by Pu-241 2. Write a nuclear…
A: (1) When Beta particle emittion take place, then atomic number of plutonium, Pu-241 changed into…
Q: The radial eigenfunction for the quantum state of lowest energy for an electron with angular…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Quantitative analysis student Jon analyzed an ore sample for its lead content and obtained a mean of…
A: Given: obtained value- 4.75% certified value- 4.50% To find: (a) percent error (b) percent accuracy…
Q: Problem to be solved and to be checked using Boyle's Law equation: In a J-shaped tube filled with…
A:
Q: Which is the best statement below? O At STP, one mole of any ideal gas occupies 22.4 L (liters). O…
A: 1) at STP, one mole of any ideal gas occupies 22.4 l (liters)
Q: Consider sample of Sr(OH)2(aq) that was made by dissolving 0.239 g Sr(OH)2(s) in enough water to…
A:
Q: If the following half-cells were combined to make a battery, what would the standard cell potential…
A:
Q: ħ² The quantized energies of the rigid rotor are given by E, = 1/-/J (J -J(J + 1). If J = 2, what…
A: Rigid rotor is a mechanical model of rotating systems. Rigid rotor is a three dimensional rigid…
Q: Consider the reaction: 2 NO₂ (g) = N₂O4 (g) At equilibrium, the partial pressures of N₂O4 and NO2…
A: Equilibrium constant is defined as products concentrations divided by the reactant concentrations…
Q: A spherical-shaped balloon 18 meters in diameter is filled with helium to a pressure of 108 kPa, 28…
A: Given that - Diameter of balloon filled with helium gas = 18 meters Pressure = 108 kPa = 108/101…
Q: 6. CH3 HNO3. H₂SO4 ?
A: In this question we have to tell the product of the reaction
Q: 3. Use a table of standard thermodynamic values to predict the standard cell potential at 71 C:…
A: Given cell notation is : Cu(s) | CuSO4 || AgNO3 | Ag (s) Use the standard thermodynamics values to…
Q: Is the change in the enthalpy of reaction or the change in entropy of reaction more sensitive to…
A: So the entropy change for a reaction delta age was specifically defined to be equal to the heat at…
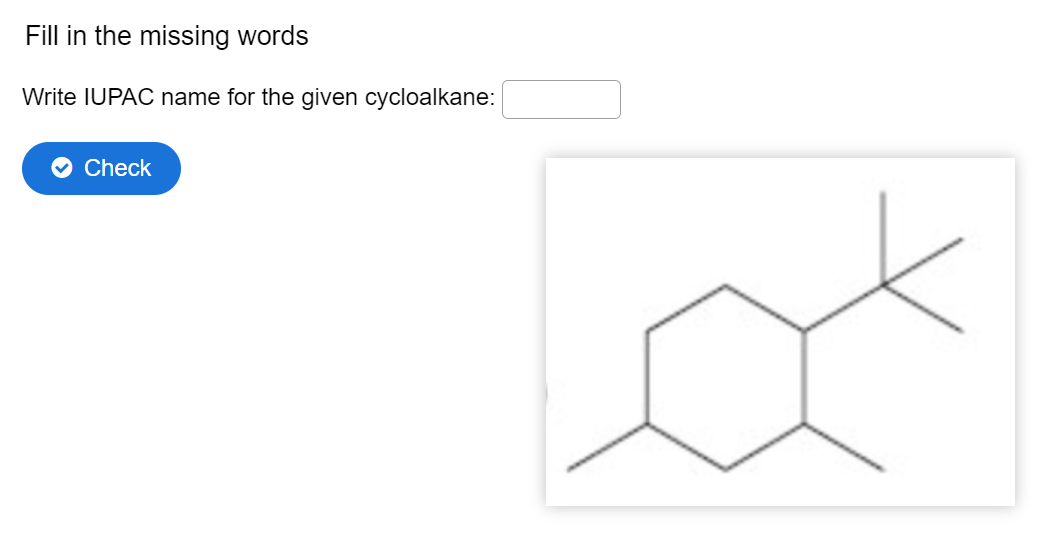
Q: Fill in the missing words What is the IUPAC name of the following compound? Check
A:
Q: What are the good titles for a community based projects?
A: There are many good titles as a community based projects some are listed below - - > Recycling…
Q: Experiment No 24: Determine the relative strength of two Acids by studying the hydrolysis of ester…
A: Given data:
Q: What is the angle between bonds
A: What is the angle between bonds in each of the following ?
Q: For given pair of compounds, identify which compound is the stronger base, and explain your choice:…
A: Electron pair donar is called Lewis base.. Electron pair acceptor is called Lewis acid...
Q: 30 mL of 0.25 mol L' HNO3(aq) is placed in a flask and titrated with 0.25 mol L NaOH(aq) solution.…
A: Given, HNO3 solution Concentration of HNO3 solution = 0.25 mol L-1 Volume of HNO3 solution = 30 mL =…
Q: 6. Draw the products formed upon Hofmann Elimination of the two isomeric amines below. (Excess CH3-I…
A: Detail mechanistic pathway is given below
Q: 10. How many molecules are in a 1.7 gram iron nail? 11. How many molecules are in a 0.35 gram piece…
A: (10) Given that - Mass of iron = 1.7 g From mole concept one of any Substance equal to - 1 mole…
Q: 2A → B+C The above reaction is run and found to follow first order kinetics with a rate constant of…
A:
Q: ACTIVITY 6.2.5 Supply the missing product in the reactions by drawing structure and giving its name…
A:
Q: The mobility of H+ ions is very high because of the small size and other reasons. The mobility of…
A: Hydrogen (H(+)) and hydroxide (OH(-)) ions in aqueous solution have anomalously large diffusion…
Q: A student burns a small snack food item and finds that it releases 40,800 J of energy. How many…
A: The conversion of quantity from one unit to another unit is known as unit conversion. During the…
Q: Calculate q , w U and H for the process during which 40.36g Ne has at 273K expands reversibly and…
A:
Q: One vial of COVID-19 vaccine, when thawed, contains 0.45 mL of solution. Before it is administered,…
A: Given: Concentration of NaCl = 0.154 M Volume of vaccine = 0.45 mL = 0.00045 L…
Q: Q6. Describe the particle motion.
A: Given: To describe: Particle in motion
Q: [B] [A] Time X у orward reaction is greater. everse reaction is greater. forward and reverse…
A: Rate of reaction is change in concentration of products and reactants in unit time interval.
Q: Why are aldehydes and ketones relatively weaker than alcohol in the context of hydrogen bond…
A: Alcohols generally contain one or more than one hydroxy, OH group, with general formula of R-OH.…
Q: Please sketch the titration curve of lysine. Then annotate X-axis, Y-axis, each pKa, PI and…
A: Here we are required to sketch the titration curve of lysine
Q: Determine the pH of a 0.20 M solution of RbNO2 at 25 °C. Kb for RbOH = 2.5 x 10¹ Ka for HNO₂ = 7.2 x…
A:
Q: Consider the following: 1) potassium, iodine 2) calcium, flourine 3) sulfur, chlorine Assume…
A: Ionic compounds are the compounds formed by transfer of electrons from one atom to another.…
Q: For items 10-12. The decomposition of NH4Cl (s) at a given temperature of 548 K, the Kp = 0.01072.…
A: Answer: In this question solid Ammonium chloride as been put into a vessel and its thermal…
Q: at a given temperature. What is the rate of production e same temperature? Enter your response in…
A: Given, Rate of consumption of A = -dA/dt = 0.378 M/s Form rate of reaction, Rate of reaction =…
Q: 1. Predict whether the following processes are spontaneous, are spon in the reverse or are in…
A: Spontaneous process occurs by its own while Non-spontaneous process required external source to…
Q: The molar enthalpy of fusion (∆Ho) for a given substance is 4.4 kJ/mol, and the molar entropy of…
A: The relationship between ∆G° , ∆H° and ∆S° is given by Gibbs-Hemholtz equation ∆G° = ∆H° - T∆S°…
Q: work If 5.50 L of water vapor at 50.2 °C and 0.121 atm reacts with an excess of iron to produce iron…
A:
Q: CEN O OCH3 1. LDA 2. CH3Br 1. LDA 2. CH3CH₂Br ? ?
A:
Q: If 25.0 mL of water are added to this system, indicate whether the following statements (Note the…
A: If we add water then concentration of acid and salt is changed due to volume increases. a) True b)…
Q: Calculate the AH for the reaction NO(g) + O(g) given the following information: NO₂(g) NO(g) + O3(g)…
A: Given aim equation is : Step equations are : Calculate the change in enthalpy of the aim equation…
Step by step
Solved in 2 steps with 2 images

- Give the IUPAC name of the following compounds. Answer the last two boxes only.What is the name of the IUPAC compound shown here? (see attatchement) First time you guys answered it was wrong, trying again. **** The answer 3-methylbut-1-ene you gave yesterday was WRONG . Please help with correct answer. This is what the question has as well: Alkanes are called saturated hydrocarbons because each carbon atom has the maximum number of hydrogen atoms. In contrast, alkenes and alkynes are unsaturated because they contain double or triple bonds that reduce the number of hydrogen atoms in the compound. When naming unsaturated hydrocarbons, a suffix reflects the type of multiple bond in the compound: ene is used for alkenes (double bond), and yne is used for alkynes (triple bond). For cyclic alkenes and alkynes, the ring is numbered such that the double or triple bond is between the first two carbon atoms. For straight-chain molecules, the numbering starts at one end of the chain, so the location of the double or triple bond must be specified in the name.Consider the synthesis scheme in Figure 12. Give the preferred IUPAC name of C? [Use lowercase letters. Do not use spaces if it is not required in the name.] *
- Questions in Cyclohexane vs Benzene Please identify their other names if any # Carbons Alkanes Complete Structure Cycloalkanes (if applicable) Shortcut Structure Complete Structure Cycloalkenes (if applicable) Shortcut Structure Alkynes Complete Structure Cycloalkynes (if applicable) Shortcut Structure 1 2 3 4 5 6 7 8The following names are all incorrect. Draw the structure represented by the incorrectname (or a consistent structure if the name is ambiguous), and give your drawing thecorrect name. ) 2-methylcyclopentene (d) 6-chlorocyclohexadieneClassify/ Name the cycloalkanes below:
- Is the straight chain alkane with molecular formula C5H12 expected to be a solid, a liquid or a gas at room temperature and pressure. Please type answer note write by hend.How To Name a Cycloalkane Using the IUPAC System ?What is the IUPAC name of the compound shown here? (view attatchement) **FIRST TIME YOU ANSWERE THIS WAS WRONG, please help with correct answer. The answer you gave me before was 1,4-dimethylcyclobut-1-ene but is was WRONG . This is what the paragraph also said next to the question "Alkanes are called saturated hydrocarbons because each carbon atom has the maximum number of hydrogen atoms. In contrast, alkenes and alkynes are unsaturated because they contain double or triple bonds that reduce the number of hydrogen atoms in the compound. When naming unsaturated hydrocarbons, a suffix reflects the type of multiple bond in the compound: ene is used for alkenes (double bond), and yne is used for alkynes (triple bond). For cyclic alkenes and alkynes, the ring is numbered such that the double or triple bond is between the first two carbon atoms. For straight-chain molecules, the numbering starts at one end of the chain, so the location of the double or triple bond must be…

