Q: Use curved arrow notation to show how the first resonance structure can be converted to the second.
A:
Q: Refer to the structure of imidazolium ion in the preceding equation and write a second resonance…
A: The movement of pi electrons within the molecule to give rise different contributing structure is…
Q: :0: H. H. H. C `N' N. нн H H B
A: The resonance structures of a molecule represent the structure with the same molecular formula and…
Q: Use a numbered list to indicate each step. (Write one sentence for each arrow that is drawn for each…
A:
Q: draw out h and show if there is any strain in each molecule!then analyze which one is more…
A: Following structure is more favourable due to the steric strain.
Q: For each set of molecules below circle the sets that represent valid resonance forms Be sure to show…
A: Welcome to bartleby ! We have to tell the correct resonating structures among all given molecules
Q: In this second exercise, you will learn how to add other bond types to your structure, such as…
A: The compound given is,
Q: CH3 .N. H3C- H
A: Rules for drawing resonance structure Only the electrons move, the nuclei of atoms never move and…
Q: Use curved arrow notation to show how the first resonance structure can be converted to the second ?
A:
Q: O2- is an ion that can from a one double bond while NO2+ will have 2. (True/False)
A: Interpretation : We have to tell which statement is true.
Q: Benzene is the simplest member of a whole class of compounds called aromatic hydrocarbons. benzene…
A: Given aromatic compound is benzene. Its structure is shown below:
Q: Explain why the next molecule is aromatic.
A:
Q: Circle the most reactive molecule with water and put an X through the least reactive. CI
A: The molecules are acid derivatives:
Q: 16. Draw all of the resonance structures for the compound shown. Circle the resonance structure that…
A: Various resonance structures are formed by delocalisation of π-electrons which are in conjugation.…
Q: How many allylic hydrogens (total number - not type!) are present in the following molecule?
A: When a compound contains atleast one double bond between two Carbon atoms, then the carbons attached…
Q: Determine the total number of resonance structures. Include the initial structure as one structure…
A:
Q: we can say that the next pair of compounds are:
A: The problem based on stereoisomerism. We convert wedge dash formula in Fischer projection and then…
Q: Check the box next to each molecule on the right that has the shape of the model molecule on the…
A: According to VSEPR theory, geometry of molecule depends upon total electron groups present on…
Q: draw the arrow formalism on the 1st structure (top) that accounts for the 2nd resonance structure…
A: The arrow formalism on the 1st structure (top) that accounts for the 2nd resonance structure…
Q: Please draw a more stable resonance structure for the following molecule. Use a curved arrow to show…
A:
Q: [References Follow the flow of electrons indicated by the curved arrows in the following polar…
A: In this reaction oh- attack on hydrogen. Hydrogen leaves it's electron and formation of double bond…
Q: Could we cut just one bond in the "starting" molecule shown in the drawing area below to create this…
A: This is possible to get the product by cutting one bond .
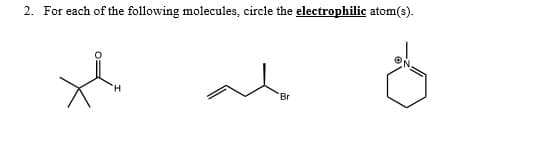
Q: For each of the following molecules, circle the electrophilic atom(s). Br
A: Electrophilic atom species are electron seeking species i.e. they are electron deficient. 1)…
Q: π
A: In first Compound CO : it's have triple bond so this Compound contain one sigma and two pi bond.…
Q: Is the molecule below conjugated? If so, circle the atoms that are part of the conjugation. Also…
A: In the given organic compound there are two lone pairs on Oxygen atoms. Positive charge and…
Q: How many σ and π bonds are in the following molecules? And what these σ and π bonds mean?
A: Since you have posted a question with multiple subparts, we will solve only first three subparts for…
Q: A. Determine whether the curved arrow(s) shown below generate a valid or invalid resonance…
A:
Q: Consider molecules A–D. Does the arrow pushing in each structure lead to an acceptable resonance…
A:
Q: Draw the resonance contributors for the species/molecules shown in the boxes below. Then, indicate…
A: Resonance is a hypothetical concept which is used to represent a molecule in more than one form via…
Q: Draw the most likely fragments for the molecular ions at: 43, 95, and 123 in figure 7. Localize any…
A: Possible fragments at 43 and 95 are shown below.
Q: Draw the lowest energy chair for the following molecules HO,
A:
Q: when a force is app When force is rema Rubber band has a It satisfies all other nly nd II and III…
A: Rubber band is considered as a solid because stretching the rubber, its shape is changes ,after it…
Q: the reaction below: a. How many non-polar and polar bonds are in the reactants? b. How many…
A: Polar Bonds:- These bonds are present between atoms which have different electronegativity. Non…
Q: 1. Draw the best possible resonance contributor. Draw curved arrows to show electron movement.
A: Resonance structure : Delocalization of π electron density in adjacent conjugated system is called…
Q: Which bond in attached pair has the higher bond dissociation energy?
A: Since, the strength of bond is proportional to the number of bonds between the elements i.e triple…
Q: What reactant would transform the molecule from the top left to the top right. Given the same…
A: In this question, we will findout the Reagent for above transformation and for below Reaction…
Q: Please give me idea why resonance is more reactive. I thought If resonance exists, it delocalizes…
A: As we know leaving group ability is increases with less hindrance . So i.e. CH3>1°>2°>3°
Q: Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas…
A: A. At first OH oxygen gets protonated to give better leaving group-OH2^+ . Then it undergo…
Q: For each of the following molecules, circle the electrophilic atom(s). TH. Br
A: Electrophile are electron loving species that means positive charged species. Example: CH3+.
Q: H. エ
A: We have to draw two resonance contributor of given structure. And predict the stability.
Q: is this z or E?? If it is E. please in details explain why cycloproyl is greater than OH group??
A:
Q: Draw all missing reactants andlor products in the appropriate boxes by placing atoms on the canvas…
A:
Q: Consider the molecule below (named squaric acid, for obvious reasons). Draw the product obtained…
A: Given Squiric acid with 2 equivalents of base Find product
Q: Which molecule, A or B, has greater angle strain? Explain. A
A: Angle ring strain present in the compound when the bond angle between two bonds differs from the…
Q: Question Consider molecules A–D. Does the arrow pushing in each structure lead to an acceptable…
A:
Q: Explain. Why methyl radical is trigonal planar while trifluoromethyl radical is pyramidal? Provide…
A: Methyl free radical Hybridisation is Sp3 because it has 3 bond pairs and one unpaired electron…
Q: a Draw the missing resonance structure on the right to account for the curved arrows on the left…
A: In this question we have to tell the resonance structure of the given structure.
Q: On a piece of paper or electronic device, draw the most and the least stable Newman projections of…
A:
Q: The molecule shown here has quite a large dipole, as indicated in its electrostatic potential map.…
A: Why the given molecule has quite a large dipole , as indicated in its electrostatic potential map…
Please answer all part of the question...thank you
Step by step
Solved in 2 steps with 1 images

- Briefly explain why the I end of the molecule is more electrophilic than the Cl end.For each structure you drew in the answer to the previous question, classify it as a strong acid,strong base, weak acid, or weak base.See attached image. For this molecule draw the 2 best resonance structures in the boxes. Circle the best.
- Add curved arrows to the following polar reactions to indicate the flow of electrons in each:Please help with row 3 highlighted in yellow (column 1 and column 2); the instructions for the specific column in row 3 are above row 1: Follow the instructions in each column. Hint for the last column: draw it with a wedge and again with adash – which gives the correct configuration?Click on all structures that are identical to the first (leftmost).
- Which are aromatic species? Please circle correct species. (assume planarity unless otherwise noted)I'm looking for an alkene with a single methyl group and an anti markovnikov Br at the last carbon and the markovnikov with Br on the bottom. The image is attached is just to illustrate positioning of the Br, and not an actual alkene

