
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 23, Problem 23.26P
The pKa, of the conjugate acid of morpholine is 8.33.
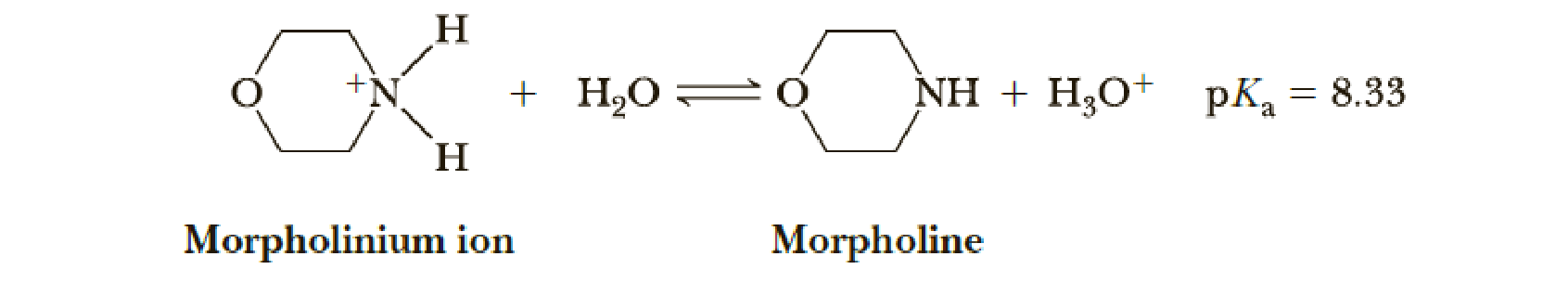
- (a) Calculate the ratio of morpholine to morpholinium ion in aqueous solution at pH 7.0.
- (b) At what pH are the concentrations of morpholine and morpholinium ion equal?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Write the equilibrium-constant expressions and obtainnumerical values for each constant in.
(a) the basic dissociation of aniline, C6H5NH2.
(b) the acidic dissociation of hypochlorous acid,HClO.
(c) the acidic dissociation of methyl ammoniumhydrochloride, CH3NH3Cl.
(d) the basic dissociation of NaNO2.
(e) the dissociation of H3AsO3to H3O+and AsO33-.
(f) the reaction of C2O42-with H2O to give H2C2O4and OH-.
show solution
Pyridine forms a stronger Lewis acid-base complex with SO3 than SO2. However, pyridine forms a weaker complex with SF6 than with SF4. Explain the difference.
Explain the observed difference in the pKa values of the conjugate acids of amines A and B.
Chapter 23 Solutions
Organic Chemistry
Ch. 23.1 - Prob. 23.1PCh. 23.2 - Prob. 23.2PCh. 23.2 - Prob. 23.3PCh. 23.2 - Prob. 23.4PCh. 23.5 - Prob. 23.5PCh. 23.5 - Prob. AQCh. 23.5 - What is the hybridization of the nitrogen in...Ch. 23.5 - Prob. CQCh. 23.5 - The pKas of the conjugate acids of aniline and...Ch. 23.5 - Prob. EQ
Ch. 23.5 - Prob. FQCh. 23.5 - Prob. GQCh. 23.5 - Select the stronger acid from each pair of...Ch. 23.6 - Prob. 23.7PCh. 23.6 - Prob. 23.8PCh. 23.6 - Prob. 23.9PCh. 23.7 - Prob. 23.10PCh. 23.8 - Prob. 23.11PCh. 23.8 - Prob. 23.12PCh. 23.8 - Prob. 23.13PCh. 23.9 - Prob. 23.14PCh. 23.10 - In Example 23.15, you considered the product of...Ch. 23 - Prob. 23.16PCh. 23 - Prob. 23.17PCh. 23 - Prob. 23.18PCh. 23 - Prob. 23.19PCh. 23 - Prob. 23.20PCh. 23 - Prob. 23.21PCh. 23 - Prob. 23.22PCh. 23 - Account for the formation of the base peaks in...Ch. 23 - Prob. 23.24PCh. 23 - Select the stronger base from each pair of...Ch. 23 - The pKa, of the conjugate acid of morpholine is...Ch. 23 - Which of the two nitrogens in pyridoxamine (a form...Ch. 23 - Prob. 23.28PCh. 23 - Prob. 23.29PCh. 23 - Prob. 23.30PCh. 23 - Prob. 23.31PCh. 23 - Suppose you have a mixture of these three...Ch. 23 - Prob. 23.33PCh. 23 - Prob. 23.34PCh. 23 - Prob. 23.35PCh. 23 - Prob. 23.36PCh. 23 - Prob. 23.37PCh. 23 - (S)-Glutamic acid is one of the 20 amino acid...Ch. 23 - Prob. 23.39PCh. 23 - Propose a structural formula for the compound...Ch. 23 - Prob. 23.41PCh. 23 - The pyrolysis of acetic esters to give an alkene...Ch. 23 - Propose steps for the following conversions using...Ch. 23 - Show how to bring about each step in this...Ch. 23 - Show how to bring about each step in the following...Ch. 23 - Prob. 23.48PCh. 23 - Prob. 23.49PCh. 23 - Methylparaben is used as a preservative in foods,...Ch. 23 - Prob. 23.51PCh. 23 - Prob. 23.52PCh. 23 - Propose a synthesis for the systemic agricultural...Ch. 23 - Prob. 23.54PCh. 23 - Several diamines are building blocks for the...Ch. 23 - Prob. 23.56PCh. 23 - Prob. 23.57PCh. 23 - Prob. 23.58PCh. 23 - Prob. 23.59PCh. 23 - Following is a retrosynthesis for the coronary...Ch. 23 - Prob. 23.61PCh. 23 - Prob. 23.62PCh. 23 - Given this retrosynthetic analysis, propose a...Ch. 23 - Prob. 23.64PCh. 23 - Following is a series of anorexics (appetite...Ch. 23 - Prob. 23.66PCh. 23 - Prob. 23.67PCh. 23 - Show how the synthetic scheme developed in Problem...Ch. 23 - Prob. 23.69PCh. 23 - Prob. 23.70PCh. 23 - Prob. 23.71PCh. 23 - Prob. 23.72P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give a suitable chemical reaction to distinguish between a primary and a secondary amine. Suggest one way of Increasing the basicity of an amine and give a specific example of its application.arrow_forwardThe compound acetophenone has a very similar molar mass to that of benzoic acid and benzamide. However, acetophenone has a much lower m.p. (20 °C) than both such that, by contrast, it is a liquid at room temperature. By considering intermolecular forces and comparing functional group structure, account for this big difference in physical properties.arrow_forwardRationalize the electronic effects responsible for the basicity of aminesarrow_forward
- Complete the following acid-base reactions and predict the direction of equilibrium (to the right or to the left) for each. Justify your prediction by citing values of pKa for the stronger and weaker acid in each equilibrium. For values of acid ionization constants, consult Table 23.2 (Acid Strengths, pKa, of the Conjugate Acids of Selected Amines) and Appendix 2 (Acid Ionization Constants for the Major Classes of Organic Acids). Where no ionization constants are given, make the best estimate from the information given in the reference tables and sections.arrow_forwardRank each of the following sets of nitrogen bases in terms of basicity and explain your answerarrow_forwardDraw the structure of chloric and chlorous acid and predict their pKa values using Pauling's rules.arrow_forward
- Provide solution and explanation for the problem below.arrow_forwardgive the scheme of Acetic acid to Terephthalic acidarrow_forwardPrimary amines can be prepared from amides by Hoffman's reaction. Write a general equation for the reaction and give reagents with reaction conditions for this reaction. What property of amines is responsible for them being basic. Suggest one way of Increasing the basicity of an amine and give a specific example of its application.arrow_forward
- Give only typing answer with explanation and conclusion Which of the following statements is/are incorrect? (6,25 p) I- In order for an organic compound to be a dyestuff, it must be chromor and auxochrochrome. II- Hydroxyl or amino groups can be given as examples of chromophore groups III- Decrease of chromophore groups in a molecule causes color deepening. A. b) II, III B. a) I only C. d) II only D. c) III onlyarrow_forwardThe side chain of cysteine is weakly acidic. Suppose in a protein, the side chain of a cysteine residue is surrounded by the side chains of several isoleucine residues. Would this make the side chain of the cysteine residue more acidic or less acidic? Please explain your answer.arrow_forwardBriefly discuss the following topics and include appropriate examples where necessary. Extremely thermophilic sulphur metabolizers.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Acid-base Theories and Conjugate Acid-base Pairs; Author: Mindset;https://www.youtube.com/watch?v=hQLWYmAFo3E;License: Standard YouTube License, CC-BY
COMPLEXOMETRIC TITRATION; Author: Pikai Pharmacy;https://www.youtube.com/watch?v=EQxvY6a42Dw;License: Standard Youtube License