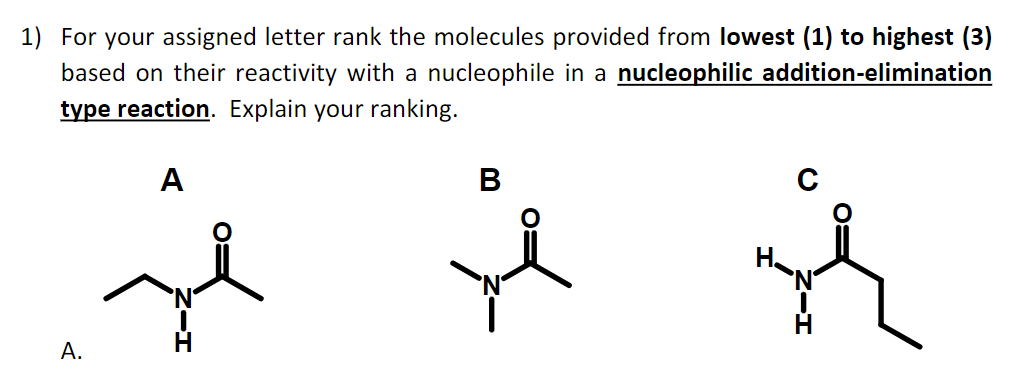
) For your assigned letter rank the molecules provided from lowest (1) to highest (3) based on their reactivity with a nucleophile in a nucleophilic addition-elimination type reaction. Explain your ranking. А В А.
Q: 8. Explain why NaCl is soluble in water whereas CaO is insoluble.
A: Strong sodium ions bound to negative chloride ions are made of salt (sodium chloride). Water can dis...
Q: likely to form when reacting Ethyl amine and excess ethyl
A: Organic reactions are those in which organic reactant react to form organic products. We have to for...
Q: Arrange the salts by their molar solubility in water. value for Most soluble Least soluble
A:
Q: QUESTION: Explain IN DETAIL at least one instance in which chemical equilibrium is utilized in our d...
A: The chemical equilibrium is the state of a reaction in which the forward reaction rate is equal to t...
Q: 7. Given the pKa information for different acids below, place the following acids in order of increa...
A:
Q: ata Endgroup analysis by titration: 8 mols of 1,6-hexanediamine were reacted rith 9.8 mols of adipic...
A:
Q: 5. A scientist has a mixture of two peptides, A and B, and wants to separate them using ion exchange...
A: (a) The resin is positively charged, i.e., anion-exchange resin. So, we need a buffer where both...
Q: In an experiment to determine the amount of aspirin in a tablet, one of the bottles of NaOH used was...
A: Here we required to predict the effect of concentration NaOH in determination of the concentration o...
Q: IBr2 FORMAL CHARGES FORMULA
A: A question based on structure of atom that is to be accomplished.
Q: FORMAL
A: Formal charge is partial or imaginary charge present on an atom in the compound. From the lewis str...
Q: A 1.00 g sample of NaOH which has been exposed to the air for some time is dissolve in water and w...
A:
Q: Provide reagents and conditions with mechanisms to produce the products indicated. HO OH o-CH3 OH OH...
A:
Q: 4. Calculate the molality of a solution prepared from 30.25 grams of NaOH (sodium hydroxide) in 5.00...
A: Molality of the solution is defined as the number of moles of solute present per kg of the solvent.
Q: Which mechanism is consistent with the reaction below? (CH3)3CBr+OH-→(CH3)3COH+Br- 1. (CH3)3CBr+(...
A: Carbocation can be defined as the species in which carbon atom bear positive charge. It can be teria...
Q: A chemist performs a gravimetric analysis. The chemist combines 1.00 L of 2.00 M AGNO3 (ag) with 1.0...
A: Given, AgNO3 solution Molarity of AgNO3 solution = 2.00 M = 2.00 mol/L Volume of AgNO3 solution = 1....
Q: What is the molality of a solution made from 100.1 g of NaCI and 440.0 g of water
A: Given, Mass of NaCl = 100.1 g Mass of water = 440.0 g = 0.440 Kg Molality of the solution = ? Note: ...
Q: litres
A:
Q: Show all steps in the synthesis of 4-methylaniline from toluene. Clearly show all reagents, reactant...
A:
Q: zinc and copper are used in the measurement of food. Are there other topics possible
A: A question based on measurement that is to be accomplished.
Q: 6. Methanal (HCHO) 7. Naphthalene (CioHe) 8. Acetanilide (CH;-NHCOCH) 9. Uracil NH Cyclohexane (CH12...
A:
Q: If your 12x concentrated stock solution contains 15g of NaCl per liter, how much NaCl would one lite...
A: Here we have to determine the mass of NaCl present per Litre of working solution if the concentrated...
Q: the injector is kept at higher temperature than the column so that it will move quickly. If the samp...
A: A multiple choice question based on analytical separations that is to be accomplished.
Q: Write the metabolism of chlordiazepoxide, name all metabolites.
A: Answer - According to the question - Chlordiazepoxide, trade name Librium among others, is a sedativ...
Q: Gibbs Energy and Phase Changes 1. The molar volumes of water and ice at 0°C and 1 atm are 18.02 and ...
A:
Q: Name Date Sect. Data Report Sheet: Quantitative Analysis of Group III Cations Unknown # 2 Draw the f...
A: Group 3 cation involve Cr3+ , Al3+ , Ni2+ and Fe3+.
Q: Calculate the [Y3*1 in a saturated solution of YF in 0.003150 M NaFna): (Kp for YF;= 8.6 x 10")
A: We have to calculate the concentration of Y+3 .
Q: Question 6 A Equation 1: X(s) + NiCl (aq) → Ni(s) + XCl, (ag) Equation 2: X(s) + MgCl2 (ag) → no rea...
A:
Q: 5, 14 12 11 16 15 -y mond shown above give the point indices for atom 15. (grs) format. So if q=0,r=...
A:
Q: What reaction converts nitrobenzene to 1-bromo-3-nitrobenzene Alkylation Acylation Halogenation ...
A: Br is halogen , in the reaction halogenation occur
Q: Part A: Percent Yield of Sodium Carbonate from Baking Soda 97.18g Mass of test tube and beaker ११. २...
A:
Q: If a student performed part A of this experiment and recorded a 122% yield, what could have been som...
A: Please find your solution below : Percentage yield is defined as the percent ratio of actual yield t...
Q: C Po undergoes four decay reactions: a, B, a, ß. Step 1. 218 Po undergoes a decay to give 84 Step 2....
A: Nuclear chemistry is the branch of chemistry in which we deal with positron emission, electron emiss...
Q: A sample of chlorine gas with an initial volume of 35.6 L was found to have a volume of 27.2 L at 15...
A:
Q: 1. The knowledge about colligative properties of solution and concentration of solution are very use...
A: A question based on properties of liquids that is to be accomplished.
Q: Carbonyl chloride (COCI), also called phosgene, was used in World War I as a poisonous gas. The equi...
A:
Q: What pressure in atm is exerted by a 56.0g of Xe in a 5.00L container at 20°C
A:
Q: What are the chemical properties of (Mesitylene) molybdenum tricarbonyl?
A: The delocalized - electron ring bonds the mesitylene group to the molybdenum centre. The ability of ...
Q: MULTIPLE CHOICE Question 12 Which of the following statements most accurately describes forever chem...
A: PFAS are a family of thousands of human-made substances – also known as “forever chemicals” because ...
Q: Finding Molecular Weights from Experimental Data a. Endgroup analysis by titration: 9.8 mols of 1,6-...
A:
Q: 1. Which of the following is the strongest acid? HO, OH а. b. F С. d. Cr e. ON
A:
Q: True/False Indicate whether the statement is true or false. 1. A system at equilibrium means the con...
A:
Q: The solubility of CO2(g) in water at 25 °C and under a pressure of 1.00 atm is 1.45 g/L. If the air ...
A:
Q: 10.00 mL sample of 1.20 M HNO2 (analyte) (pKa = 3.34 and Ka = 4.57x10-4) is titrated with 1.20 M NaO...
A: Given , pka = 3.34 Molarity of HNO2 = 1.20 M Volume of HNO2 = 10 mL Mmol of HNO2 = M × V = 1.20 ×1...
Q: Please identify the each stament below whether it is true or false, and leave an explanation. Thank ...
A: Given:- Identify whether it is true or false, and leave an explanation:- 1. All the gases in the w...
Q: Question 4 Calculate the wavelength of visible light (in nanometers) that has a frequency of 6.831 x...
A: We are given : Frequency of light = 6.831 * 1014 Hz
Q: 5. Explain why metals exhibit the following properties. – page 269 shiny flexible Electrical conduct...
A: Metals are shiny-The positive metal ions are surrounded by pool of electrons. The electrostatic forc...
Q: Consider the five esters below with formula C ,H 1202: (a) (b) (c) (d) (e) (i) Which is/are mad e fr...
A: The ester is made from the condensation reaction between a carboxylic acid and an alcohol.
Q: Determine the Ksp expression for Al2(SO4)3(s) ⇌ 2Al3+(aq) + 3SO42-(aq)
A: Given :- Al2(SO4)3(s) ⇌ 2Al3+(aq) + 3SO42-(aq) To determine :- Ksp expression of given equation
Q: . identify the structure of A and B in the following synthetic scheme: Write out complete reactions...
A: By observing the nature of the reagents used, we can expect ketone and imine as products respective...
Q: Consider the reaction: C(s) + H2O + heat (double arrow) CO(g) + H2, what will happen when you remove...
A: Le Chatelier's principle states that when concentration, temperature or pressure of the system is ch...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- p-Fluoronitrobenzene is more reactive toward hydroxide ion than is p-chloronitrobenzene. What does this tell you about the rate-determining step for nucleophilic aromatic substitution?Define the General Mechanism–Acid-Catalyzed Nucleophilic Addition ?Given a target molecule below, propose a synthetic route involving either the Wittig reaction or HornerEdmonson approach. Justify your choice, considering both reagents and selectivity
- Please I need help with this question Based on the experiment- Synthesis of Iodosalicylamide – An Electrophilic AromaticSubstitution (a derivative of benzene, salicylamide was used and substituted an iodine atom onto the ring; the ring is the nucleophile while the atom/molecule that is being substituted onto the ring has anelectrophilic character) Please I need help with this QUESTION: Draw the structure of the final product(s) based on the ATTACHED IR analysis. Also label the signals (both the frequency and molecular motion) that help determine the final product(s). Thanks in advanceStep 2 of the iodination of benzene shows water acting as a base and removing a proton from the sigma complex. We did notconsider the possibility of water acting as a nucleophile and attacking the carbocation, as in an electrophilic addition to an alkene.Draw the reaction that would occur if water reacted as a nucleophile and added to the carbocation. Explain why this type ofaddition is rarely observed.Decide which compounds from the list below are best suited for nucleophilic addition reactions and which ones are more appropriate for nucleophilic substitution reactions.
- Please provide 3 possible mechanisms for bromination of trans-cinnamic acid and pyridinium tribromide (Br2) with stereochemistry (wedge and dash bond) (1 for anti addition, 1 for syn addition, and 1 for 50:50 syn and anti additon).Arrange the structure on the image with regard to the reactivity towards nucleophilic acyl substitution 1 being the least and 3 being the mostFrom 1 being the slowest and 4 being the fastest rank the aromatic hydrocarbons in terms of increasing raters of nitration via electrophilic aromatic substitution
- Determine which nucleophilic aromatic substitutions are likely, and proposemechanisms for both the addition–elimination type and the benzyne type.In the chemical reaction between crotyl chloride and ethanolic silver nitrate solution, a precipitate is definitely formed. Using the skeletal structures of the molecules, write out the skeletal structure of each reaction (don't include the mechanism). Identify what the nucleophile is in each type of reaction, especially within this SN1 reaction. Explain the reactivity of each electrophile/substrate in terms of whether they are tertiary, secondary, primary, etc. for each reaction. Include the overall chemical reaction.In the chemical reaction between benzyl chloride and ethanolic silver nitrate solution, a precipitate is barely formed. Using the skeletal structures of the molecules, write out the skeletal structure of each reaction (don't include the mechanism). Identify what the nucleophile is in each type of reaction, especially within this SN1 reaction. Explain the reactivity of each electrophile/substrate in terms of whether they are tertiary, secondary, primary, etc. for each reaction. Include the overall chemical reaction.

