For which isomer would you expect a greater equilibrium percentage of molecules with the alkyl group in the axial position, isopropylcyclohexane or propylcyclohexane? Explain. Isopropylcyclohexane Propylcyclohexane
For which isomer would you expect a greater equilibrium percentage of molecules with the alkyl group in the axial position, isopropylcyclohexane or propylcyclohexane? Explain. Isopropylcyclohexane Propylcyclohexane
Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 25VC: The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy...
Related questions
Question
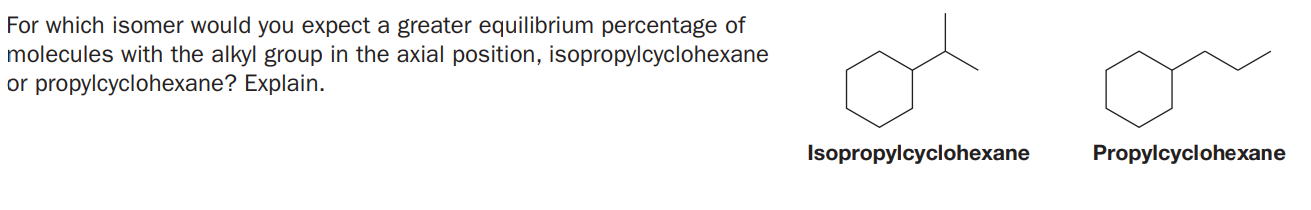
Transcribed Image Text:For which isomer would you expect a greater equilibrium percentage of
molecules with the alkyl group in the axial position, isopropylcyclohexane
or propylcyclohexane? Explain.
Isopropylcyclohexane
Propylcyclohexane
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 5 images

Recommended textbooks for you

