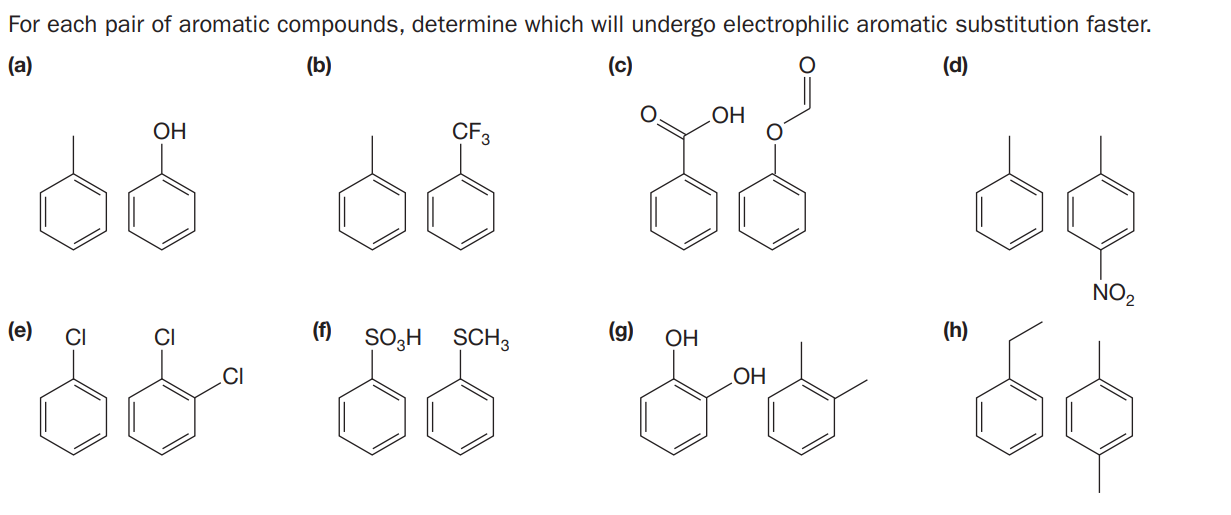
For each pair of aromatic compounds, determine which will undergo electrophilic aromatic substitution faster. (a) (b) (c) (d) LOH ОН CF3 NO, (e) CI CI (f) SO3H SCH3 (g) (h) OH LOH
Q: Draw a detailed mechanism for the FeBr3@catalyzed reaction of ethylbenzene with bromine, and show…
A: The ethylbenzene on reaction with bromine in the presence of FeBr3 gives 1-Bromo-4-ethylbenzene and…
Q: v.. Using curved arrow notation, propose a detailed mechanism that is plausible for the following…
A: This is an example of formation of bromonium ion followed by intramolecular SN2 reaction.
Q: at is the correct order of decreasing reactivity ost reactive first) of these compounds toward…
A: When electron donating groups are attached to benzene ring.so, they increase the reactivity towards…
Q: From 1 being the slowest and 4 being the fastest rank the aromatic hydrocarbons in terms of…
A: The compounds given are,
Q: Below is one of four steps in an electrophilic aromatic substitution reaction. Braw the curved arre…
A:
Q: What is the slow (rate-determining) step in any electrophilic aromatic substitution reaction? Please…
A: Please find below a example of the reaction mechanism
Q: Complete the mechanism of the given acid-catalyzed imine to enamine tautomerization by adding any…
A: This question is simply asking the mechanism of enamine formation through Tautomerisation process.…
Q: Identify the conjugate base of the molecule in the box. Provide a curved arrow mechanism showing the…
A:
Q: The reaction shown here yields three different nucleophilic substitution products that are…
A: Nucleophilic Substitution reaction- The replacement one atom or group of atom from reactant…
Q: Amides are moderately acidic at the N atom, so they can be alkylated in a fashion that is quite…
A: In this reaction, we have an amide compound which is treated with sodium hydride (a base) and then…
Q: Which of the following is expected to be most reactive towards a nucleophilic aromatic substitution…
A: We have to tell which compound is more reactive towards a nucleophilic aromatic substitution via the…
Q: Q3. Rank the compounds in each of the following groups according to their indicated property )…
A: An electrophilic aromatic substitution is a process where an atom or aromatic ring get replaced by…
Q: NaOH NH, + NH3 H20
A:
Q: In both examples below the reactants shown are combined to bring about a nucleophilic substitution…
A: If weak base is used (dil.NaOH), substitution reaction takesplace Benzyl chloride undergo SN1…
Q: (c) Nitrobenzene will undergo electrophilic aromatic substitution at meta position, but not at the…
A:
Q: Predict the main mechanism that occur for the following reaction
A: Nucleophilic substitution reactions the electron-rich nucleophile attacks on a positively charged…
Q: Which of the following is the rate- determining step in the electrophilic aromatic substitution of…
A: In Electrophilic aromatic substitution of benzene an electrophile replaces the hydrogen atom present…
Q: . Predict the products of the following acid-base reactions, and draw a curved arrow mechanism…
A: # Bronsted Acid- The species that can donate H+ ion. # Bronsted…
Q: In a nucleophilic substitution reaction, complete inversion of atom is observed. Would the mechanism…
A: In substitution reaction in which one nucleophile is substituted with another nucleophile is called…
Q: a) Carry out the following transformation and provide mechanism. C,H; CH: H a) H.C. C NH2 -COOH Ph…
A:
Q: Electrophilic aromatic substitution involves a cationic intermediate in which positive charge is…
A: Mechanism is given below
Q: N. H. c) Carry out the following conversion and provide mechanism. NO2 NO2 i) N. COCH3 .COOH ii) N.…
A:
Q: During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed.…
A:
Q: At what position, and on what ring, would you expect the following substance to undergo…
A:
Q: Please explain the following clearly. 1.Benzene undergoes electrophilic aromatic Substitution rxns.…
A: As per our guidelines, we can only solve one question at a time please post other questions as next…
Q: Give two possible [1,2]-rearrangement products where an alkyl group migrates in the following…
A: Organic reaction mechanisms:
Q: For each sequence of reactions, draw the complete, detailed mechanism and predict the major organic…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 5) Provide a complete and detailed mechanism for these transformations. OH heat, acid +
A: We have to predict the mechanism for given reaction.
Q: H₂O TfOH
A: Solution: Alkynes can be hydrated to form enols that immediately tautomerize into ketones. The…
Q: 3/5 5.1 each indicated reaction. Mechanisms. Using curved arrows, suggest a reasonable mechanism for…
A: The reaction of alkene with phosphoric acid leads to the formation of carbocation. The oxygen of -…
Q: Which nucleophilic substitution mechanisms would require a polar protic solvent? A SN1 B) SN2
A: A nucleophilic substitution reaction is a class of chemical reaction . In this , an electron rich…
Q: Suggest a reasonable mechanism for the following reaction Ph OCH3 Ph. OCH3 FeCla Ph Ph OCH3
A: FeCl3 acts as a catalyst. It helps in removal of methoxy group to produce electrophile. Attack of…
Q: Which of the following carbocations is (are) likely to undergo rearrangement through a hydride…
A:
Q: Which of the following is most likely to act as a base rather than a nucleophile?
A: From the given alkoxide most like base is tertiary-butoxide Because it contains more alkyl and it…
Q: During electrophilic aromatic substitution, a resonance-stabilized cation intermediate is formed.…
A: An electrophile attacks at more nucleophilic site. In case of electron donating group, ortho and…
Q: Which of the following is the correct order of increasing reactivity in nucleophilic substitution…
A: Acid halides, esters , anhydrides, amides etc. are the carboxylic acid derivatives. Their reactivity…
Q: Which of the following statements is FALSE? OA Bromobenzene is less reactive than benzene in an…
A: All the electron donating substituents on the ring activates the ring towards electrophilic aromatic…
Q: Provide a reasonable mechanism for the following transformation. Includeall intermediates as well as…
A: The mechanism is as follows:
Q: Predict the most likely sites of electrophilic aromatic substitution in each of the following…
A: Predict the most likely sites of electrophilic aromatic substitution in the above molecules are…
Q: Here is a reaction leading to the formation of a racemic mixture of alcohols as products majors:…
A:
Q: Draw the mechanism of the following reaction taking into account the stereochemistry Cl2 H20
A: The mechanism can be given as follows: Step 1: The attack of the alkene on the chlorine is shown…
Q: For each pair of compounds determine which will undergo electrophilic substitution faster. (a) (b)…
A: As there are more than three parts of this question, we have solved the first three parts for you,…
Q: 1) СО, Р, Д 2) H20, H*
A:
Q: Predict the site on each molecule that is most likely to undergo electrophilic aromatic…
A: (a)
Q: The reaction of aniline with benzoyl chloride (an acid chloride) produces a mixture of two products,…
A:
Q: On the left-hand (reactants) side of the reaction, provide the curved arrows (using the formalism…
A:
Q: SN2 reactions takes place with inversion of configuration, while SN1 reaction takes place with…
A: An organic problem based on substitution reactions, which is to be accomplished.
Q: Provide a plausible arrow pushing mechanism for the reaction below. Note that the starting material…
A: Applying basic rule of reagents and chemical reaction.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Which sequence ranks the following aromatic rings of this compound in order of increasing reactivity in an electrophilic aromatic substitutionreaction (slowest to fastest reacting)?Predict the most likely sites of electrophilic aromatic substitution in each of the following molecules.Which of these compounds would undergo nucleophilic aromatic substitution the fastest?
- Usually at the 2- or 3- position, Thiophene undergoes electrophilic aromatic substitution. Draw the intermediates and all resonance structures for the reaction at each position, and then decide what position should be more reactive depending on your intermediates.Provide an arrow pushing mechanism for the following transformation and clearly circle the slowest (rate-determining) step in the mechanism.(a) For which aromatic compound do you expect nitration to take place faster: furan or thiophene? (b) For each of these compounds, at which C atom do you expectelectrophilic aromatic substitution to predominantly takeplace? Explain your reasoning.
- Provide a detailed mechanism for the following reactionOrganometallic compounds act as nucleophiles under very specific conditions, and can generate alcohols, alkanes, among others. Describe in detail what these reaction conditions are so that organometallics can perform the addition reaction to carbonyl.Propose a reasonable mechanism for the following reaction.
- Please explain the following clearly. 1.Benzene undergoes electrophilic aromatic Substitution rxns. Why is that? 2. Giving and example and mechanisms, explain the aromatic Substitution reactions.Predict the major product(s) of electrophilic aromatic substitutionArrange the compounds in set in order of decreasing reactivity (fastest to slowest) toward electrophilic aromatic substitution.

