Gasohol is a mixture of ethanol (C2H5OH) and gasoline. Write the chemical equation for the combustion of ethanol. (Use the lowest possible whole number coefficients. Include states-of-matter under SATP conditions in your answer.) b. Determine the maximum amount of work that can be obtained from the combustion of 1 gal of ethanol at 298.15 K and standard conditions. The density of ethanol is 0.789 g/mL, and 1 gal is 3.79 L. (See Thermodynamic Properties pictures attached)
Gasohol is a mixture of ethanol (C2H5OH) and gasoline. Write the chemical equation for the combustion of ethanol. (Use the lowest possible whole number coefficients. Include states-of-matter under SATP conditions in your answer.) b. Determine the maximum amount of work that can be obtained from the combustion of 1 gal of ethanol at 298.15 K and standard conditions. The density of ethanol is 0.789 g/mL, and 1 gal is 3.79 L. (See Thermodynamic Properties pictures attached)
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter2: Matter
Section: Chapter Questions
Problem 22A
Related questions
Question
100%
a. Gasohol is a mixture of ethanol (C2H5OH) and gasoline. Write the chemical equation for the combustion of ethanol. (Use the lowest possible whole number coefficients. Include
b. Determine the maximum amount of work that can be obtained from the combustion of 1 gal of ethanol at 298.15 K and standard conditions. The density of ethanol is 0.789 g/mL, and 1 gal is 3.79 L. (See
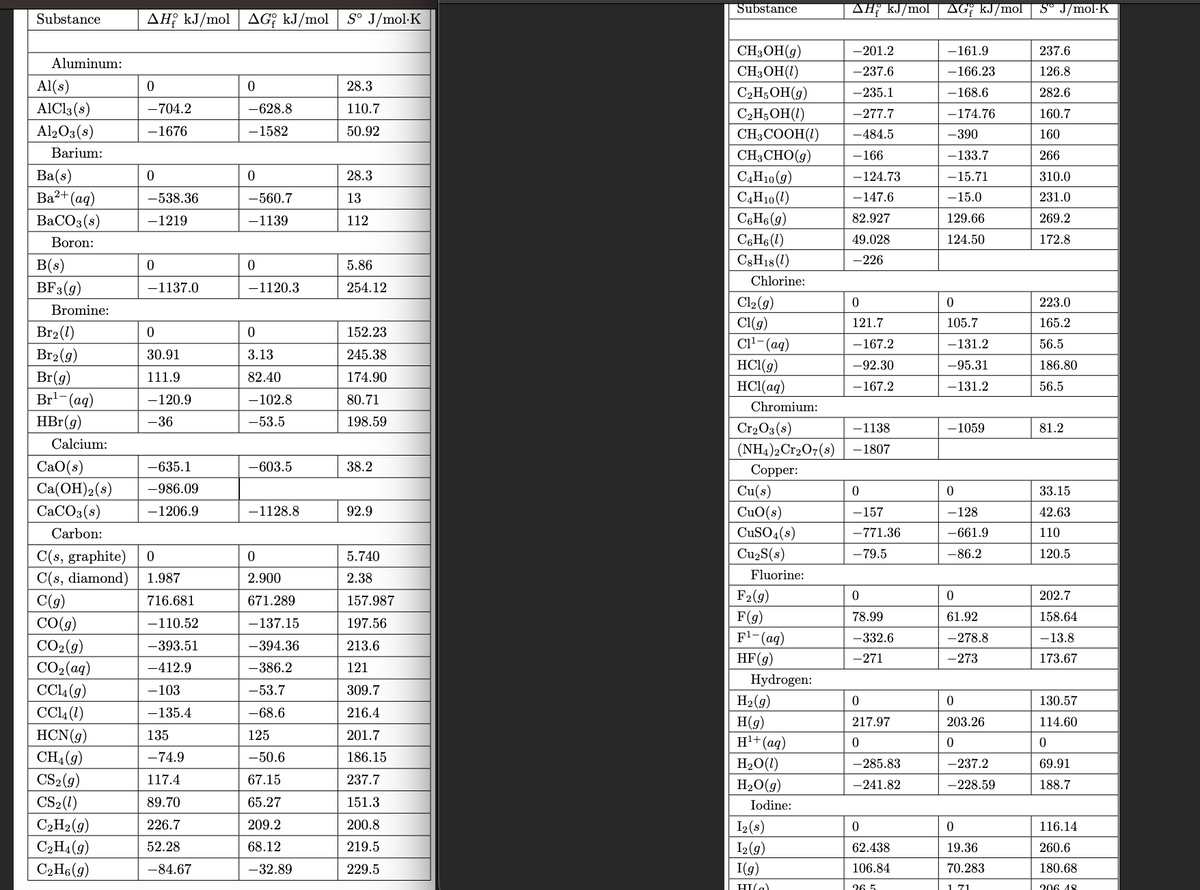
Transcribed Image Text:Substance
Aluminum:
Al(s)
AlCl3(s)
Al2O3(s)
Barium:
Ba(s)
Ba²+ (aq)
BaCO3(s)
Boron:
B(s)
BF3 (9)
Bromine:
Br₂ (1)
Br₂(g)
Br(g)
Br¹- (aq)
HBr (g)
Calcium:
CaO(s)
Ca(OH)2 (s)
CaCO3(s)
Carbon:
CC14 (9)
CC14 (1)
HCN (g)
CH4 (9)
CS₂ (g)
CS₂ (1)
C₂H₂(g)
AH kJ/mol AG kJ/mol
C₂H₁ (9)
C₂H6 (9)
0
-704.2
-1676
0
-538.36
-1219
0
-1137.0
0
30.91
111.9
-120.9
-36
C(s, graphite) 0
C(s, diamond) 1.987
C(g)
716.681
CO(g)
-110.52
CO₂(g)
-393.51
CO2(aq)
-412.9
-103
-135.4
-635.1
-986.09
-1206.9
135
-74.9
117.4
89.70
226.7
52.28
-84.67
0
-628.8
-1582
0
-560.7
-1139
0
-1120.3
0
3.13
82.40
-102.8
-53.5
-603.5
-1128.8
0
2.900
671.289
-137.15
-394.36
-386.2
-53.7
-68.6
125
-50.6
67.15
65.27
209.2
68.12
-32.89
Sº J/mol K
28.3
110.7
50.92
28.3
13
112
5.86
254.12
152.23
245.38
174.90
80.71
198.59
38.2
92.9
5.740
2.38
157.987
197.56
213.6
121
309.7
216.4
201.7
186.15
237.7
151.3
200.8
219.5
229.5
Substance
CH3OH(g)
CH3OH(1)
C₂H5OH (9)
C₂H5OH (1)
CH3COOH (1)
CH3CHO(g)
C4H10 (9)
C4H10 (1)
C6H6 (9)
C6H6 (1)
C8H18 (1)
Chlorine:
Cl₂ (g)
Cl(g)
Cl¹- (aq)
HCl(g)
HCl(aq)
Chromium:
Cr₂O3(s)
(NH4)2Cr2O7(s)
Copper:
Cu(s)
CuO(s)
CuSO4(s)
Cu₂S(s)
Fluorine:
F₂(g)
F(g)
F¹-(aq)
HF (g)
Hydrogen:
H₂(g)
H(g)
H¹+ (aq)
H₂O(l)
H₂O(g)
Iodine:
I₂ (s)
I₂ (g)
I(g)
HI(₂)
AH kJ/mol
-201.2
-237.6
-235.1
-277.7
-484.5
-166
-124.73
-147.6
82.927
49.028
-226
0
121.7
-167.2
-92.30
-167.2
-1138
-1807
0
-157
-771.36
-79.5
0
78.99
-332.6
-271
0
217.97
0
-285.83
-241.82
0
62.438
106.84
26.5
AG kJ/mol S° J/mol K
-161.9
-166.23
-168.6
-174.76
-390
-133.7
-15.71
-15.0
129.66
124.50
0
105.7
-131.2
-95.31
-131.2
-1059
0
-128
-661.9
-86.2
0
61.92
-278.8
-273
0
203.26
0
-237.2
-228.59
0
19.36
70.283
1 71
237.6
126.8
282.6
160.7
160
266
310.0
231.0
269.2
172.8
223.0
165.2
56.5
186.80
56.5
81.2
33.15
42.63
110
120.5
202.7
158.64
-13.8
173.67
130.57
114.60
0
69.91
188.7
116.14
260.6
180.68
206 18
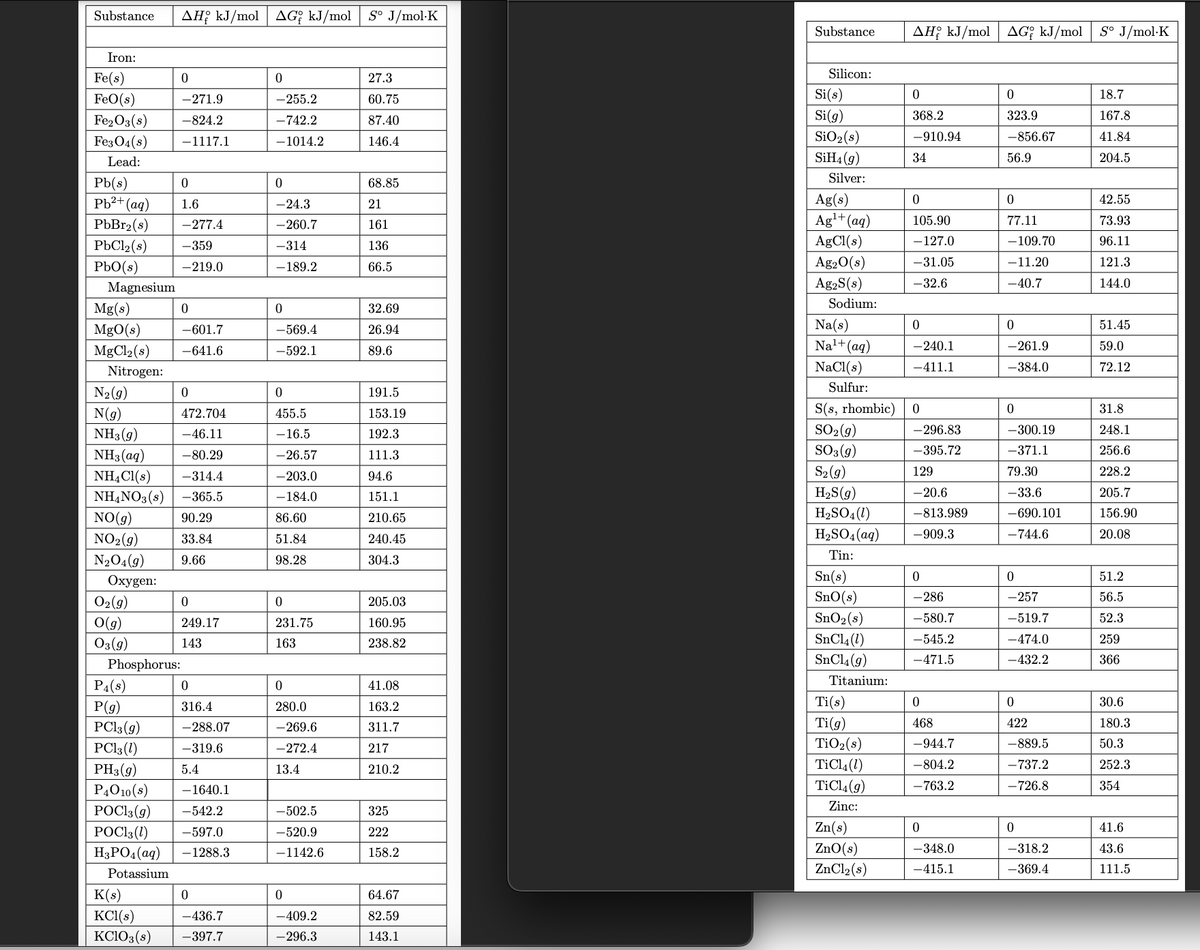
Transcribed Image Text:Substance AH kJ/mol AG kJ/mol S° J/mol.K
Iron:
Fe(s)
FeO (s)
Fe₂O3(s)
Fe3O4(s)
Lead:
Pb(s)
Pb²+ (aq)
PbBr₂ (s)
PbCl₂ (s)
PbO(s)
Magnesium
Mg(s)
MgO(s)
MgCl₂ (s)
Nitrogen:
Oxygen:
O2(g)
O(g)
03(g)
P4(s)
P(g)
PC13 (9)
PC13 (1)
N₂(g)
N(g)
NH3(g)
NH3(aq)
-80.29
NH4Cl(s) -314.4
NH4NO3(s) -365.5
NO(g)
90.29
NO2(g)
33.84
N₂O4(9)
9.66
PH3(g)
P4010(s)
0
POC13 (9)
POC13 (1)
H3PO4 (aq)
-271.9
-824.2
-1117.1
Potassium
0
1.6
-277.4
-359
-219.0
0
-601.7
-641.6
Phosphorus:
0
472.704
-46.11
0
249.17
143
0
316.4
-288.07
-319.6
5.4
-1640.1
-542.2
-597.0
-1288.3
0
K(s)
KCl(s)
-436.7
KClO3(s) -397.7
0
-255.2
-742.2
-1014.2
0
-24.3
-260.7
-314
-189.2
0
-569.4
-592.1
0
455.5
-16.5
-26.57
-203.0
-184.0
86.60
51.84
98.28
0
231.75
163
0
280.0
-269.6
-272.4
13.4
-502.5
-520.9
-1142.6
0
-409.2
-296.3
27.3
60.75
87.40
146.4
68.85
21
161
136
66.5
32.69
26.94
89.6
191.5
153.19
192.3
111.3
94.6
151.1
210.65
240.45
304.3
205.03
160.95
238.82
41.08
163.2
311.7
217
210.2
325
222
158.2
64.67
82.59
143.1
Substance
Silicon:
Si(s)
Si(g)
SiO₂ (s)
SiH4 (9)
Silver:
Ag(s)
Ag¹+ (aq)
AgCl(s)
Ag₂O(s)
Ag2S (s)
Sodium:
Na(s)
Na¹ + (aq)
NaCl(s)
Sulfur:
S(s, rhombic)
SO₂(g)
SO3(g)
S2(g)
H₂S (g)
H₂SO4 (1)
H₂SO4 (aq)
Tin:
Sn(s)
SnO(s)
SnO₂ (s)
SnCl4 (1)
SnCl4 (9)
Titanium:
Ti(s)
Ti(g)
TiO₂ (s)
TiCl4 (1)
TiCl4 (9)
Zinc:
Zn(s)
ZnO(s)
ZnCl₂ (s)
AH kJ/mol AG kJ/mol
0
368.2
-910.94
34
0
105.90
-127.0
-31.05
-32.6
0
-240.1
-411.1
0
- 296.83
-395.72
129
-20.6
-813.989
-909.3
0
-286
-580.7
-545.2
-471.5
0
468
-944.7
-804.2
-763.2
0
-348.0
-415.1
0
323.9
-856.67
56.9
0
77.11
-109.70
-11.20
-40.7
0
-261.9
-384.0
0
-300.19
-371.1
79.30
-33.6
-690.101
-744.6
0
-257
-519.7
-474.0
-432.2
0
422
-889.5
-737.2
-726.8
0
-318.2
-369.4
Sº J/mol K
18.7
167.8
41.84
204.5
42.55
73.93
96.11
121.3
144.0
51.45
59.0
72.12
31.8
248.1
256.6
228.2
205.7
156.90
20.08
51.2
56.5
52.3
259
366
30.6
180.3
50.3
252.3
354
41.6
43.6
111.5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning