Q: Calculate pAl when 25 mL of 0.0274 M Al3+ solution was titrated with 28 mL 0.0180 M EDTA at pH 5.…
A: 25 mL of 0.0274 M Al3+ solution was titrated with 28 mL of 0.0180 M EDTA at pH 5 .log Kf = 16.4…
Q: Give the periodic group number and number of valence electrons for each of the following atoms. a. S…
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: ✓ The molecular formula of this molecule is NH
A: chemical formula gives the information about the total number of atoms of each element in the…
Q: QUESTION 7 Give the structure of the product of the following reaction: acid catalyst H₂C=CH₂ + H₂O…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Practice problems: Propose reasonable mechanisms for the following reactions. Use arrows correctly…
A: Detailed mechanism is provided for each question
Q: What is the result of heating cobalt (II) chloride hexahydrate? Group of answer choices The cobalt…
A: On heating cobalt(II) chloride hexahydrate, it decomposes into cobalt chloride and water. Reaction…
Q: Identify the letters that correspond to the C atoms that would yield the most number of products in…
A:
Q: write the net ionic equation for lithium plus water.
A:
Q: Triglycerides are formed when the fatty acids react with the alcohols of the glycerol to form as…
A: 19)•In triglycerides alcohol of glycerol and acid of fatty acid are meet and form ester functional…
Q: The reaction H2 + I2 → 2HI takes place in the gas phase, is 2nd order, the constant its speed is…
A: Given, The order of the reaction is second order and for the second order reaction rate law will be,…
Q: 2. For each of these equations, determine whether it is a redox re- action. For each redox reaction,…
A: #2: In a redox reaction, the reactants undergo both oxidation and reduction to form the products.…
Q: When copper is heated in air, copper(II) oxide is formed. In a given experiment, 3.12 g copper was…
A:
Q: The following Lewis diagram represents the valence electron configuration of a main-group element.…
A: Valence electrons The number of electrons present in the outermost shell of the atom are called as…
Q: a laccd sign in - Sear X tab caps lock shift 1 A chemist determines by measurements that 0.080 moles…
A: Given, The moles of Iodine solid precipitates in the chemical reaction = 0.080 mole. The mass of…
Q: 14) Do a retrosynthetic analysis for the following target molecule. Identify the synthetic…
A:
Q: What is the pH of a 0.130 M solution of HNO₃?b
A:
Q: d. Draw the elimination product that would form upon reaction of (4S)-1-bromo-4- methylhexane with…
A: The reactant used is (4S)-1-bromo-4-methylhexane. The reagents used are (CH3)3COH and (CH3)3COK.
Q: How many grams of sodium hydroxide is required to make 250.0 mL of a 0.410 M sodium hydroxide…
A: Given, The molarity of sodium hydroxide ( NaOH) = 0.410 M. The volume of the solution = 250.0 mL.…
Q: 27. (10) Write a reasonable mechanism for the following transformation. H₂SO4 OH OH 28 + Predict the…
A: Here we have to write the mechanism of the following given reactions. Here the first reaction is…
Q: For which K= 130.0.Assume that 0.406 mol C (g) is placed in a cylinder. The Temperature is 300.0…
A: The Given reaction is Ag+Bg→Cg; The K is 130mol
Q: Which solution, if any, would create the higher osmotic pressure (compared to pure water): one…
A: In the given question two types of solution is prepared. One is 2 g of LiBr prepared in 40 ml water…
Q: What is the mole-to-mole ratio between aluminum ion and potassium aluminum sulfate dodecahydrate?…
A: We need to determine moles of Al present in 1 mol of potassium aluminum sulfate dodecahydrate.
Q: MISSED THIS? Read Section 6.5 (Pages 225-228); Watch IWE 6.8. A sample of gas has a mass of 0.555 g…
A: Given data P = pressure of the gas = 734 mm Hg =734760= 0.966 atm ( because 1 atm = 760 atm) V =…
Q: Which substrate concentration range is best for determining the catalytic efficiency of an enzyme…
A: In enzyme catalyzed reaction the catalytic effeciency is defined as how efficiently enzyme converts…
Q: 2. What is the molecular formula of your mononitrated reaction product? 8. Based on all of the data…
A: Benzene undergo Electrophilic substitution reactions in which H of benzene ring is replaced by…
Q: Why are multiple heating of the potassium chlorate necessary? Group of answer choices To make sure…
A: We ate to discuss why slow/multiple heating of potassium chloraye is necessary.
Q: Hydrated coppper (II) sulfate is heated, how do you know when the dehydration is complete? Group of…
A:
Q: Datasheet A. Preparation of Aspirin A.1 Mass of flask A.2 Mass of flask and salicylic acid Mass of…
A:
Q: Rank the following aqueous solutions in order of decreasing freezing point. Select 1 (highest…
A:
Q: A steel container of volume 0.40 L can withstand pressures up to 88 atm before exploding. Part A…
A: Given, Volume(V) = 0.40 L Pressure(P) = 88 atm Temperature(T) = 295 K R is the ideal gas constant…
Q: Name the major product of the following reaction: H H3C- CH3 CH3 + HCI
A:
Q: heat [4+2] cycloaddition
A: We have given pericyclic reaction which is carried out under thermal condition. Given reaction is…
Q: (NH4)2S(aq) + SrCl₂(aq) - Express your answer as a chemical equation. Enter NOREACTION if no…
A: Given reactions are examples of double displacement reaction . In double displacement reaction…
Q: CHEMICAL REACTIONS Calculating and using the molar mass of diatomic elements A chemist measures the…
A:
Q: The carbon dioxide pressure in a bottle of champagne is about 7 atm. At this pressure about 0.65 g…
A:
Q: What is occurring in section C? B C temperature (°C) forening D BONG Cat time (minutes) OA.…
A: We are given a Temperature(oC) versus Time(min) graph for the conversion of an unknown material from…
Q: -3 The osmotic pressure of 8.46 x 10 M solutions of CaCl₂ and urea at 25 °C are 0.511 atm and 0.207…
A:
Q: What are the structures of the products below? Type the letter labels of your answers. Type NONE if…
A:
Q: Enter the complete ionic equation to show the reaction of aqueous lead(II) nitrate with aqueous…
A: Given that, there is a reaction between aqueous lead(ll) nitrate (Pb(NO3)2 with aqueous potassium…
Q: 1 Charles’s law states that the volume of a gas is directly related to the absolute temperature when…
A: Given, V1= 1.18 × 103 L T1 = 30℃ = (30 + 273.15 ) =…
Q: Galaulate the PH and the Pott FOR 9.6M solution OF MH₂ инз K. FOR NH3=1.97105
A: Here we are required to find the pOH and pH of 0.6M of ammonia solution
Q: Draw the product(s) of the following reactions. CH3CH₂CH₂-CEC-CH₂CH₂CH3 1. BH3/THF 2. H₂O₂ / aqueous…
A: This is hydroboration-oxidation reaction of alkynes. Here in the reaction hydroboration and…
Q: Using the thermodynamic information in the ALEKS Data tab, calculate the standard reaction free…
A: Given reaction is : 2Al (s) + Fe2O3 (s) ------> Al2O3 (s) + 2Fe (s) Calculate the standard change…
Q: Use appropriate data to determine which statement is true of the voltaic cell picture here.
A:
Q: What is the concentration of citric acid H3C6H5O7 in a 0.400 M solution of citric acid? please…
A: •Concentration:-It is amount of solute dissolved in given amount of solvent or solution. •We use…
Q: 5. Calculate the number of oxygen atoms present in 755.0 mg of nitrogendioxide gas.
A:
Q: Show how to convert bromocyclohexane to each of the compounds shown below. ol H (a) (b) CH3 N (c) H
A:
Q: What is the sum of the coefficients of reactants and products for the dehydration equation of nickel…
A: Given, The sum of the coefficients of products and reactants in the dehydration equation of nickel…
Q: 3. How to covert the reactant to the product? Show all reagents and intermediates involved in the…
A: Ethyl acetoacetate contain ester and keto group making methylene hydrogen acidic.In presence of base…
Q: Question 10 Identify the organic class to which the compound belongs based on the functional groups…
A: functional group are those species which defines the physical and chemical properties of a given…
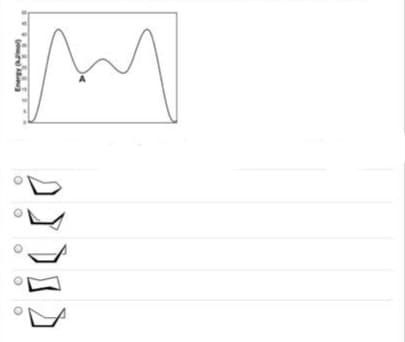
Give a clear handwritten answer with explanation...give below a energy diagram..choose the what represent energy point A in the diagram..give detailed answer
Step by step
Solved in 2 steps with 1 images

- How does the coordination geometry of a metal center influence its reactivity in bioinorganic chemistry? Provide an example to illustrate your answer. asap give correctly or else downvoteIn the case of ligand substitution at square planar platinum(II), which is the stronger trans effect ligand: chloride, or iodideThe flowing compound is the amino acid glycine that contains glycinate bidentate chelating group since it contains two ligand groups amine and carboxyl group.

