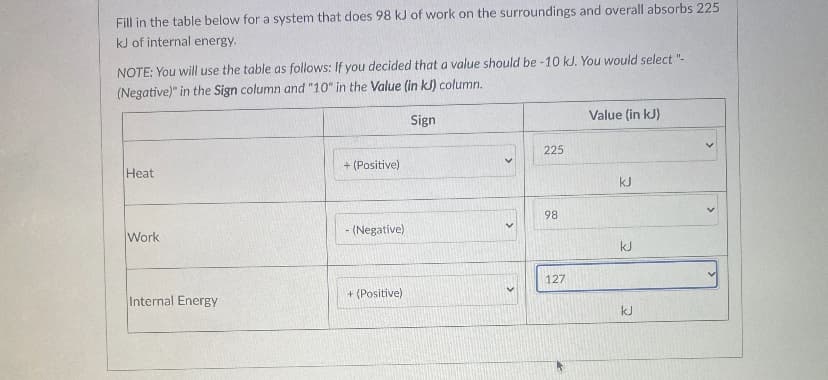
Fill in the table below for a system that does 98 kJ of work on the surroundings and overall absorbs 225 kJ of internal energy. NOTE: You will use the table as follows: If you decided that a value should be -10 kJ. You would select "- (Negative)" in the Sign column and "10" in the Value (in kJ) column. Value (in kJ) Sign 225 + (Positive) Heat kJ 98 Work - (Negative) kJ 127 + (Positive) Internal Energy kJ
Thermochemistry
Thermochemistry can be considered as a branch of thermodynamics that deals with the connections between warmth, work, and various types of energy, formed because of different synthetic and actual cycles. Thermochemistry describes the energy changes that occur as a result of reactions or chemical changes in a substance.
Exergonic Reaction
The term exergonic is derived from the Greek word in which ‘ergon’ means work and exergonic means ‘work outside’. Exergonic reactions releases work energy. Exergonic reactions are different from exothermic reactions, the one that releases only heat energy during the course of the reaction. So, exothermic reaction is one type of exergonic reaction. Exergonic reaction releases work energy in different forms like heat, light or sound. For example, a glow stick releases light making that an exergonic reaction and not an exothermic reaction since no heat is released. Even endothermic reactions at very high temperature are exergonic.
15) is this right?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps









