Given the reaction below has AH = -118.9 kJ, which one of the following actions will cause an increase in the equilibrium concentration of NO2 in a closed reaction chamber? 2 NO (g) + O₂ (g) = 2 NO₂ (g) adding O₂ (g) to the system increasing the volume of the system while temperature is kept constant increasing the temperature of the system adding a catalyst removing O₂ (g) from the system
Given the reaction below has AH = -118.9 kJ, which one of the following actions will cause an increase in the equilibrium concentration of NO2 in a closed reaction chamber? 2 NO (g) + O₂ (g) = 2 NO₂ (g) adding O₂ (g) to the system increasing the volume of the system while temperature is kept constant increasing the temperature of the system adding a catalyst removing O₂ (g) from the system
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter16: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 101CWP
Related questions
Question
Answer and Explain
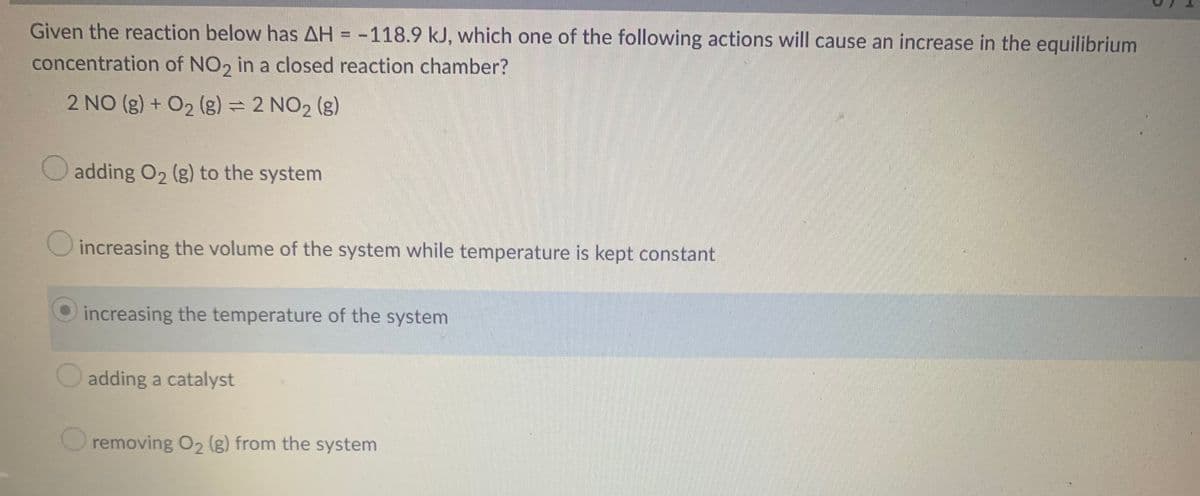
Transcribed Image Text:Given the reaction below has AH = -118.9 kJ, which one of the following actions will cause an increase in the equilibrium
concentration of NO2 in a closed reaction chamber?
2 NO(g) + O₂ (g) = 2 NO₂ (g)
adding O₂ (g) to the system
increasing the volume of the system while temperature is kept constant
increasing the temperature of the system
adding a catalyst
removing O₂ (g) from the system
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning