Q: Indicate which enzyme works on each molecule. a) Amylose b) Hydrogen peroxide c) DNA
A: a. Amylose Salivary amylase breaks down amylose, amylopection into smaller chain of glucose called…
Q: OH | || CH3CHCH,CSR → CH3CH=CHCSR
A:
Q: List the four factors that influence the rate of a chemical reaction and state whether increasing…
A: The speed of consumption of reactants or the formation of products in a reaction per unit of time is…
Q: Describe several general ways that Ab-Ag reactions are detected.
A: Antigens are the protein molecules capable of inducing an immune response. Antibodies…
Q: In the partial reactions shown below, is the reactant undergoing oxidation or reduction?
A: Oxidation is a process, in which electrons are removed, whereas reduction is just the opposite. In…
Q: Show that the half-life for a first-order reaction is inversely proportional tothe rate constant,…
A: First-order reaction is the reaction that depends on the concentration of only one reactant.
Q: what are the factors affecting the rate of reactions and show the mechanism of each factors.
A: The rate of reaction is the speed of reaction at which the product is formed from the substrate in a…
Q: Does AG´° determine whether a reaction is spontaneous? Why?
A: Free energy of Gibb's free energy indicates the portion of the total energy of a system that is…
Q: Identify the specific molecules that powers endergonic reactions
A: An endergonic reaction (also known as an energy-absorbing nonspontaneous reaction) is a chemical…
Q: Which statement describes the results of this reaction: 6 CO2 + 6 H2O + Energy C6H1206 + 6 02 ?
A: CO2 is reduced and H2O is oxidised.
Q: Define reversible reaction.
A: A reversible reaction is a reaction in which conversion of reactant to product and conversion of…
Q: What are the Determinants of Reaction Rates?
A: Study of chemical reactions is known as reaction kinetics. Chemical reaction refers to the reaction…
Q: Under what conditions might a reaction be endothermic but exergonic? Explain.
A: The exothermic reaction is a process, in which energy is released, whereas the endergonic process…
Q: Use the following illustration of a biochemical reaction to answer the questions below. a. Is this…
A: Enzyme catalysed reaction means that enzyme help in binding the substrate in its active right and…
Q: Define the following terms: a. hydride ion b. oxidation reaction c. energy d. FAD e. electron…
A: As per the honor code, we answer only three subparts at a time. Therefore we are answering the first…
Q: Give the name of the enzyme in this reaction along with detailed
A: Coenzyme A is a coenzyme which plays important role in the synthesis and oxidation of fatty acids,…
Q: Experiment #1: The Effect of Concentration on Reaction Rate Number of Drops of Enzyme 2 4 6 Reaction…
A: During a biochemical reaction, substrate molecules get converted into product according to the…
Q: For the reaction below: CH3CH2I DMF
A: It is an williamson ether synthesis
Q: Which curve represents the spontaneous reaction, and which the nonspontaneous?
A: Gibbs Free energy is the energy that was available to do work under constant temperature and…
Q: Show the complete reaction mechanism (including arrow pushing) for this reaction.
A: Deamination is the process of removal of the amino group from the amino acids as NH3. The liberated…
Q: Draw a Gibbs free energy plot for a spontaneous reaction, and then draw how enzymes affect this…
A: the energy required to convert substrate into the product is called Gibbs energy.
Q: Enzymes sometimes work as a key that opens the lock, if enzyme is the key then what is the…
A: Enzymes are the catalysts that elevate the pace of chemical reactions that occur within the living…
Q: Define the following terms:a. exergonic reactionb. endergonic reactionc. phosphoryl group transfer…
A: Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: Draw a Gibbs free energy plot for a spontaneous reaction, and then draw how enzymes affect this plot
A: Gibbs free energy graph shows whether a reaction is spontaneous or not. It shows if the reaction is…
Q: Label #1-5 with appropriate terms related to enzymes: Binds here (1) and is changed to 2 Must bind…
A: The enzymes are biocatalyst that perform specific reaction within the biological system. It is…
Q: Explain the effects of wavelength of light on the rate of reaction?
A: The wavelength is an important factor for the reactions which are driven by the light or…
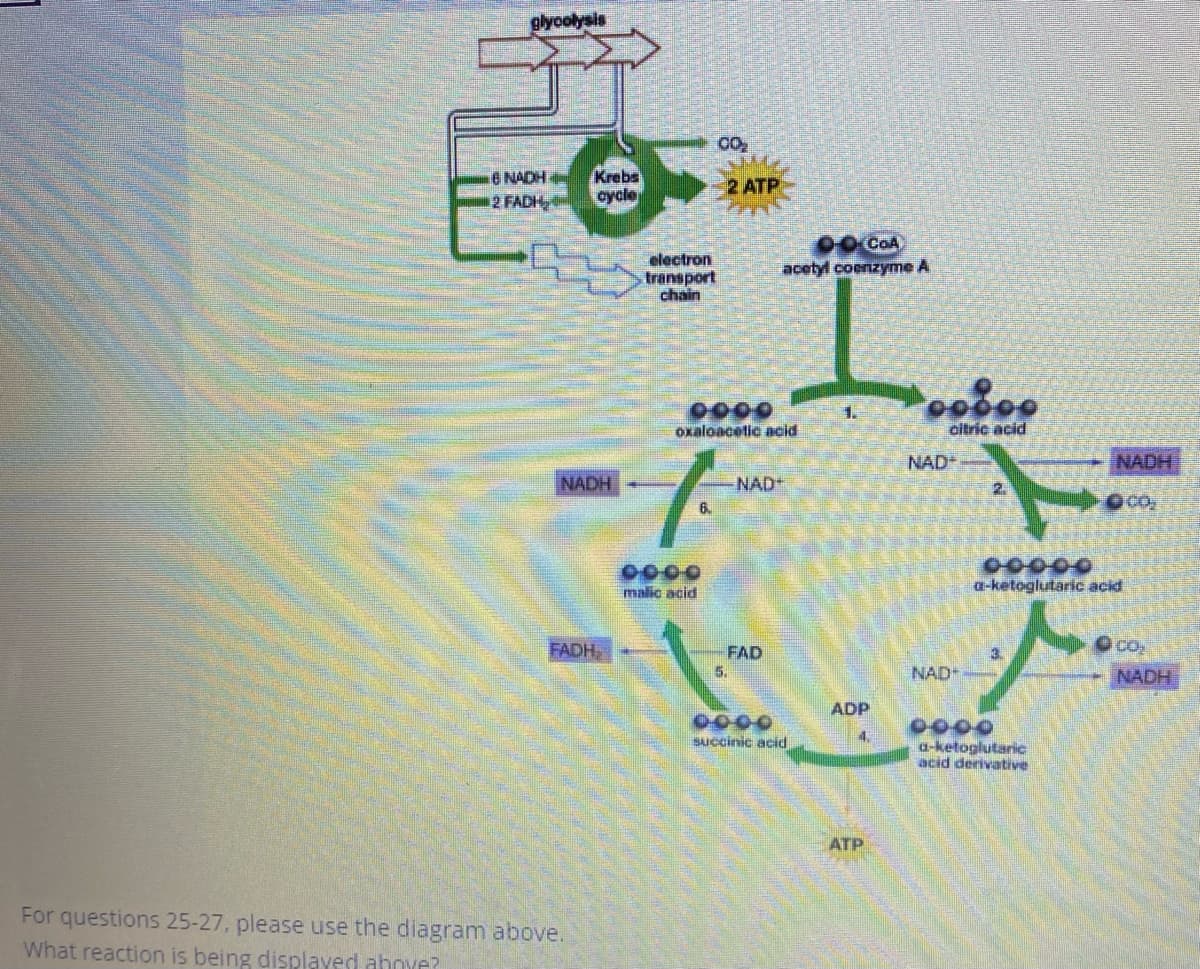
Q: Compare between Kreb's reversible reactions add structures and equations# 14:0
A: Krebs cycle is also known as the citric acid cycle or tricarboxylic acid cycle (TCA). It is one of…
Q: WHAT MAY HAPPEN IF THE FOLLOWING ENZYMES IS ABSENT? answer briefly.
A: Several reactions occur simultaneously inside a cell. These reactions are accelerated by catalysts…
Q: How does INCREASING temperature affect the rate of reaction (how fast the reaction occurs)?
A: Rate of a reaction, in addition to the product produced and having an uninterrupted supply of…
Q: Define the following terms: a. adenosine triphosphate b. redox reaction c. oxidizing agent d.…
A: The molecule is defined as a particle that is made up of two or more atoms that are bonded together…
Q: || CH3CSCOA "02CCH2CSCOA
A:
Q: a. Refer to the reaction below and provide A,B,C and D. b. What type of reaction is taking place…
A: Different reactions involving biomolecules are catalyzed by specific catalyst or enzyme that lowers…
Q: Name the phase of this reaction and Label the diagram Note fand I are locations AD H
A:
Q: NH2 fofota HO-P- ÓH ОН ОН Using the graphic above, answer the following questions. Is removing…
A: The oxidation reaction is the chemical reaction where the reactants lose either one or a few…
Q: Complete the pathway below by writing the intermediate (I) name, pathway name (P) and enzyme (E)…
A: 1.P- Glycogenolysis 2.P- Glycogen synthesis 3.I- Glucose-6-phosphate 4.P- Pentose phosphate pathway…
Q: CH3CSCOA -02CCH2CSCOA O=
A: The reaction showed the carboxylation of acetyl-CoA to produce malonyl-CoA. This is a…
Q: Explain why using higher concentration of substrate does not produce a significant change in the…
A: Enzymes are catalysts that help speed up the rate of a chemical reaction. They do so by lowering the…
Q: Draw the product of the reaction below. (Upload your answer here) H- но ? NaBH4 HO -H H- ČH2OH
A: Given structure :- D-GALACTOSE (monosaccharide) In presence of NaBH4 , REDUCTION reaction…
Q: Use the data from the table below to answer the questions on an ezyme obtained from potato extract.…
A: Enzyme activity is regarded as the amount of enzyme that will be required to convert 1 mole of a…
Q: Define the following terms:a. reaction orderb. turnover numberc. double-displacement reactiond.…
A: Enzymes are the biological catalyst which result in the catalysis of the reaction and the formation…
Q: catalyzes the reaction
A: pyruvate +NADH+ H+ ® Lactate +NAD+
Q: Define the following terms: a. catalytic dyad b. transition state theory c. hydroxide ion catalysis…
A: BASIC INFORMATION ENZYMES They are the catalyst. They help in accelerating the chemical reaction.…
Q: Describe activation energy.
A: Biology terms are fundamental concepts and terms used in biology, which is the study of life and…
Q: explain the difference between a "spontaneous" reaction and a "non-spontaneous" reaction
A: A chemical reaction is a process by which a substance is converted into another substance which has…
Q: CH3CSCOA "0,CCH2CSCOA
A: In the given reaction acetyl-CoA is converted into malonyl-CoA and this reaction occurs during fatty…
Q: Distinguish between exergonic and endergonic reactions, and give examples of how they may be…
A: Exergonic and endergonic chemical reactions enable organisms to carry out their functions, such as…
Q: Give an example of each Irreversible and Reversible Reactions?
A: Cells, the most fundamental and vital unit of life, are found in all living things. A cell is in…
Step by step
Solved in 2 steps

- Consider oleic acid (18:1D9): Total number of ATP produced from oleic acid complete oxidation (exclude transport)?Calculate the ATP yields in respiration given a certain type of substrate and process. Some common substrates and the place in the aerobic pathway where they insert:2-carbon amino acids @ Acetyl CoA3-carbon amino acids @ Pyruvate4-carbon amino acids @ Succinate5-carbon amino acids @ a-ketoglutarateFatty acids oxidized into 2-carbon units that enter @ Acetyl CoAGlycerol enters at glyceraldehyde phosphate 1) a) A dipeptide containing a 4-carbon amino acid and a 5-carbon amino acid b)A dipeptide containing a 4-carbon amino acid and a 5-carbon amino acid c)A 20-unit polypeptide (each unit = a 2-carbon amino acid)Glucose is completely metabolized to six molecules of CO 2. What specifi c reactions generate each molecule of CO 2?
- 1. The enzyme that catalyzes the transformation of C2H2 + H2 to C2H4 is likely to be classified as2. The enzyme that catalyzes the conversion of glucose + ATP to glucose-6-phosphate + ADP is likely to be classified asa. Isomeraseb. Ligasec. Transferased. Lyasee. Hydrolasef. OxidoreductaseCHOOSE THE CORRECT LETTERHow many cycles of B-oxidation are necessary to completely oxidize 3 molecules stearic acid (C18H3602)?A. 24B. 18C. 27D. 21myristic acid (14:0) to carbon dioxide and watera. rounds of the beta oxidation pathway will be involvedb. how many moles of acetyl CoA will be produced after complete beta oxidationc. how many moles of ATP will be obtained after complete beta oxidation
- You experiementally apply the compund antimyctin, which inhinits the functioning of complex III, cytochrome reductase, from participating in redox reactions. You follow this with the addition of 6 molecules of FADH2. How many total ATP molecules can be created using the number of protons that were pumped?Make a concept map using all of the following terms: GlycolysisOxidation of PyruvateCitric Acid CycleElectron Transport ChainChemiosmosisGlucoseOxidative Phos.Substrate level phosphatePyruvateacetyl-CoACO2OxygenWaterreduced elec. carriers (NADH)oxidized elec. carriers (NAD+)High enerGy Elec.Low enerGy Elec.H+ GradientADP + PiATP1. Explain the Chemiosmotic theory with regard to ATP generation in oxidative phosphorylation. 2. Explain how Phosphorylase activation differs between the liver and the muscle. What is the role of Epinephrine in Glycogen metabolism. 3. List all the materials required for beta-oxidation of cholesterol from six-carbon intermediate Mevalonate.
- Calculate the number of molecules of ATP formed by the complete catabolism of arachidic acid, C 20H 40O 2.myristic acid (14:0) carbon dioxide and watera. how many moles of ATP produced from 1 round of the TCA cycleb. what is the net yield of atp(moles) after completion of oxidation of myristic acidc. 2 activated isoprene involved cholesterol biosynthesisWhich of the following statements about the malate-aspartate shuttle is false? a) 2.5 ATPs are formed per cytoplasmic NADH shuttled. b) Transamination is involved. c) Cytoplasmic oxaloacetate is converted to malate. d) Mitochondrial malate is converted to oxaloacetate. e) Antiport transport is not involved.

