Freezing point Tr of pure TBA, °C Mass of the empty test tube, g Mass of the test tube with TBA, g Mass of the test tube with TBA and benzoic acid, g Mass of TBA, g and kg Mass of benzoic acid, g Freezing point Tr of the benzoic acid-TBA mixture, °C Freezing temperature depression AT, °C Molality of solution, mol/kg Moles of benzoic acid
Freezing point Tr of pure TBA, °C Mass of the empty test tube, g Mass of the test tube with TBA, g Mass of the test tube with TBA and benzoic acid, g Mass of TBA, g and kg Mass of benzoic acid, g Freezing point Tr of the benzoic acid-TBA mixture, °C Freezing temperature depression AT, °C Molality of solution, mol/kg Moles of benzoic acid
Chapter4: Least-squares And Calibration Methods
Section: Chapter Questions
Problem 12P
Related questions
Question
Please show your calculations all organized and neat for each box
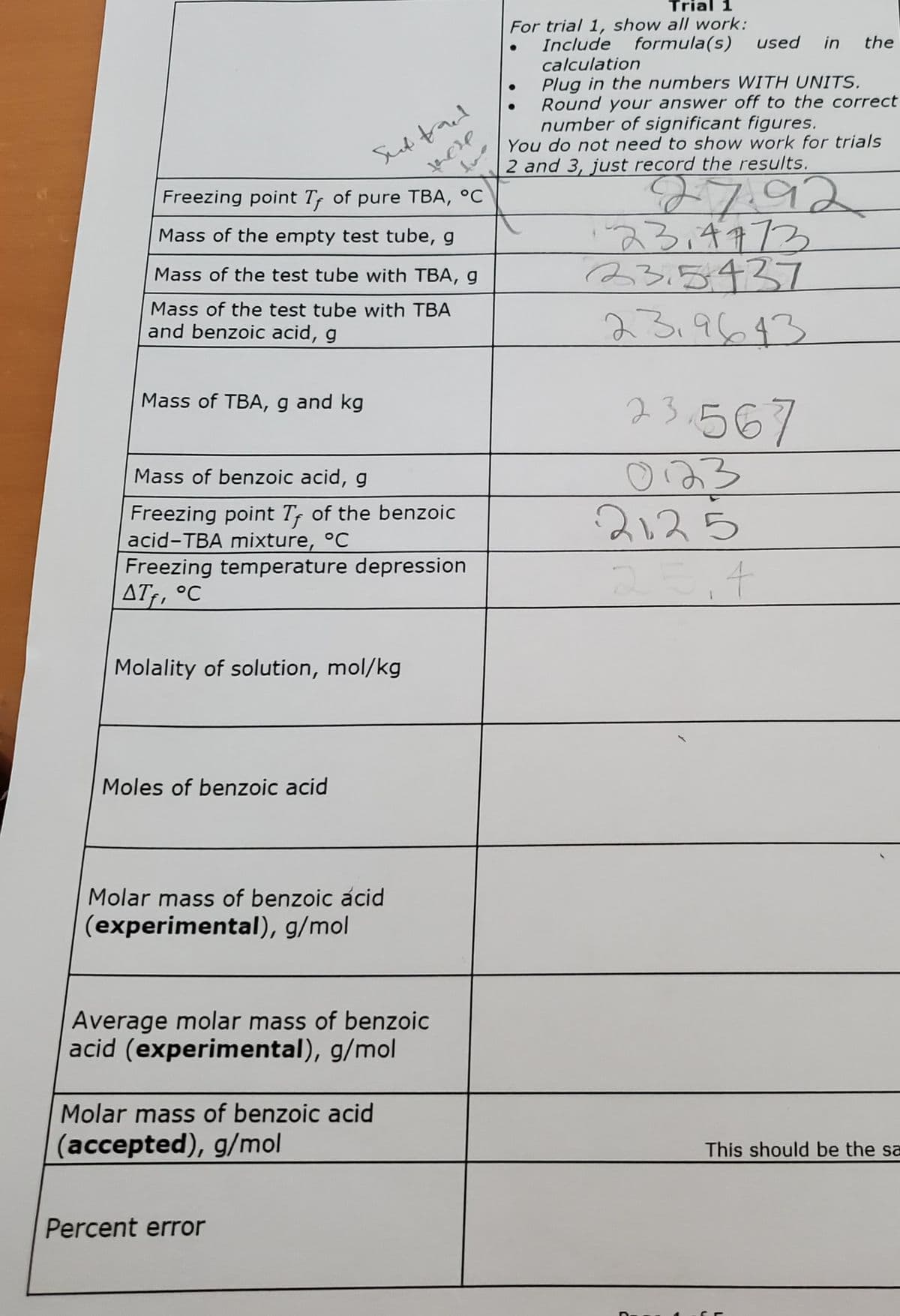
Transcribed Image Text:Trial 1
For trial 1, show all work:
Include formula(s)
calculation
Plug in the numbers WITH UNITS.
Round your answer off to the correct
number of significant figures.
You do not need to show work for trials
2 and 3, just record the results.
used
in
the
Sud tacd
27.92
スろチャアろ
23.5437
23.9643
Freezing point Tr of pure TBA, °C
Mass of the empty test tube, g
Mass of the test tube with TBA, g
Mass of the test tube with TBA
and benzoic acid, g
23.567
Mass of TBA, g and kg
o23
2125
Mass of benzoic acid, g
Freezing point Tr of the benzoic
acid-TBA mixture, °C
Freezing temperature depression
AT, °C
Molality of solution, mol/kg
Moles of benzoic acid
Molar mass of benzoic acid
(experimental), g/mol
Average molar mass of benzoic
acid (experimental), g/mol
Molar mass of benzoic acid
(accepted), g/mol
This should be the sa
Percent error
Expert Solution
Step 1
Since you have posted a question with multiple sub-parts, we will solve first three subparts for you. To get the remaining sub-part solved please repost the complete question and mention the sub-parts to be solved.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

