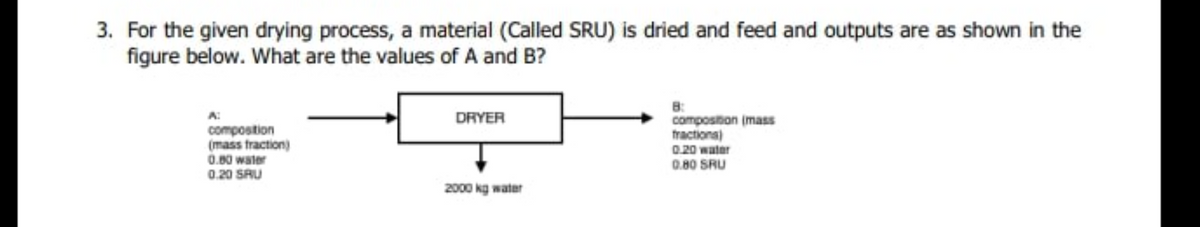
For the given drying process, a material (Called SRU) is dried and feed and outputs are as shown in the figure below. What are the values of A and B? 8: composition (mass tractions) 0.20 water 0.80 SRU DRYER compostion (mass fraction) 0.80 water 0.20 SRU 2000 kg water
Q: The rate of a certain reaction is given by the following rate law: rate =] Use this information to a...
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for yo...
Q: mass
A:
Q: For the following two structures compared in parts A, B, and C determine whether they are enantiomer...
A: Concept : Enantiomer: In chemistry, an enantiomer is one of two stereoisomers of a compound that ar...
Q: 1.00 mol of a perfect monoatomic gas at pressure 1.00 atm and 300 K is compressed adiabatically and ...
A:
Q: Consider the equilibrium system described by the chemical reaction below. Calculate the value of Qc ...
A:
Q: QUESTION 4 Which of the following molecules has a net dipole moment of zero? Please select all the c...
A: In a alkene if 2 same group is situated at trans position than the dipole mement of this compound wi...
Q: Nitrogen and oxygen react at high temperatures. For the reversible reaction, N2(g) + O2(g) ⇌ 2NO(g)...
A: Since you have posted question with multiple subparts as per guidelines we can answer three per ques...
Q: Carbon reacts with water vapor at elevated temperatures. C(s) + H2O(g) ⇌ CO(g) + H2(g) Kc=0.2 a...
A:
Q: Predict the 13C NMR spectra of the 2-chloro-2-methylpropan-1-ol. 1 CH3 2 13 HO-CH2 CI 4CH3
A: The analysis of 13C NMR spectra of the above compound is given below
Q: Consider the four ethers below. Three of them can be made using one (or more) of the methods we disc...
A:
Q: Which of the following are correctly ranked in order of decreasing acidity? The least acidic compoun...
A: The compound in which the congugate base is more stable is more acidic in nature.
Q: How many milliliters of 0.110 MHNO3 contain 7.68 g HNO3?
A: Given :- molar concentration of HNO3 solution = 0.110 mol/L mass of HNO3 = 7.68 g To calculate :- ...
Q: what is the strongest intermolecular force in BH3
A: Intermolecular force acts between molecules
Q: Heptane: yellow flame
A: This is a multipart question.According to the guidelines, in multiple parts question, we can solve o...
Q: Give two examples of common radioisotopes used in medicine. Be sure to write the full name of the is...
A: In medical world radioisotopes used in various ways.Radioisotopes can occurred as naturally or artif...
Q: Mom bought 2 kilos of bangus(fish) for our party next week. To prevent it from spoiling, he sprinkle...
A: Colligative property depend on the number of solute particle present in given amount of solution. T...
Q: Step 3: Number the longest chain of carbon atoms so that the substituents have the lowest possible n...
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: solve and compare the % dissociation of 0.1M and 0.001M solutions of boric acid (Ka= 3.8 x 10-10)
A:
Q: Analysis of sphalerite using X-ray Fluorescence technique is shown in Table 1. Sphalerite may be nea...
A: A question based on molecules that is to be accomplished.
Q: Draw milleY's modutus of tbe 312 Simensiobal Surface with the surface on the A ,2 cookdihates
A:
Q: 3. The two most important inorganic buffer systems in the body are the phosphoric acid and carbonic ...
A:
Q: owances (RDAS) for Vitamin Female Pres 40 mg* 50 mg 15 mg
A: To find the percentage using the given table as,
Q: How many milliliters of water have to be added to 143.8 ml of 0.25 M HCl to reduce the concentration...
A: Given :- Initial concentration of HCl solution = 0.25 M Initial volume = 143.8 mL Final concentra...
Q: What is An for the following equation in relating K c to K p? LICIO 3( s) = LICIO( s) + O 2( 8) 0. 3...
A: Solid lithium chlorate decomposes into solid lithium hypochlorite and oxygen gas. The equation for t...
Q: standard free energy change
A:
Q: Combustion of hydrocarbons such as decane ( C 10 H 22 ) produces carbon dioxide, a "greenhouse gas."...
A:
Q: There are two isomerism in coordination complexes which are structural isomerism and stereoisomerism...
A:
Q: At active muscle I. PCO2 is high II. acidity is low III. carbamate forms IV. hemoglobin is stabilize...
A: 1. True During exercise, CO2 production is increased and thus pCO2 is high. 2. False pH of a normal ...
Q: Draw the reactants that would react to form this cyclohexene derivative in a Diels-Alder reaction. I...
A: Diels Alder reaction is the cycloaddition reaction between diene and dienophile. This is also known ...
Q: In the Fischer projection below, what are the configurations of the two asymmetric centers? H2CH HO ...
A: Rules for assigning R and S notation to fischer projection. • Assign priorities to the atoms/groups...
Q: What is/are the major product(s)_of the following reaction? + Br2 FeBr3 Br Br Br Br II III IV O|| O ...
A:
Q: Given these compounds, what would the chemical shifts look like? Predict them for each compound
A: The structure of an unknown molecule can be determined by analyzing the spectroscopical results such...
Q: c) Compound 3 with molecular formula of C,H,O, 3H ΙΗ ΝMR 3H 2H 15
A:
Q: Br Compound C has Br Compound D has D. Br Br Br Compound E has E. Br C.
A: The number of 1H NMR signals of the cyclopropane derivatives are given below
Q: A 0.04M solution was found to be 1.6% ionized. what is the concentration of the conjugate base?What ...
A:
Q: Calculate the vapour pressure of water at 75 °C. Take the enthalpy of vaporization of water as 40.66...
A: As per our guidelines, we are supposed to answer only one question. Kindly, repost other question as...
Q: H₂(g) + F₂(g) ⇌ 2HF(g) the equilibrium concentrations in a 10.00-L rigid container are [H₂] = 0.060...
A:
Q: For each of the following pairs or reactions, which is correctly labeled as having the larger equili...
A:
Q: When a is on an atom directly attached to a benzene ring, the benzene ring will stabilize it by reso...
A:
Q: conjugate acid
A:
Q: e acids re
A:
Q: how can non-beneficial chemical reactions be minimized or stopped ?
A: how can non-beneficial chemical reactions be minimized or stopped ? Explanation is given below.
Q: Write the empirical formula of at least four binary ionic compounds that could be formed from the fo...
A:
Q: What is the expression for the equilibrium constant? 2B1F5(g) = Br2(g) + 5F2(g) O a) Keq = [Br2] [F2...
A: The correct answer about equilibrium constant is given below
Q: I need to analyze the Ir spectrum and circle the frequency identify the groups and appearance stron...
A:
Q: Draw the major 1,2- and 1,4-addition products formed when this diene reacts with HBr. Do not include...
A:
Q: 5.0 m aqueous solution containing which solute will have the lowest freezing point? A. CaCl2 B. Na...
A: Freezing point of solvent is greater than freezing point of solutions
Q: You react 10.0 g of nitrogen gas with hydrogen gas according to the following reaction: N2(g) + 3 H2...
A: Given reaction: N2(g) + 3 H2(g) → 2 NH3(g) We have to find the mass of hydrogen gas that is require...
Q: 500 1000 1500 2000 2500 3000 seconds created Is BF, being created or destroyed by the chemical destr...
A: The pressure v/s time curve of BF3 is given as,
Q: Draw the two resonance structures of the carbocation intermediate formed in this reaction. Do not in...
A:
Step by step
Solved in 2 steps with 1 images

- A given soil sample is subjected for proximate analysis - specifically moisture content and analysis of inorganic matter (or ash) content. A cleaned and tared crucible is weighed in an analytical balance and found to be 4.5015 grams . The soil sample is weighed-in and the crucible, together with the plant sample, measured a total of 6.2525 grams. The set-up is then placed in an oven and dried to 105 degrees Celsius for at least two hours. The setup is cooled to room temperature in a dessicator and then measured to a constant weight of 5.9775 grams. Afterwards, the setup is placed inside the muffle furnace and ignited to 720 degrees Celsius for at least an hour. The set-up is cooled to room temperature again inside the dessicator and then measured to the constant weight of 4.9375 grams. (A) What is the percent moisture in the plant sample? Write the answer in THREE SIGNIFICANT FIGURES. (B) What is the percent volatile (organic) matter in the plant sample? Express answer in THREE…Hot mix asphalt (HMA) is commonly used in the construction of roadways in the United States at a rateestimated to be 500,000,000 tons/year. A typical upper layer (there may be as many as 5 layers ofdifferent thicknesses and compositions) of a high-volume highway is composed of crushed stone (alsoknown as crushed aggregate) and asphalt binder and is approximately 1.25 inches thick. The stone,which has a specific gravity of about 2.7, is mixed with asphalt binder (SG = 1.03) to form the HMAwith a composition of approximately 95 wt% aggregate and 5% asphalt. What is the volume ft3 of asphalt binder and weight (tons) of HMA required for the top layer of a 15-mile segment of interstatehighway with 5 lanes, each of which has a width of 12 ft?A binary mixture with components A and B is to be separated in a distillation column to obtain 95 mol%. A as the top product. The binary mixture has a con stant relative volatility AB = 2. The column feed is a a - saturated liquid containing 50 mol % of A. Under the usual simplifying assumptions such as constant molal overflow, negligible heat loss, ideal trays, the mini mum reflux ratio for this separation is (rounded off to one decimal place)
- A solution containing 100 lbm KNO 3/100 lbm H 2O at 80°C is fed to a cooling crystallizer operated at 25°C. Slurry from the crystallizer (KNO3 crystals suspended in saturated solution) is fed to a filter, where the crystals are separated from the solution. Use the solubility data in Figure 6.5-1 to determine the production rate of crystals (lbm/lbm feed) and the solid-to-liquid mass ratio (lbm crystals/lbm liquid) in the slurry leaving the crystallizer.If 35,000 kg of whole milk containing 4% fat is to be separated in a 6-hour period into skim milk with 0.45% fat and cream with 45% fat, what are the mass flow rates of the two output streams from a continuous centrifuge which accomplishes this separation? (Ans; Cream=464.8335kg/h, Skim milk= 5368.4998kg/h)A spent sulfuric acid solution is brought up to strength for a pickling process in a mixer. Spent solution at 3% sulfuric acid (by weight) is mixed with a 50% solution (by weight) to obtain the desired product concentration of 40% acid by weight. All are aqueous solutions. Determine all flow rates on the basis of 100 lbm per hour of product. If the actual flow of the spent stream is 300 lbm per hour. What must the flow rates of the stream be?
- The aqueous solution of acetic with a flow rate of 1000 kg/hr contains 30% acetic acid by mass and will be extracted in a countercurrent multistage process with pure isopropyl ether (IPE) to reduce the acetic acid concentration in the final raffinate phase (LN) to 2% by mass. a) Calculate the minimum solvent flow. b) Determine the theoretical number of steps required when using a solvent of 1.5 minimum solvent flow rate. c) Determine the number of theoretical steps using the McCabe-Thiele method. Chart. Acetic acid (A)-Water (B)-isopropyl ether (IPE) (C) LIQUID-Liquid Balances at 1 atm pressure and 293 K Water layer (%) Water (B) Isopropyl ether layer (%) Water (B) 0.6 Acetic acid (A) IPE (C) Acetic acid (A) IPE (C) 98.8 1.2 99.4 99.3 98.9 0.69 98.1 1.2 0.18 0.5 1.41 97.1 1.5 0.37 0.7 2.89 95.5 1.6 0.79 98.4 91.7 1.9 1.0 97.1 6.42 13.30 25.50 36.70 1.9 93.3 4.82 11.40 2.3 84.4 71.1 3.4 3.9 84.7 71.5 21.60 31.10 58.9 4.4 6.9 58.1 48.7 10.6 10.8 45.1 37.1 44.30 46.40 16.5 36.20 15.1…An environmental study was performed to measure the transport of a DDT (a banned pesticide) throughout the globe. A lake in northern Canada was sampled to see if low levels of DDT were present there in spite of it not being administered within 1000 miles of that location. 1.000 L of the lake water was spiked with 100.0 uL of a 2.451 ug/L solution of tetrachlorobenzene, which acted as the internal standard. The entire 1.000 L spiked solution was concentrated onto an SPE column. After washing the SPE column with ~5 mL of pure water, the analyte and internal standard were eluted from the SPE column using ~5 mL of CH2Cl2 solvent. The eluted sample was evaporated under a stream of nitrogen to a final volume of 1.000 mL. 1 uL of this final solution was analyzed using GC-MS, and gave a signal of 24835 for DDT and 11597 for tetrachlorobenzene. To calculate the response factor, a standard solution was prepared consisting of 1.482 ug/L of DDT and 2.451 ug/L of tetrachlorobenzene.…Amylum, commonly known as starch is a white powder with variable molecular formula due to the different number of repeating sugar units based on the plant that it is obtained from. A sample of amylum in the form of spherical beads was subjected to series of analyses to determine physicochemical properties in the quality control unit of a factory. From the results each bead is found to have a 4.5 mL volume and 1.47 g/mL density. 4 of the beads were placed in a reaction chamber combusted, as a result, 43.1 g CO2 and 14.7 g H2O were obtained. If the molecular weight of the amylum is 972.84 g/mol how many repeating units the starch molecule consist and what is the molecular formula of the starch? [Empirical Formula=CxHyOz, Repeating unit=n, Molecular Formula=( CxHyOz)*n= CnxHnyOnz]
- Various parameters in a gas absorption experiment are as follows; Water flow rate =5 L/min, Air flow rate=25 L/min, CO2 flow rate=6 L/min. Then, to find the amount of CO2 absorbed into the water 1 hour after the system is started and stabilized, 50 mL samples are taken from the inlet and outlet of the tank and titrated with 0.01M NaOH. 100mL samples are taken from the tower entrance and exit and sent to the hemp apparatus. The consumptions measured with the help of the Hemp apparatus are 2.1 mL (inlet) and 1.9 mL (output), respectively. According to this, find the amount of CO2 absorbed by titration? (Note: The relative error in the experiment is 10.)An evaporation-crystallization process is used to obtain solid potassium sulfate from an aqueous solution of this salt. The fresh feed to the process contains 19.6% wt K2SO4 The wet filter cake consists of solid K2SO4 crystals and 40 % wt K2SO4 solution, in a ratio 10 kg crystals/kg solution. The filte a 40% solution is recycled to join the fresh food. Of the water fed to the evaporator, 45% is evaporated The evaporator has a maximum capacity of 175 kg/hr water evaporales Calculate a) The maximam production rate of solid K2SO4 b) The rate at which fresh feed must be supplied to achieve this production rate c) The ratio kg recycle kg fresh feedEthyl alcohol (C2H5OH) may be prepared by the fermentation of glucose (C6H12O6) as indicated by the equation: yeastC6H12O6 ----> C2H5OH + CO2 74.12 mL of ethyl alcohol (specific gravity = 0.790) was collected by this fermentation pro- cess. What mass of glucose was used?

