Q: Show the curved arrow formalism in order to arrive in the major final product
A: This reaction proceeds via an electrophilic addition mechanism. This reaction proceeds in a…
Q: OR A ZI
A: We have to choose which one is more basic.
Q: How many carbon atoms in this remdesivir are considered chiral? Please circle each one.
A: The given compound: The chiral carbon atoms in the given compound have to be determined.
Q: s sugar X a reducing or a non-reducing sugar? Reducing, because one/both of the rings can open…
A: The carbohydrate containing free aldehyde or ketone groups in its molecular structure is said to be…
Q: Determine whether each of the following molecules has a plane of symmetry and draw it in
A: A plane of symmetry is the imaginary plane that can bisect a molecule given, into two equal halves…
Q: Terminal alkyne is considered a more acidic compound because it has a lower s character. True or…
A: In this question, we will discuss about this Statement is true or false. You can see details…
Q: Convert attached three-dimensional model to a Newman projection aroundthe indicated bond ?
A: INTRODUCTION: Newman projection is defined as it visualizes the conformation of a chemical bond from…
Q: NH2
A:
Q: How does this chair conformer make this other this in a synthesis reaction?
A:
Q: Draw the following molecule in a chair conformation. ok
A: Cyclohexane exist in two non-planar forms chair and boat out of these two forms chair form is highly…
Q: a) Which of the following compounds is (are) capable of H-bonding to itself? Circle all that apply.…
A: Given compounds are: In the above given compounds, A is a ketone, B is an alkene, C is alcohol, D…
Q: Draw both possible chair conformations for the and identity which is lower in following molecule…
A: Lower energy chair conformation can be figure out by the bulky group interaction.
Q: regiochemistry and/or configuration
A: The given blank has to be filled correct regiochemistry and/or configuration
Q: In the following species, circle the most acidic hydrogen
A: The most acidic hydrogen is the one which removes to form a stabilized species.
Q: Circle the most reactive molecule with water and put an X through the least reactive. CI
A: The molecules are acid derivatives:
Q: Q5. Determine whether each of the following curved arrow(s) are acceptable. For those that are, draw…
A:
Q: Q5. Determine whether each of the following curved arrow(s) are acceptable. For those that are, draw…
A: The answer to the following questions is- Arrows are used to indicate movement of electrons in the…
Q: Label the most stable and the second most stable carbocation. HOHO
A:
Q: How many allylic hydrogens (total number - not type!) are present in the following molecule?
A: When a compound contains atleast one double bond between two Carbon atoms, then the carbons attached…
Q: Give a clear handwritten answer with explanation..given below a energy diagram...what represent A in…
A:
Q: O Add the missing curved arrow notation.
A:
Q: In each case below, draw a Newman projection as viewed from the angle indicated:
A:
Q: Q. How many different C environment for molecule shown?
A: For different environment carbon connectivity sho
Q: H.C. CH3 CH3 CH3
A:
Q: B. Draw the correct curved arrow(s) needed to generate the resonance structures shown below and…
A:
Q: For each of the following molecules, circle the electrophilic atom(s). Br
A: Electrophilic atom species are electron seeking species i.e. they are electron deficient. 1)…
Q: Which of the following is the most accurate depiction of a structure at the point shown on the…
A:
Q: Which one is more stable S2-, S4-, S6-?
A: The given ions of Sulphur are S2-, S4-, and S6-.
Q: What structure has the lowest energy?
A: For trans-1,3-substituted cyclohexane, the substituents should be in (a,e) or (e,a) positions to…
Q: Draw the lowest energy Newman projection of the following molecule sighting down the C3-C4 bond.
A: Given that : We have to draw the lowest energy Newman projection of the following molecule sighting…
Q: Circle the following molecules that have the R configuration
A: R indicates that a clockwise circular arrow that goes from higher priority to lower priority crosses…
Q: For the following molecule draw both possible chair conformers and predict the direction of the…
A: Chair conformation: It is more stable conformation rather than other conformations .Angle strain and…
Q: Which C-H bond has the lowest BDE?
A: Given : structure of molecules This is based on stability of radical.
Q: он
A: When molecules with same chemical differ from one another in orientation of atoms in surrounding…
Q: Which of the molecules and ions given in diagram are aromatic according to the Hückel criteria?…
A: The conditions for a compound to be aromatic are: 1) It should be closed ring structure. 2) It…
Q: Add the hydrogens to the following structure and circle its most acidic hydrogen(s).
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided resonance structures,…
A: The arrows pushing mechanism can be shown as:
Q: Draw the orbital interaction that illustrates the hyperconjugation that takes place in propyne.
A: The hyper conjugation is the delocalization of the δ electron from the Cα-H bond to an adjacent…
Q: For each of the following pairs of molecules, which do you think should have the more acidic a…
A: A strong acid is an acid that dissociates to make protons available for reaction. Acid mainly…
Q: For each of the following molecules, circle the electrophilic atom(s). H. Br
A: Electrophilic atom species are electron-seeking species i.e. they are electron deficient. 1. There…
Q: Please do ring flip of this and determnine both if trans or cis. Thanks
A: Ring flipping in cyclohaxane is the conversion of one chair conformation into another chair…
Q: Which of the following species are aromatic or anti-aromatic (assuming n-systems are fully planar)?…
A: Hello, if multiple questions are present , first question is allowed to answer, hence post…
Q: Can someone explain how to convert a condensed formula to a bond. I get confused about atoms that…
A: In the bond line angle formula, each covalent bond between carbon-carbon or carbon-heteroatom is…
Q: For each of the following molecules, circle the electrophilic atom(s). TH. Br
A: Electrophile are electron loving species that means positive charged species. Example: CH3+.
Q: H. エ
A: We have to draw two resonance contributor of given structure. And predict the stability.
Q: HO, H+ How do you araw the deailcd mechanism with arou formalisms?
A:
Q: Please circle the most acidic hydrogen(s) in the molecule below
A: Alcohol are more acidic then alkynes .
Q: Shown on the left is a cationic intermediate for the electrophilic addition of chlorine to the para…
A: Since the carbocation will be more stable on the carbon attached to the O as it will make back bond…
Q: Draw an arrow on the carbocation in the box on the left to show how it rearranges to the carbocation…
A: As we know that ; The Tertiary carbocation is more stable than the secondary carbocation and the…
Q: following compounds. Also, draw the resonance hybrid and indicate the major contributor in the drawn…
A:
Q: Properly, draw the Newman structure for a staggered and anti depiction of 2,3-dimethylbutane. How…
A: Gauche conformation is one in which the dihedral angle between the two atom or group is 60° The…
Q: D. Draw the products of the second step of the mechanism. Be sure to add lone pairs and charges,…
A: Given information Reactants are Butyl Chloride and Iron(III)chloride In Friedel Craft alkylation an…
Q: Which molecule, A or B, has greater angle strain? Explain. A
A: Angle ring strain present in the compound when the bond angle between two bonds differs from the…
Q: Which aonance structure of the anion in the box? Y
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: a Draw the missing resonance structure on the right to account for the curved arrows on the left…
A: In this question we have to tell the resonance structure of the given structure.
Q: Label each attached compound as cis or trans. Then draw the second chair conformation.
A: This given compound is 1,2 axial/equatorial trans dichlorocyclohexane. Axial means above the plane…
Q: Which of the molecules and ions given in diagram are aromatic according to the Hückel criteria?…
A:
Q: Although the hypothetical molecule shown here has alternating single and double bonds, those double…
A: Conjugation: Conjugation is defined by saying that a group of molecules having alternate double…
Q: For the same molecule, which of the images corresponds to the correct Newman projection along the…
A:
8
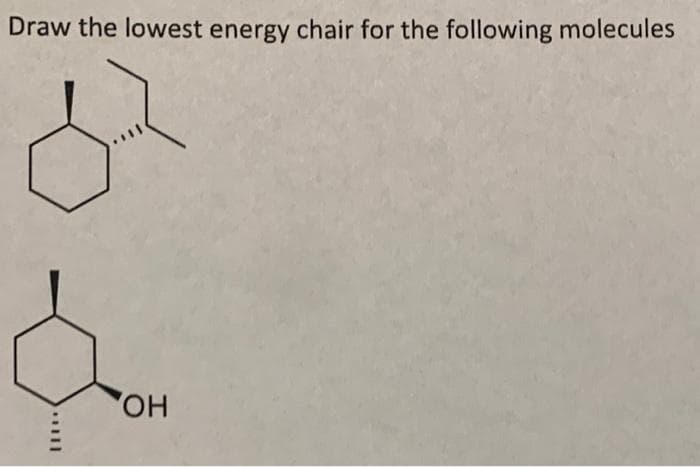
Step by step
Solved in 2 steps with 2 images

- ! ( these are very easy please do all , if u hv plan to do only 3 plz skip, lets order do ) give typed ansPlease determine the Identity using the IR spect and the given, show bonds, would love to compareSolve correctly please, both ques answer need with full explanation for Upvote. (Gpt/ai wrong answer not allowed)

