Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter23: Amines
Section: Chapter Questions
Problem 23.26P: The pKa, of the conjugate acid of morpholine is 8.33. (a) Calculate the ratio of morpholine to...
Related questions
Question
100%
For each of the maxima given in the UV spectra of Figures 8.51-8.53, determine the value of A and of log € (epsilon).
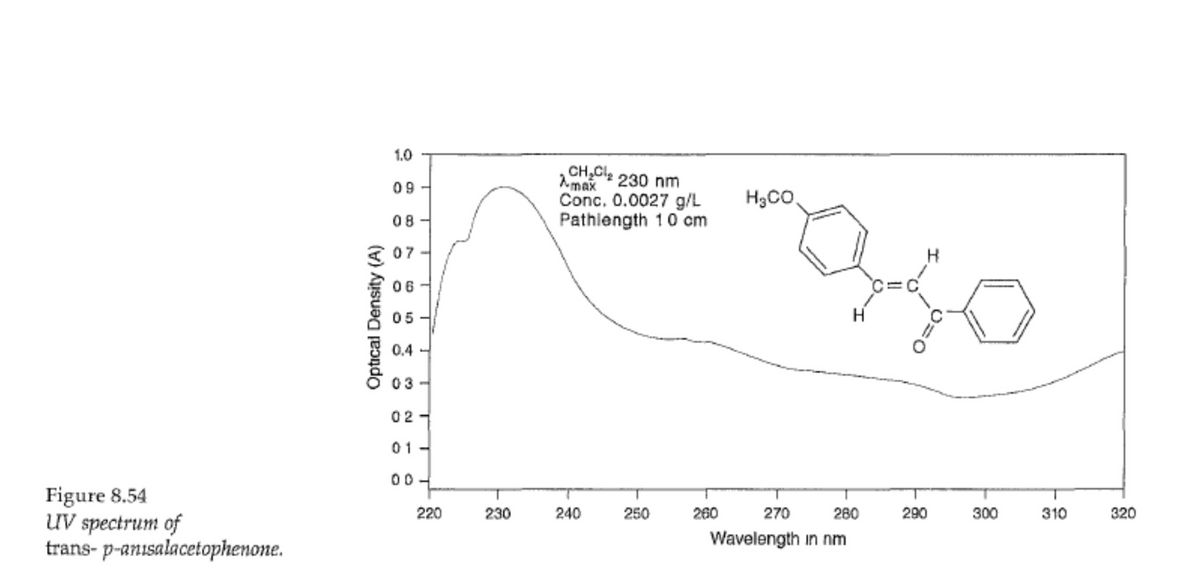
Transcribed Image Text:Figure 8.54
UV spectrum of
trans-p-anısalacetophenone.
Optical Density (A)
1.0
09
08
07
06
a
ເດ
0.4
03-
02-
01-
00
220
230
CH₂Cl₂ 230 nm
max
Conc, 0.0027 g/L
Pathlength 10 cm
240
250
HgCO.
260
aso
C=C
280
270
Wavelength in nm
290
300
310
320
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning