- For a solution in which u = 8.0 X 10 2, calculate K, for %3D
Q: 5. Coomassie Brilliant Blue G-250, a dye used in the Bradford assay, has a maximum absorbance at 595…
A: Proteins can be separated based on their size, net charge and affinity towards a particular ligand.
Q: 2- How much solution (in ml) of silver nitrate at a molar concentration of 0,0540 mol/l is needed as…
A: The double replacement reactions occur when ionic chemicals in a solution react with each other.…
Q: -8 Given that the concentration of the hydroxy anion is 10 °M, what is the pH of the resulting…
A: The pH of the solution is a measure of hydrogen ion concentration. The pH of water is neutral…
Q: Compute the pI value of tyrosine. If the gel in an electrophoresis experiment is at pH 7, in which…
A: Step 1 Gel electrophoresis (Tiselius,1937) is a technique of separating biomolecules under an…
Q: You have prepared a BSA serial dilution (ug ml) to construct a protein standard curve, for which the…
A: The standard equation for calculating concentration is Beer Lambert's law, A= ebc, where A is…
Q: Calculate the unknown concentration of the PROTEIN E with an absorbance value of A678 given the…
A: Proteins have aromatic amino acids that absorb the maximum light of 280 nm wavelength. This…
Q: 6 Concanavalin (ConA) is a 25.5KDa protein with pI of 4.7 and optical absorbance (A 0.1% 289) of…
A: Proteins are polymers of amino acids with specific molecular weight and pI (isoelectric point, pH at…
Q: A solution of pH 5 contains--- -times more H + than the same amount of a solution at pH 8.
A:
Q: 2 a. You are trying to purify protein C from a mixture of proteins noted in the above Table. If you…
A: Chromatography is biochemical separation method for organic molecules or solutes of a compound…
Q: The order is for Moxam 5 gm IM every 8 hours. On hand is 10 gram of Moxam with the following…
A: Medication order is the terms used to define the directions for a drug needed to be given to the…
Q: Sketch the Hill plot for a system with negative cooperativity. Be sure to a) label your axes, and to…
A: A protein with multiple binding sites for a ligand can exhibit cooperativity. Cooperativity is the…
Q: Standard Curve 1.4 1.2 y = 12.235x R = 0.97758 0.8 0.6 0.4 0.2 0.02 0.04 0.06 0.08 0.1 0.12 BSA (mg)…
A: In the given question, we are given a straight line graph between Absorbance at 595 nm versus BSA…
Q: If you were presented with 2 L of a 2 M sucrose stock solution, how many grams of sugar would be in…
A: Molarity: The moles of a solute per liter of a solution is called as molarity (M).
Q: Protein concentration can readily be determined using the Beer-Lambert law: A = e l c where A =…
A: According to Beer-Lambert law: A = e l c where A = absorbance e = molar absorption coefficient…
Q: 4. Work through the following derivation: Starting with the equation of the line from the…
A: Lineweaver-Burk plot was postulated by Hans Lineweaver and Dean Burk in the year 1934. The equation…
Q: Given the following Hill Plot, what is the Kp for the last binding site? log Y 1-Y 2.5 0 -2.5 2.5…
A: Kd is dissociation constant and used to express the affinity of a protein for a ligand. A lower…
Q: 8. In an experiment, the concentration of the pesticide Lindane (L) was determined by GC analysis by…
A: Given in the question : The peak area of lindane standard solution = 44000 (0.01 M) So, 44000 gives…
Q: Zns) + Cu²+ce (aq) -> Zn²*(aq) + Cu(s) 1 A. Is Zn being oxidized or reduced? Why? Is Zn the anode or…
A: An electrochemical cell where electrolysis occurs is called an electrolytic cell. It is made up of…
Q: A protein sample of unknown concentration was placed in a cuvette with a path length of 1 cm and the…
A: The concentration of a protein is directly proportional to the amount of light it absorbs. The…
Q: At what concentration of S(expressed as a multiple of KM) will νo=0.95Vmax?
A: The enzyme catalyzed reaction is a biochemical reaction involving conversion of substrate molecules…
Q: Suppose you have a mixture of the following proteins protein A: pl = 3.5, mw = 35 kDa protein B: pl…
A: Cation exchange chromatography is defined as part of the ion-exchange chromatography. It is used to…
Q: The Ksp values of silver chromate Ag2CrO4 and silver iodate Ag(IO3) are given below.…
A: Given : Ag2CrO4 Ag(IO3) Ksp…
Q: concentration of each enzyme and with [X] = 1 µM. Which curve corresponds to which enzyme? Explain.
A: Given, the substrate concentration is [X] =1 μM Km of enzyme A = 2.0 μM Km of enzyme B = 0.5 μM
Q: There is a mixture of four proteins with their respective MW (in kD) in parentheses: A (60) B (100)…
A: Chromatography is a method for separating a mixture in the laboratory. The liquid is immersed in a…
Q: Two proteins of different molecular weight (mw) are mixed and need to be separated. When eluted…
A: Gel filtration chromatography/Size exclusion chromatography- It uses porous beads having defined…
Q: A 40 umol.l-1 solution of flavin mononucleotide (FMN) gave an absorbance of 0.8 at 470 nm using a 1…
A: Beer law states that the concentration of the solution is proportional to the amount of light it…
Q: 6. If the y-intercept of a Lineweaver-Burk plot = 1.91 (sec/millimole) and the slope = 75.3 L/sec,…
A: From the Lineweaver Burk plot the following information is given that is 1.91 sec/millimole and the…
Q: A cellophane bag containing a solution of iodine is placed in a beaker containing a starch solution…
A: Starch which is also known as amylum is defined as the polymeric carbohydrate, which consists of…
Q: -8 K, for HCO3 ` = 2.38 × 10 Calculate Ka for the conjugate acid ( H¿CO;) For the reaction :…
A: Conjugate acid-base pairs are those that differ from each other by a proton or H+ ion. For example,…
Q: If a data from an enzyme experiment is plotted as a Lineweaver - Burk plot, and the Vmax is 0.02…
A: Enzymes are macromolecular biological catalysts that accelerate chemical reactions. The molecules…
Q: Scenario: First, 0.1 mL of the unknown is mixed with 0.1 mL of distilled water. Then, 0.1 mL of the…
A: Beer-Lambert Law is applied in absorption spectrometry for biochemical analysis and measurement…
Q: 32. Which of the following is true? A. An exergonic reaction, energy is released and the delta G is…
A: Exergonic reactions produce free energy, hence the G value for that reaction will be negative. So…
Q: For the serial dilution, your stock solution must have a concentration of 3.5 mg/mL. How much…
A: RBC is the red blood cell and RBC suspension is normally used in serological testing and are usually…
Q: You assay 0.5 ml of 1/2000 diluted egg white for protein, as described in the practical schedule,…
A: Standard calibration curves are plotted by taking concentration of the solution in x axis and…
Q: BSA (mg/ml) Absorbancy 595nm 0.225 1 0.310 2 0.420 3 0.510 4 0.610 0.720 6 0.810 7 0.915 8 0.950…
A: The Bradford Assay is is a sensitive assay to detect and quantify protein in solution.
Q: What will happen when each of the following is added to a solution that contains 1.0 uM Y and 0.6…
A: Enzymes are protein molecules which increases the rate of chemical reaction without being costumed…
Q: What is the type of leaf outline in these figures?
A: Leaves are the vegetative organs of a plant that are specialised to perform photosynthesis mainly as…
Q: A solution contains 0.25 M Ni(NO3)2 and 0.25 M Cu(NO3)2. A. Can the metal ions be separated by…
A: Sodium carbonate is generally while dissolved in water, sodium carbonate forms carbonic acid and…
Q: ou resuspend 1 ml of of a purified chlorophyll sample in 10 ml of 80% acetone, and determine that…
A: The Beer-Lambert Law (also called Beer’s Law) is used to determine the concentration of unknown…
Q: Part B: Preparation of Standard Graph & Determination of Concentration of Original Unknown Indigo…
A: Standard curves are plotted by using the light absorbance value and the solution concentration.…
Q: if you perform a 1:5 dilution of a 1 M solution , how many parts of the 1 M solution do you use, and…
A: Dilution factor describes ratio of aliquot volume to final volume. It is a notation often used in…
Q: Given that the molar extinction coefficient of lysozyme is 37200 Abs M-1cm-1, calculate the…
A: According to the Lambert Beer's law: Absorbance = (extinction coefficient) x (concentration) x (path…
Q: choices for question 1: H LOH NH2 INH3* HO H 'N' `NH2 `NH2 a b H LOH LOH NH2 INH2 `NH2 `NH2 H A H A…
A: Hi! Since you have posted multiple questions, we will answer the first one for you because we are…
Q: 10 Determine the Hill coefficient for the following binding data using a linear method. [L] in…
A: The Hill plot is a graph graph which enables us to check whether the protein in question exhibits…
Q: -6 Draw the Fischer projection formula for each of the following molecules. CI b. ОН а. НО CH3 Cl…
A: Fischer projection is a method of representing the three dimensional structures of molecules on a…
Q: With the constants you have for each objective, compute for the sizes of the given cells below.…
A: Given Image A seen under scanner Image B seen under low power objective Image C seen under high…
Q: Concanavalin (ConA) is a 25.5KDa protein with pl of 4.7 and optical absorbance (A 0.1% 289) of 1.14.…
A: Proteins are polymers of amino acids with specific molecular weight and pI (isoelectric point, pH at…
Q: Calculate the DNA concentration in µg/mL from the following information: a. Absorbance reading…
A: Deoxyribonucleic acid or DNA is the hereditary material that is present in all organisms. DNA…
Q: A two-dimensional gel electrophoresis of a protein preparation revealed the presence of five…
A: Size exclusion chromatography is another name for gel filtration chromatography. Gel filtration…
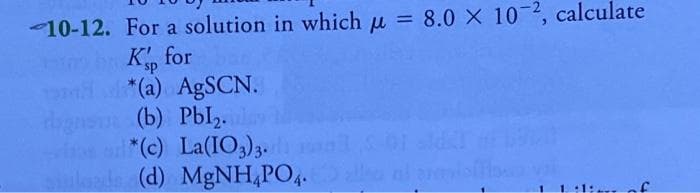
Step by step
Solved in 2 steps with 2 images

- Why are hydrogen bonds and van der Waals interactions necessary for cells?20. According to the package insert, adding 2.5 mL of sterile water to a 1 g cefazolin vial yields a final volume of 3 mL. What volume of this solution would you need to obtain 200 mg of cefazolin?##6.question. a drug is order 100mg/m2 po BID. How many milliliters will a patient receive if her BSA is 1.80m2 and concentration of solutions is 100mg/5ml?
- 10, 11 please answer all give the significance/role/effect of the reagent/condition in the isolation or analysis of a biomolecule. limit answers to two short sentencesDextrose 5% in water solution is an example of hypertonic solution. True False?15. How much powder and water to make a 0.1 concentration of resazurin and please explain the reaction from resazurin to resorufin?
- 16. The lipopolysaccharide of Gram-negative bacteria is composed of the following EXCEPT: Polysaccharide Lipoteichoic acid Phospholipid O-Polysaccharide6 After running an Ion exchange chromotograpraphy column and using cation exchange, will the elution contain any activity?Why is it impossible for humans to digest food that contains cellulose?
- Why do ions have a difficult time getting through plasma membranes despite their small size?When you do a load of laundry, why do you not just drop a bar of soap Into the washing machine? In other words, why is laundry detergent sold as a liquid or powder?Plasma contains more sodium than chloride. How can this be if individual ions of sodium and chloride exactly balance each other out, and plasma is electrically neutral?



