Q: Andy and his college friends decided to go on a summer vacation after a long and stressful final…
A: Select the correct answer :
Q: Which of these two contains a stereocenter? Draw the structure on your answer sheet and mark the…
A: From the given two structures : The one which has stereocenter = ?
Q: What is the effect of adding salt to the ice cubes? Select the correct response: It decreases the…
A: The melting point of a substance is the temperature at which it changes state from solid to liquid.
Q: Which of the following will show a Tyndall Effect? Select the correct response: Aqueous solution of…
A: Tyndall effect is an optical property of a colloidal solution. When a beam of light passed through a…
Q: How many grams of pure sodium carbonate are needed to standardize a 0.20 N HCl when you expect the…
A: Given that - Normality of HCl = 0.20 N Volume of HCl used = 25.0 mL Then mass of HCl can be…
Q: A Zn electrode for Zn2+ analysis is what type of indicator electrode? a. glass electrode b.…
A: Answer: c metallic electrode of the first kind.
Q: Which of the following choices is a diastereomer of the first structure shown? OH OH O OH NH₂ NH₂ NH…
A:
Q: 200-2.5 acid
A: According to Bartleby guidelines, we're supposed to answer first question among non related type of…
Q: In which equation(s) do/does water act as a Brønsted-Lowry base? You may choose one or more…
A: According to Bronsted-Lowry theory the substance which donate proton (H+ ) is acid and which accept…
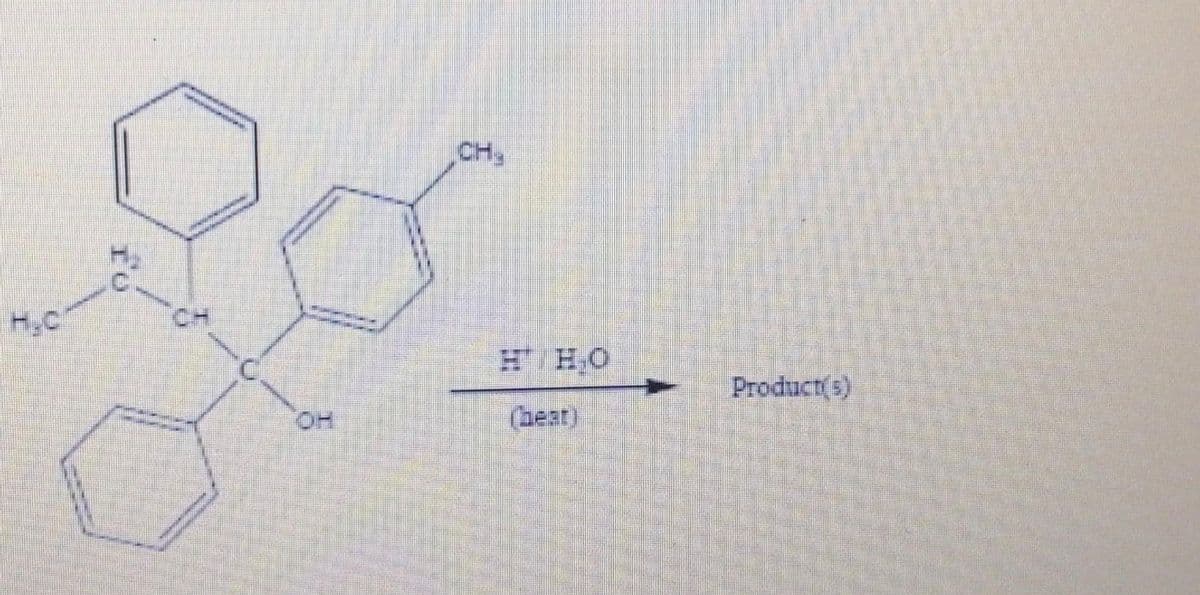
Q: 06. Predict the major product(s) of the following reactions. Show stereochemistry where applicable.…
A:
Q: Using the following data determine the reaction order and rate constant for the reaction: H2CO3(aq)…
A: The rate law expression for nth order reaction may be written as: Rate=k[Reactant]n
Q: Metals A,B and C have a standard reduction potentials of +0.68V, -1.48V and -0.80V. Which…
A: Standard reduction potential is given for three cells A B and C we have to find the combination…
Q: Hex-1-ene gas, C6H12, burns in oxygen to produce carbon dioxide and water vapour. a. Write an…
A:
Q: ● Why is the reaction with acetaminophen regiospecific?
A: As multiple questions are given, I am allowed to answer one question at a time. Please re upload the…
Q: 1. Illustrate and explain how the compound below can be used to produce different alkenes.
A: In this question, we will illustrating and explaining how the given compound ( which have alkyne…
Q: 10. Give the structure corresponding to each name. a. 7,7-dimethyl-4-octanol b.…
A:
Q: Why is it necessary to know the concentration of the solutions? Select the correct response: To…
A: Concentration of any solution is defined as the amount of solute dissolved in a particular amount of…
Q: 2 A student determined the concentration of a weak acid, acetic acid, by a titrimetric method using…
A: A question based tools in analytical chemistry that is to be accomplished.
Q: Write (+) if the compound would test positive in the following qualitative tests and (-) if not.
A: given Write (+) if the compound would test positive in the following qualitative tests and (-) if…
Q: CH3 CH3 心心 ·Br Cl Br CH3 Br d, C. H3C H Br H OH CH3 CH3 CH3 e. 士人人 CI CI- ||||| Cl HO CH3 CH3 CH3 a.…
A: ->If configuration is reversed these are enantiomer . ->If configuration is same then these…
Q: 1. Answer BOTH parts a) and b). a) i) Explain the difference between spin-lattice relaxation (T₁)…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: PROBLEM 3: Given the following half reaction: a. 1₂lt b. Pt| Fe,Fe+3 c. ZnZn¹2 d. Ag | Ag For each…
A: a) The following are the separate two half-cell balanced reactions for the given redox reactions: i)…
Q: Question 42 1 A 150-mL sample of gas exerts a pressure of 4.20 atm at 20.0 °C. What volume would…
A: Here we are required to find the volume of gas sample at 100°C and 4 atm pressure .
Q: What gas law best explains the explosion of the heated aerosol container? Select the correct…
A:
Q: Pressure in atm 217.75 (Critical pressure) 1.00 0.0060 D Phase Diagram for Water C Normal freezing…
A:
Q: 2. A mixture containing only BaSO4 (FW 233.38) and CaSO4 (FW 136.14) contains one half as much Ba2+…
A: In the given question we have to calculate the percentage of CaSO4 in the given mixture. Given…
Q: d. o-cresol and anisole e. benyl alcohol and anisole
A: There are various specific reaction which only shows by some specific compounds only.
Q: Which of the following conditions will result to MnO4² as product for the reaction between MnO4 and…
A:
Q: How many milliliters of water can be added to 5 mL of 12 M HCl solution to make a 3 M HCl solution?
A: Molarity is defined as number of moles of solute divide by volume of solution
Q: Q.20 SINGLE CHOICE CH3 I H₂O/H+ H₂C O 18 CH3 Where A and B are, respectively, A O 18 H₂C and Me,C-OH…
A: ->Acidic hydrolysis of ester give acid and Alcohol as product .
Q: Which of the following is NOT a guideline in writing a good recommendation for a research paper?…
A: A research paper is written by a researcher in which he explain what he had learned after exploring…
Q: Find the connection among the words below and choose the letter of the word which is different.* 1.…
A: Two questions based on adsorption that is to be accomplished.
Q: W h i c h p a r a m e t e r i s u s e d t o d e t e r m i n e t h e a n a l y t e c…
A: The UV-visible electrophotometric can be used to determine the analyte concentration by plotting…
Q: How do we define the concentration of a solution? Select the correct response: Predict the amount of…
A: We have to find correct options.
Q: Question 17 Which of the following substances has the HIGHEST melting point? Select one: O a. C₂H5OH
A: Melting point is the temperature at which substance melts to form liquid.
Q: 3 A 3.00 mol sample of an ideal diatomic gas expands adiabatically from a volume of 0.1610 m³ to…
A: Using ideal gas equation, PV=nRT Initial conditions,P1=1 atmV1=0.1610 m3=161 Ln=3…
Q: C. Determine the strongest intermolecular force experienced by each of the following substances…
A: Polar Molecules take part in dipole-dipole forces. Non-polar molecules like CO2, Cl2, Br2 have…
Q: Derive the expression for calculating the isothermal compressibility of a certain gas that obeys the…
A:
Q: Draw 2 resonance structures for the following: Upload answer as jpeg or pdf file only. Make sure you…
A: Here we are required to draw the resonating structures of the given compound
Q: A gaseous mixture of methane, ethane and propane has their percent volume of 38%, 42% and 20%…
A:
Q: Br 1. Li 2. Cul S
A:
Q: What part of research refers to wrapping up of ideas and leaving a strong final impression on the…
A: Research generally addresses a particular problem and probable solutions to the problem. While…
Q: 1. Draw 3 isomers of C7H12O2 then state where you will dispose it (the kind of waste disposal should…
A: Given in following question a compound C7H12O2 draw given compound 3 isomers where you will dispose…
Q: A sample of pure sodium oxalate weighing 0.1050 g is ignited [Na2C2O4 (s) --> Na2CO3 (s) + CO (g)]…
A:
Q: In the chemical formula Mg(NO3)2, the numerical coefficient is equal to…
A: Chemical formula are representation of molecules or compounds by fixed proportions of elements in…
Q: 4. A mixture containing only BaO (FW 153.33) and CaO (FW 56.08) weighs 2.00g. The oxides are…
A:
Q: A student has synthesised propyl acetate and collected spectroscopic evidence for a successful…
A:
Q: Given reaction rate date for: 2NO(g) + Br₂(g) Trial [NO] (M) 1 0.20 2 0.20 3 0.30 2NOBr(g) [Br2] (M)…
A:
Q: Moving to another question will save this stion 9 What is the theoretical efficiency of an engine…
A:
Q: Identify the functional groups in the given org CH 3 CH 3 H3C CH3 ****** OH CH 3 Select the correct…
A:
Please give. ASAP

Step by step
Solved in 2 steps with 3 images


