Five students set out to determine the average mass of a half-dollar coin. In performing their measurements, they obtained the following measurements: Student 1 Student 2 Student 3 Student 4 Student 5 Coin # Mass (g) 1 11.369 Coin # Mass (g) Coin # Mass (g) Coin # Mass (g) Coin # Mass (g) 1 11.378 1 11.357 1 11.401 1 11.330 2 11.433 2 11.363 2 11.264 2 11.352 2 11.424 3 11.380 3 11.419 3 11.274 3 11.278 3 11.368 4 11.333 4 11.439 11.323 4 11.399 4 11.386 4. 5 11.348 5 11.405 5 11.331 5 11.340 5 11.325 6 11.326 6 11.434 6 11.382 7 11.345 6 11.359 6 11.306 7 11.310 7 11.371 7 11.362 7 11.355 8 11.305 9 11.401 10 11.316 8 11.284 8 11.429 9 11.406 10 11.419 8 11.443 8. 11.369 9 11.369 10 11.271 6. 11.446 9 11.260 10 11.345 10 11.263 The average mass of a half-dollar coin is supposed to be 11.340 grams. Of the students, which one was the most precise in their measurements [Select ] Student 2 Student 3 Student 4 Student 1 Student 5 Next
Five students set out to determine the average mass of a half-dollar coin. In performing their measurements, they obtained the following measurements: Student 1 Student 2 Student 3 Student 4 Student 5 Coin # Mass (g) 1 11.369 Coin # Mass (g) Coin # Mass (g) Coin # Mass (g) Coin # Mass (g) 1 11.378 1 11.357 1 11.401 1 11.330 2 11.433 2 11.363 2 11.264 2 11.352 2 11.424 3 11.380 3 11.419 3 11.274 3 11.278 3 11.368 4 11.333 4 11.439 11.323 4 11.399 4 11.386 4. 5 11.348 5 11.405 5 11.331 5 11.340 5 11.325 6 11.326 6 11.434 6 11.382 7 11.345 6 11.359 6 11.306 7 11.310 7 11.371 7 11.362 7 11.355 8 11.305 9 11.401 10 11.316 8 11.284 8 11.429 9 11.406 10 11.419 8 11.443 8. 11.369 9 11.369 10 11.271 6. 11.446 9 11.260 10 11.345 10 11.263 The average mass of a half-dollar coin is supposed to be 11.340 grams. Of the students, which one was the most precise in their measurements [Select ] Student 2 Student 3 Student 4 Student 1 Student 5 Next
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter1: Basic Concepts Of Chemistry
Section: Chapter Questions
Problem 42GQ: Make a drawing, based on the kinetic-molecular theory and the ideas about atoms and molecules...
Related questions
Question
100%
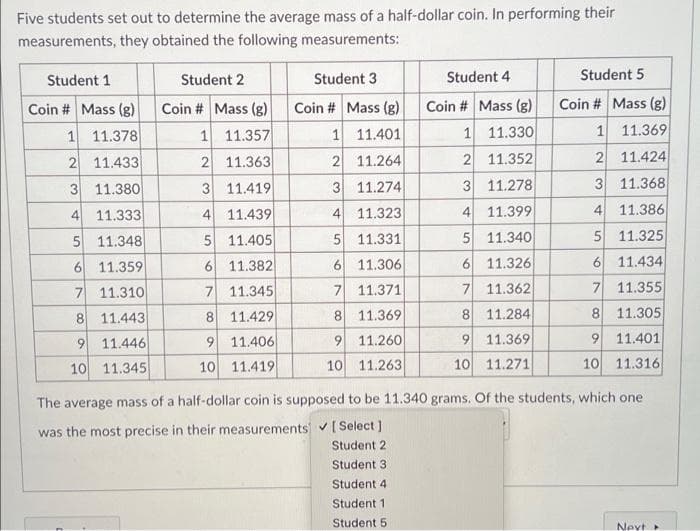
Transcribed Image Text:Five students set out to determine the average mass of a half-dollar coin. In performing their
measurements, they obtained the following measurements:
Student 1
Student 2
Student 3
Student 4
Student 5
Coin # Mass (g)
1 11.369
Coin # Mass (g)
Coin # Mass (g)
Coin # Mass (g)
Coin # Mass (g)
1 11.378
11.357
1 11.401
1 11.330
2 11.433
2 11.363
2 11.264
2 11.352
2 11.424
3 11.380
3 11.419
3 11.274
3 11.278
3 11.368
4 11.333
4 11.439
11.323
4 11.399
41
11.386
4.
5 11.348
5 11.405
5 11.331
5 11.340
5 11.325
6 11.434
7 11.355
6 11.359
6 11.382
6 11.306
6 11.326
7 11.310
7 11.345
7 11.371
7 11.362
8 11.443
8 11.429
8.
11.369
8 11.284
8 11.305
9 11.369
10 11.271
9 11.446
9 11.406
9 11.260
9 11.401
10 11.345
10 11.419
10 11.263
10 11.316
The average mass of a half-dollar coin is supposed to be 11.340 grams. Of the students, which one
was the most precise in their measurements [Select )
Student 2
Student 3
Student 4
Student 1
Student 5
Next
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning