Freezing Point of pure TBA (experimentally determined) = 22.90 °C. (C)(kg solvent) mol solute Molal Freezing Point Depression Constant Data - Freezing Point with 1" addition of Solute Time min'sec" Temperature Time min'sec" Temperature (decimal) (°C) (decimal) (°C) 0'00" (0.00) 37.00 0'15" (0.25) 31.40 0'30" (0.50) 27.50 0'45" (0.75) 23.80 1'00" (1.00) 21.00 1'15" (1.25) 18.80 1'30" (1.50) 16.60 Freezing Point Data A Time (min) Temp (C) 0.00 0.25 0.50 0.75 1.00 1.25 1.50 1.75 2.00 2.50 3.00 3.50 4.00 4.50 5.00 5.50 6.00 23.80 21.00 18.80 16.60 37.00 Freesing Point Duta B 31.40 Time (min) Temp (C) 27.50 0.50 m1 -13.271 bl 35.111 Intersection Temp 0.75 1.00 1.25 1.50 1.75 2.00 2.50 3.00 3.50 4.00 4.50 5.00 m2 5.50 6.00 -1.35 b2 17.75 15.79 "C 3'00" (3.00) 13.70 3'30" (3.50) 13.00 4'00" (4.00) 12.20 4'30" (4.50) 11.60 15.50 15.00 14.50 13.70 13.00 12.20 11.60 11.00 10.40 9.80 1'45" (1.75) 15.50 2'00" (2.00) 15.00 2'30" (2.50) 14.50 2 40.00 35.00 30.00 25.00 20.00 15.00 10.00 5.00 0.00 ТВА' k,= 1.00 y=-13.271x 35.311 a) What is the freezing point of the solution? b) What is the molar mass of the unknown? 8,09 Freezing Point Determination Temperature (°C) vs Time (min) 2.00 Time min'sec" Temperature (decimal) (°C) 1.00 5'00" (5.00) 11.00 5'30" (5.50) 10.40 6'00" (6.00) 9.80 4.00 Time (min) y=-1.3457x+17,75 5.00 6.00 7.00
Freezing Point of pure TBA (experimentally determined) = 22.90 °C. (C)(kg solvent) mol solute Molal Freezing Point Depression Constant Data - Freezing Point with 1" addition of Solute Time min'sec" Temperature Time min'sec" Temperature (decimal) (°C) (decimal) (°C) 0'00" (0.00) 37.00 0'15" (0.25) 31.40 0'30" (0.50) 27.50 0'45" (0.75) 23.80 1'00" (1.00) 21.00 1'15" (1.25) 18.80 1'30" (1.50) 16.60 Freezing Point Data A Time (min) Temp (C) 0.00 0.25 0.50 0.75 1.00 1.25 1.50 1.75 2.00 2.50 3.00 3.50 4.00 4.50 5.00 5.50 6.00 23.80 21.00 18.80 16.60 37.00 Freesing Point Duta B 31.40 Time (min) Temp (C) 27.50 0.50 m1 -13.271 bl 35.111 Intersection Temp 0.75 1.00 1.25 1.50 1.75 2.00 2.50 3.00 3.50 4.00 4.50 5.00 m2 5.50 6.00 -1.35 b2 17.75 15.79 "C 3'00" (3.00) 13.70 3'30" (3.50) 13.00 4'00" (4.00) 12.20 4'30" (4.50) 11.60 15.50 15.00 14.50 13.70 13.00 12.20 11.60 11.00 10.40 9.80 1'45" (1.75) 15.50 2'00" (2.00) 15.00 2'30" (2.50) 14.50 2 40.00 35.00 30.00 25.00 20.00 15.00 10.00 5.00 0.00 ТВА' k,= 1.00 y=-13.271x 35.311 a) What is the freezing point of the solution? b) What is the molar mass of the unknown? 8,09 Freezing Point Determination Temperature (°C) vs Time (min) 2.00 Time min'sec" Temperature (decimal) (°C) 1.00 5'00" (5.00) 11.00 5'30" (5.50) 10.40 6'00" (6.00) 9.80 4.00 Time (min) y=-1.3457x+17,75 5.00 6.00 7.00
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.12QAP
Related questions
Question
Q1 please

Transcribed Image Text:PRE-LAB QUESTIONS
1. The following experimental data were collected and graphed:
Mass of pure TBA used = 15.348 g
1.081 g
Mass of FIRST Sample in TBA=
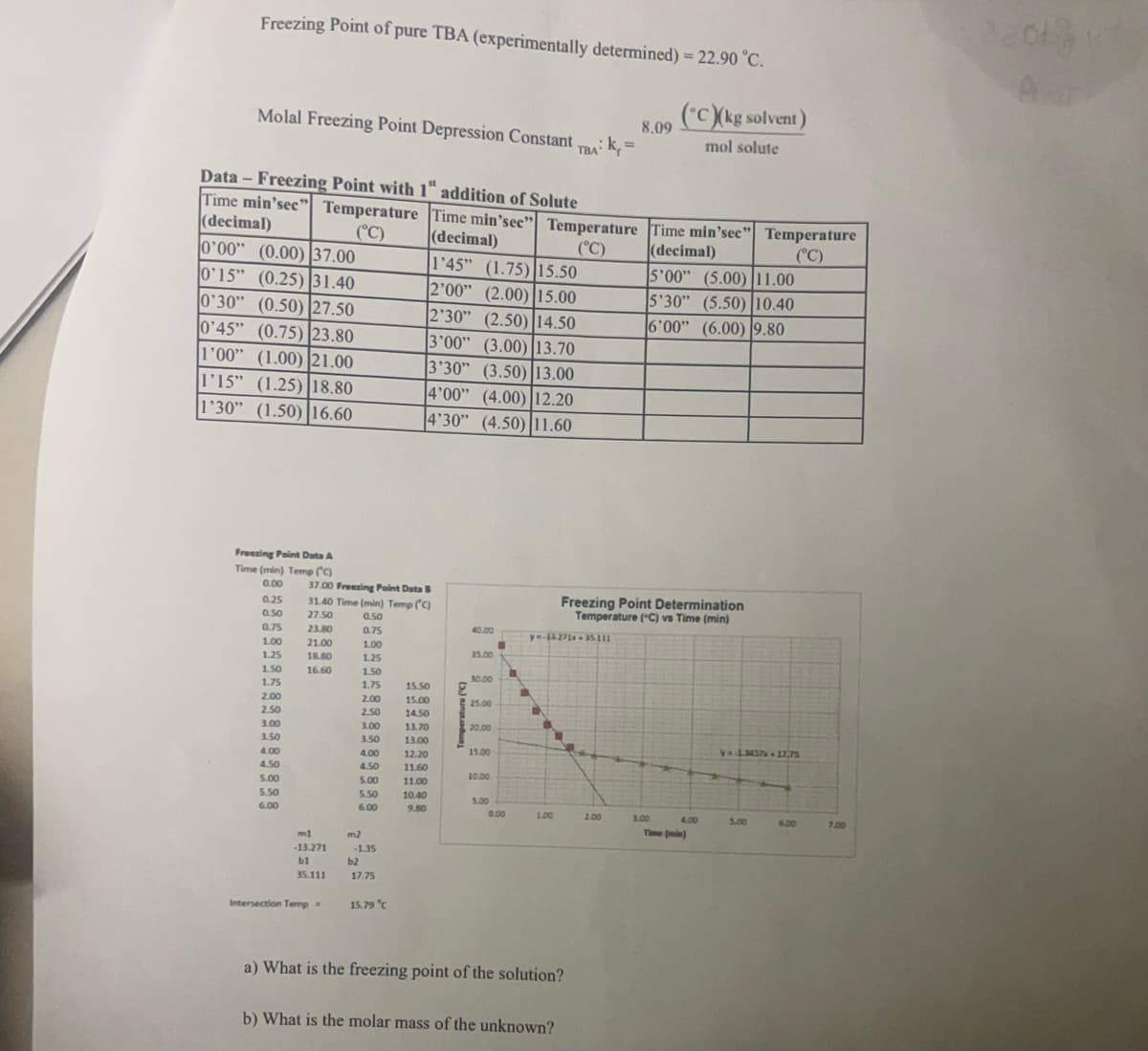
Transcribed Image Text:Freezing Point of pure TBA (experimentally determined) = 22.90 °C.
(C)(kg solvent)
mol solute
Molal Freezing Point Depression Constant.
0'00" (0.00) 37.00
0'15" (0.25) 31.40
0'30" (0.50) 27.50
0'45" (0.75) 23.80
1'00" (1.00) 21.00
1'15" (1.25) 18.80
1'30" (1.50) 16.60
Data- Freezing Point with 1" addition of Solute
Time min'sec" Temperature Time min'sec" Temperature Time min'sec" Temperature
(decimal)
(°C)
(decimal)
(°C)
(decimal)
(°C)
Freezing Point Data A
Time (min) Temp (C)
0.00
0.25
0.50
0.75
1.00
1.25
1.50
1.75
2.00
2.50
3.00
3.50
4.00
4.50
5.00
5.50
6.00
37.00 Freezing Point Data B
31.40 Time (min) Temp (C)
27.50
0.50
23.80
0.75
21.00
1.00
18.80
1.25
16.60
1.50
1.75
m1
-13.271
bl
35.111
Intersection Temp.
2.00
2.50
3.00
3.50
4.00
4.50
5.00
5.50
6.00
m2
-1.35
b2
17.75
15.79 °C
15.50
15.00
14.50
13.70
13.00
12.20
11.60
11.00
10.40
9.80
1'45" (1.75) 15.50
2'00" (2.00) 15.00
2'30" (2.50) 14.50
3'00" (3.00) 13.70
3'30" (3.50) 13.00
4'00" (4.00) 12.20
4'30" (4.50) 11.60
E
40.00
35.00
30.00
25.00
20.00
15.00
10.00
5.00
0.00
TBA: K
1.00
y=-13.271x + 35.311
a) What is the freezing point of the solution?
b) What is the molar mass of the unknown?
8,09
Freezing Point Determination
Temperature (°C) vs Time (min)
2.00
5'00" (5.00) 11.00
5'30" (5.50) 10.40
6'00" (6.00) 9.80
3.00
Time (min)
y=-1.3457x + 17,75
5.00
6.00
7.00
201
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning